Pay-off Phase Reactions - The Energy Windfall
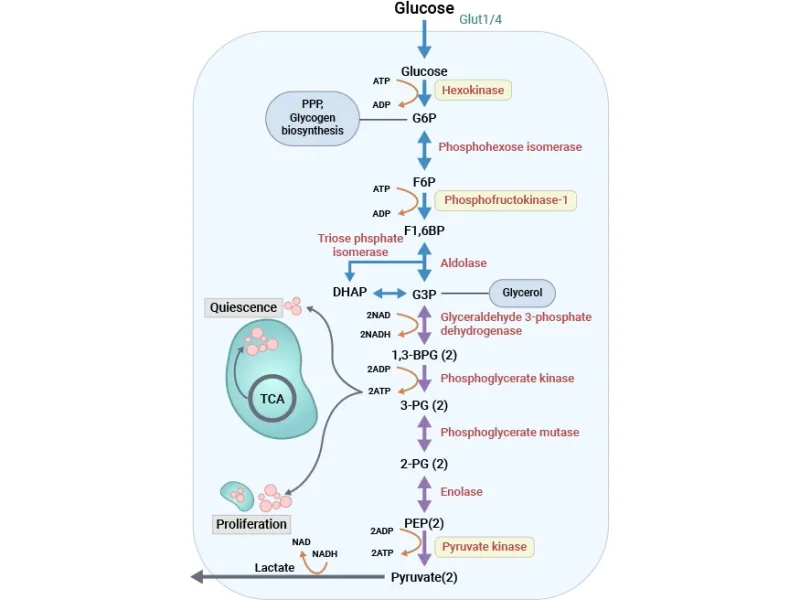
This phase converts the initial 3-carbon sugars into pyruvate, capturing energy in the form of ATP and NADH. For each molecule of glucose, this entire sequence occurs twice.
-
Net Yield per Glucose: 2 ATP & 2 NADH.
-
Step 1: Oxidation of G3P
- Enzyme: Glyceraldehyde-3-P dehydrogenase.
- Reaction: $Glyceraldehyde-3-P + NAD⁺ + Pi ⇌ 1,3-BPG + NADH + H⁺$.
- Produces 1,3-Bisphosphoglycerate (1,3-BPG), a high-energy compound, and 2 NADH (per glucose).
-
Step 2: First ATP Generation (Substrate-Level Phosphorylation)
- Enzyme: Phosphoglycerate kinase.
- Reaction: $1,3-BPG + ADP ⇌ 3-Phosphoglycerate + ATP$.
- Produces 2 ATP (per glucose), balancing the investment phase.
-
Step 3 & 4: Isomerization & Dehydration
- Phosphoglycerate mutase shifts the phosphate from C3 to C2.
- Enolase dehydrates 2-Phosphoglycerate to form Phosphoenolpyruvate (PEP), another high-energy compound. ⚠️ Inhibited by Fluoride.
-
Step 5: Second ATP Generation (Substrate-Level Phosphorylation)
- Enzyme: Pyruvate kinase.
- Reaction: $PEP + ADP → Pyruvate + ATP$.
- Produces 2 ATP (per glucose).
⭐ Arsenate Poisoning: Arsenate mimics inorganic phosphate (Pi) and uncouples the Glyceraldehyde-3-P dehydrogenase reaction. This prevents the synthesis of 1,3-BPG and bypasses the subsequent ATP-generating step by phosphoglycerate kinase, leading to a net yield of 0 ATP from glycolysis.
📌 Mnemonic for Enzymes: High Profile People Act Too Glamorous, Picture Posing Every Place (P=Phosphoglycerate Kinase, P=Phosphoglycerate Mutase, E=Enolase, P=Pyruvate Kinase).
Regulation & Clinical Pearls - Control and Chaos
-
Pyruvate Kinase (PK) Regulation: The final, irreversible step; a major control point.
- Feed-Forward Activation: ↑ by Fructose-1,6-bisphosphate (product of PFK-1).
- Allosteric Inhibition: ↓ by ATP & Alanine (signals of high energy & building blocks).
- Hormonal Control (Liver only): Glucagon → PKA activation → Phosphorylation → PK inhibition. Prevents liver from consuming glucose needed by the brain during fasting.
-
Pyruvate Kinase Deficiency:
- Autosomal recessive disorder causing chronic hemolytic anemia.
- RBCs are exclusively affected as they lack mitochondria and depend entirely on glycolysis for ATP.
- ↓ ATP → dysfunctional membrane ion pumps → RBC dehydration → Echinocyte (burr cell) formation.
- Leads to extravascular hemolysis by splenic macrophages → Splenomegaly.
- 📌 PK deficiency makes RBCs Pale & Kaput.
⭐ In Pyruvate Kinase deficiency, the lack of ATP disrupts the Na⁺/K⁺ pump in RBCs, leading to dehydration, membrane damage (echinocytes), and phagocytosis by splenic macrophages. This is the second most common enzyme-related cause of hemolytic anemia after G6PD deficiency.
High‑Yield Points - ⚡ Biggest Takeaways
- The payoff phase produces a net of 2 ATP and 2 NADH per glucose molecule.
- Substrate-level phosphorylation occurs at two steps, catalyzed by Phosphoglycerate Kinase and Pyruvate Kinase.
- Pyruvate Kinase is a key regulated, irreversible enzyme, subject to feed-forward activation by fructose-1,6-bisphosphate.
- Arsenate poisoning uncouples G3P dehydrogenase, leading to zero net ATP production from glycolysis.
- Fluoride inhibits the Enolase enzyme, blocking the formation of phosphoenolpyruvate.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

