SLP Fundamentals - The Direct Deposit
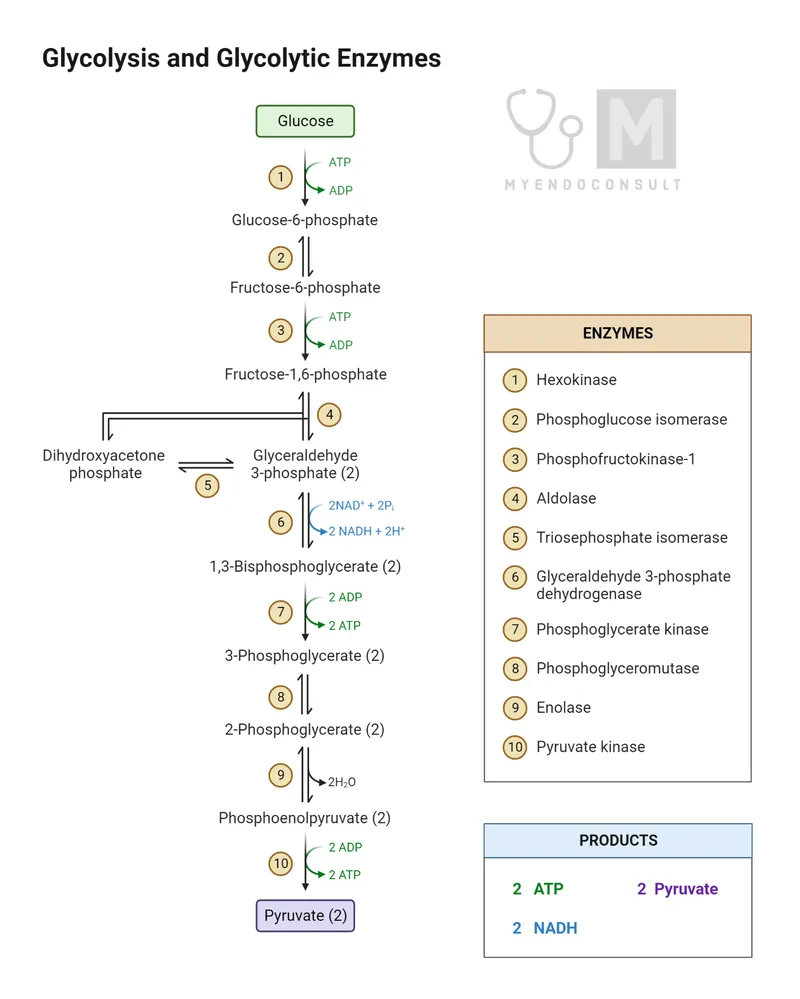
Substrate-level phosphorylation (SLP) is the direct enzymatic transfer of a phosphate group ($P_i$) from a substrate to ADP, forming ATP. This contrasts with oxidative phosphorylation, which uses a proton gradient and ATP synthase. In glycolysis, SLP is the only method of ATP synthesis.
- Step 7: 1,3-Bisphosphoglycerate → 3-Phosphoglycerate
- Enzyme: Phosphoglycerate Kinase
- Yield: 2 ATP (as 2 G3P molecules are processed)
- Step 10: Phosphoenolpyruvate (PEP) → Pyruvate
- Enzyme: Pyruvate Kinase
- Yield: 2 ATP
⭐ Arsenate (arsenic poison) can prevent net ATP synthesis in glycolysis by competing with phosphate for binding to glyceraldehyde-3-phosphate, forming an unstable compound that hydrolyzes without producing ATP at the phosphoglycerate kinase step.
Key Reactions & Enzymes - The Money Makers
Substrate-level phosphorylation is the direct transfer of a phosphate group from a substrate to ADP, forming ATP. In glycolysis, this occurs at two key steps, each happening twice per glucose molecule, yielding the "payoff."
- Reaction 1: 1,3-Bisphosphoglycerate → 3-Phosphoglycerate
- Reaction 2: Phosphoenolpyruvate (PEP) → Pyruvate
| Feature | Reaction 1 | Reaction 2 |
|---|---|---|
| Enzyme | Phosphoglycerate Kinase (PGK) | Pyruvate Kinase (PK) |
| Substrate | 1,3-Bisphosphoglycerate | Phosphoenolpyruvate (PEP) |
| Product | 3-Phosphoglycerate | Pyruvate |
| ATP Yield | 2 ATP (per glucose) | 2 ATP (per glucose) |
⭐ Exam Favorite: Pyruvate Kinase deficiency is an autosomal recessive disorder causing chronic hemolytic anemia. RBCs depend solely on glycolysis for ATP; without sufficient PK, they cannot maintain the Na+/K+ pump, leading to cell swelling and lysis.
Pathway Regulation - Controlling the Cashflow
- Pyruvate Kinase (PK): The final, irreversible, and highly regulated step of glycolysis.
- Feed-Forward Activation: An ↑ in Fructose-1,6-bisphosphate (from the PFK-1 step) signals substrate availability and allosterically activates PK.
- Inhibition (Energy is High):
- ↑ ATP (allosteric)
- ↑ Alanine (allosteric, signals building blocks are plentiful)
⭐ Tissue-Specific Regulation: In the liver, glucagon activates PKA, which phosphorylates and inactivates PK. This conserves glucose during fasting. Muscle PK is not regulated by this mechanism.
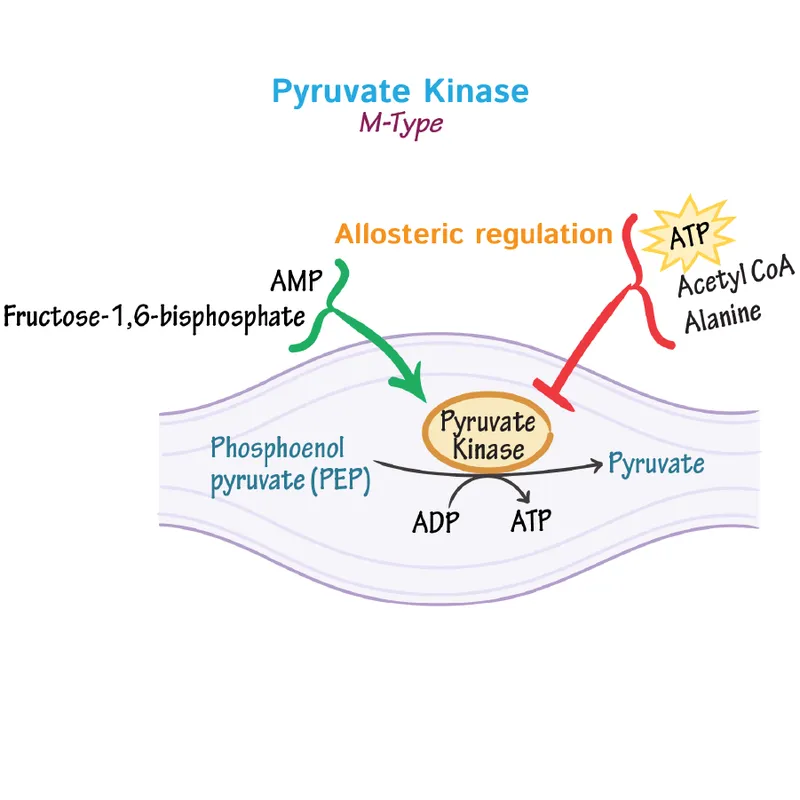
📌 Mnemonic: ATP & Alanine Arrest PK activity.
Clinical Correlates - When the ATM Breaks
- Pyruvate Kinase (PK) Deficiency: An autosomal recessive disorder causing ↓ ATP production in RBCs.
- Pathophysiology: ↓ ATP leads to RBC membrane instability, resulting in chronic extravascular hemolysis.
- Clinical Findings:
- Presents in newborns with hemolytic anemia, jaundice, and splenomegaly.
- Labs: Normocytic anemia, ↑ reticulocytes, ↑ unconjugated bilirubin.
- Peripheral smear shows burr cells (echinocytes).
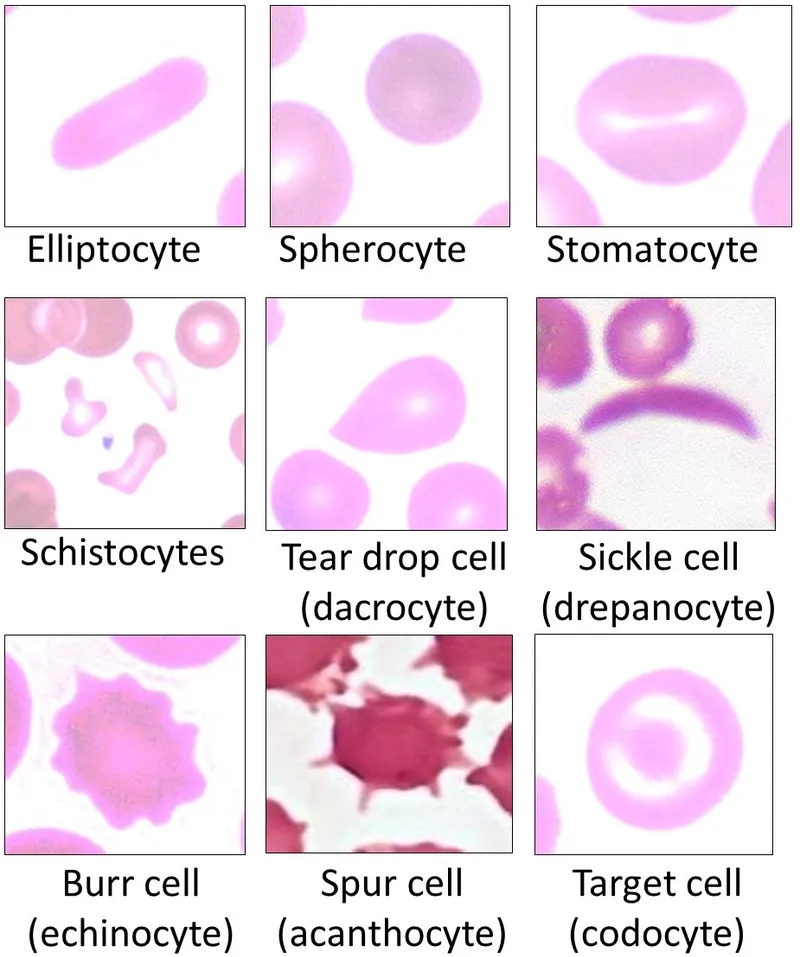
⭐ Unlike G6PD deficiency, hemolysis is chronic and no Heinz bodies are formed, as the HMP shunt is intact.
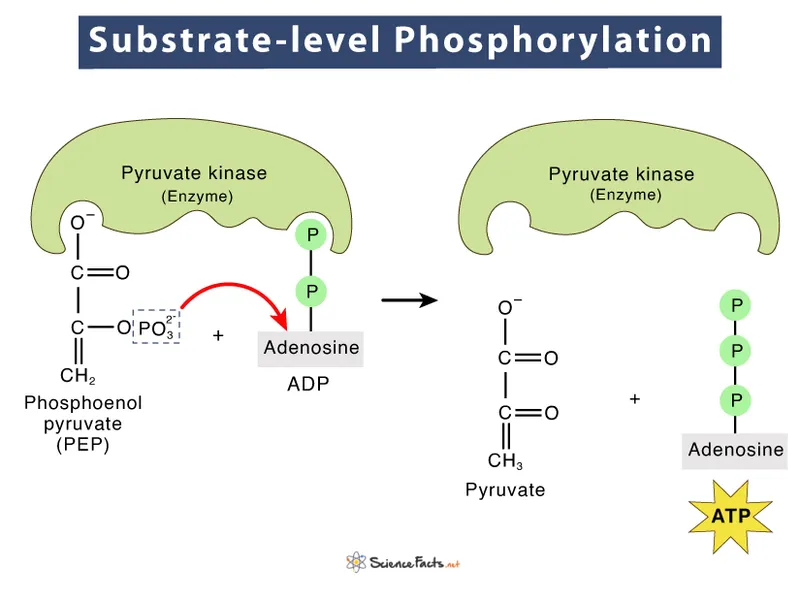
- Direct ATP synthesis where a phosphate group is transferred from a high-energy substrate directly to ADP.
- Occurs twice in the payoff phase of glycolysis, catalyzed by Phosphoglycerate Kinase and Pyruvate Kinase.
- The high-energy donor molecules are 1,3-Bisphosphoglycerate and Phosphoenolpyruvate (PEP).
- Produces a net total of 2 ATP per glucose molecule through this mechanism.
- It is an anaerobic process, not requiring oxygen or the electron transport chain.
- Crucial for ATP production in RBCs.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

