Overview - The Metabolic Metro Map
- Metabolism is a network of pathways linked by shared intermediates.
- Key Junctions: Glucose-6-Phosphate (G6P), Pyruvate, and Acetyl-CoA act as metabolic traffic circles.
- These hubs direct substrates towards energy production (catabolism) or storage/synthesis (anabolism) based on the cell's energy state.
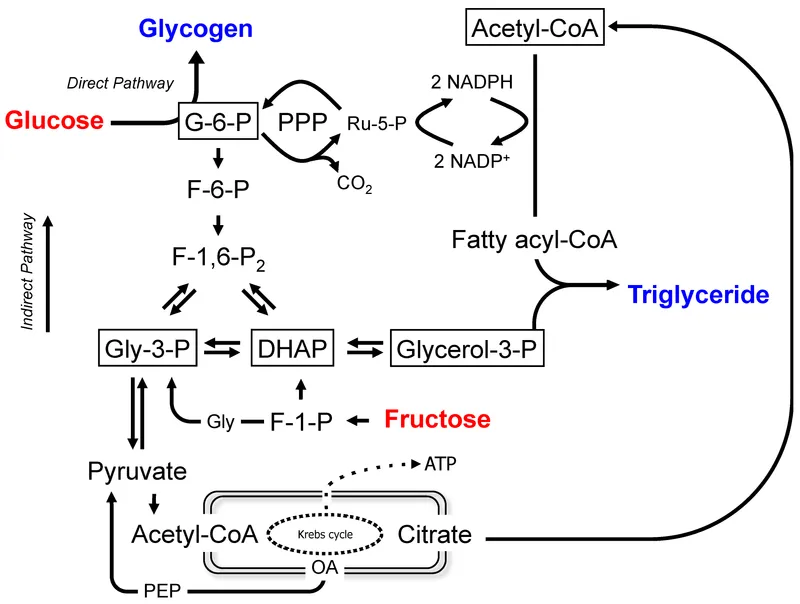
⭐ The Pyruvate Dehydrogenase Complex reaction (Pyruvate → Acetyl-CoA) is irreversible. This is why fatty acids (which yield Acetyl-CoA) cannot be used for net glucose synthesis.
Fed State - Post-Meal Power-Up
- Primary Driver: High insulin-to-glucagon ratio promotes anabolism.
- Main Fuel: Glucose.
- Metabolic Priorities: Use glucose, store the excess.
- Liver & Muscle: ↑ Glycolysis & ↑ Glycogenesis (storage as glycogen).
- Adipose Tissue: ↑ Glucose uptake for glycerol synthesis; ↑ Lipoprotein Lipase (LPL) activity to store dietary fats as triacylglycerols (TAGs).
- Protein: ↑ Amino acid uptake and protein synthesis.
⭐ GLUT4 is the key! This insulin-responsive transporter moves to the membrane in muscle and adipose tissue to facilitate glucose uptake.
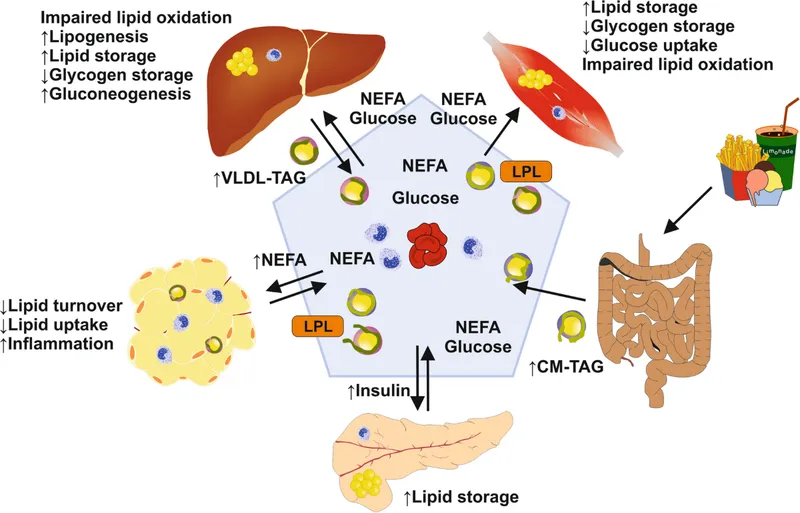
Fasting State - Fasting Fuel Frenzy
- Hormonal Profile: ↓ Insulin, ↑ Glucagon, ↑ Epinephrine.
- Primary Goal: Maintain euglycemia for brain and RBCs.
- Liver Metabolism:
- Glycogenolysis: Initial glucose source (first 12-18 hours).
- Gluconeogenesis: Becomes primary source later. Key substrates: lactate, alanine, and glycerol.
- Adipose Tissue:
- Lipolysis: Hormone-sensitive lipase breaks down triglycerides into fatty acids and glycerol.
- Fuel Utilization:
- Muscle/Liver: Switch to fatty acid oxidation.
- Brain: Utilizes glucose, then ketone bodies after prolonged fasting.
⭐ Alanine is the principal amino acid shuttle from muscle to liver for gluconeogenesis (Cahill Cycle).
Starvation State - Ketone Survival Mode
- Timeline: Prolonged fasting (> 3 days).
- Primary Goal: Muscle protein-sparing by ↓ gluconeogenesis.
- Liver Metabolism:
- ↑ Fatty acid oxidation produces excess Acetyl-CoA.
- Acetyl-CoA is converted to ketone bodies (β-hydroxybutyrate & acetoacetate).
- Brain Fuel Shift: Adapts to use ketone bodies as its main fuel. This spares glucose for obligate tissues like RBCs.
⭐ The brain derives up to 75% of its energy from ketone bodies during prolonged starvation, a key adaptation to spare essential proteins.
Organ-Specific Metabolism - The Body's Orchestra
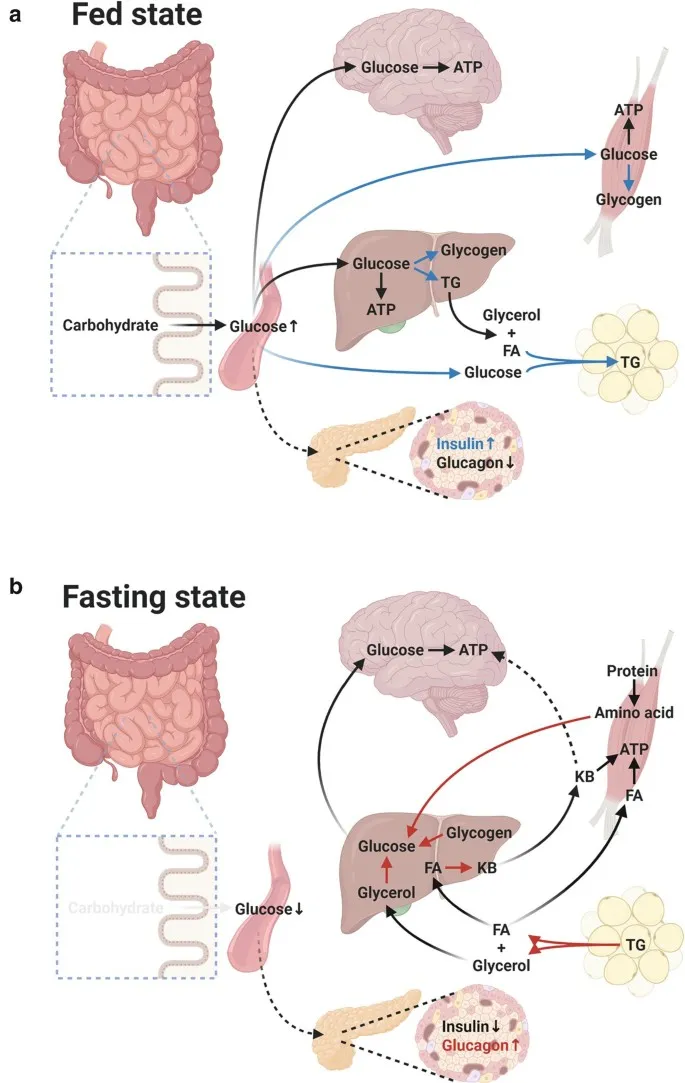
- Liver: The central metabolic processor.
- Fed: Glycolysis, glycogenesis, lipogenesis.
- Fasting: Gluconeogenesis (primary source), glycogenolysis, ketogenesis, urea cycle.
- Skeletal Muscle: Major glucose user & storage site.
- Fed: Glucose uptake (GLUT4) & glycogen storage.
- Fasting: Switches to fatty acid oxidation. Exports lactate (Cori cycle) & alanine (Cahill cycle).
- Brain: High, constant energy demand.
- Uses glucose almost exclusively (~120g/day).
- Adapts to use ketone bodies in prolonged starvation.
- Adipose Tissue: Body's main energy reservoir.
- Fed: Stores triglycerides.
- Fasting: Releases free fatty acids & glycerol via lipolysis.
⭐ The brain's absolute requirement for glucose drives the body's gluconeogenic and ketogenic responses during fasting.
- Insulin (anabolic, fed state) and glucagon (catabolic, fasting state) are the primary hormonal regulators of metabolism.
- Acetyl-CoA is the central metabolic hub, linking carbohydrate, fat, and protein breakdown.
- The liver is the key organ for maintaining blood glucose homeostasis through gluconeogenesis and glycogenolysis.
- The brain depends on glucose but adapts to use ketone bodies during prolonged starvation.
- Muscle utilizes glucose, fatty acids, and ketones, releasing alanine and lactate during exertion.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

