Fates of Pyruvate - The Metabolic Crossroads
Pyruvate is a key metabolic intermediate. Its fate is determined by oxygen availability and the cell's energy requirements.
- Aerobic Conditions (Mitochondria): Pyruvate → Acetyl-CoA via Pyruvate Dehydrogenase Complex (PDC). This is the link to the TCA cycle.
- Anaerobic Conditions (Cytosol): Pyruvate → Lactate via Lactate Dehydrogenase (LDH). Regenerates $NAD^+$ for glycolysis.
- Gluconeogenesis (Mitochondria): Pyruvate → Oxaloacetate via Pyruvate Carboxylase.
- Transamination (Cytosol): Pyruvate ⇌ Alanine via Alanine Aminotransferase (ALT) (Cahill Cycle).
⭐ Warburg Effect: Cancer cells often favor converting pyruvate to lactate even with ample oxygen, a phenomenon supporting rapid growth.
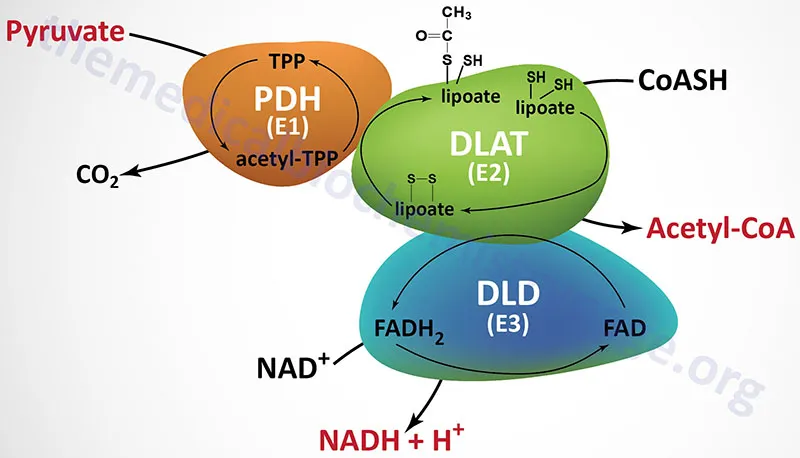
Aerobic Respiration - To Acetyl-CoA
- Site: Mitochondrial matrix.
- Enzyme: Pyruvate Dehydrogenase Complex (PDC).
- Reaction: An irreversible oxidative decarboxylation linking glycolysis to the TCA cycle.
- $Pyruvate + NAD⁺ + CoA \rightarrow Acetyl-CoA + NADH + H⁺ + CO₂$
- PDC Cofactors: Requires 5 coenzymes derived from vitamins.
- Thiamine pyrophosphate (TPP, B1)
- Lipoic acid
- Coenzyme A (CoA, B5)
- FAD (B2)
- NAD⁺ (B3)
- 📌 Mnemonic: Tender Loving Care For Nancy.
- Regulation:
- Activators: ↑ ADP, ↑ Ca²⁺, ↑ NAD⁺/NADH ratio.
- Inhibitors: ↑ ATP, ↑ Acetyl-CoA, ↑ NADH (product inhibition).
⭐ Clinical Pearl: Arsenic poisoning inhibits lipoic acid, inactivating the PDC. This leads to a backup of pyruvate and lactate, causing vomiting, rice-water stools, and a characteristic garlic breath.
Anaerobic Fermentation - Lactate & Alcohol
-
Lactate Fermentation (Human cells)
- Goal: Regenerate NAD⁺ to sustain glycolysis in anaerobic conditions.
- Sites: Erythrocytes, lens, cornea, kidney medulla, and exercising skeletal muscle.
- Enzyme: Lactate Dehydrogenase (LDH).
- Reaction: Reversible conversion.
- $Pyruvate + NADH + H^+ \rightleftharpoons Lactate + NAD^+$
- Lactate enters the Cori cycle, traveling to the liver for conversion back to glucose.
-
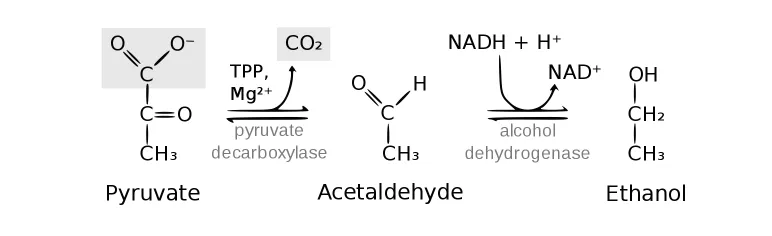
Alcohol Fermentation (Yeast & microbes)
- Goal: Also regenerates NAD⁺ for glycolysis.
- Process: A two-step pathway.
- Pyruvate decarboxylase converts pyruvate to acetaldehyde, releasing CO₂.
- Alcohol dehydrogenase reduces acetaldehyde to ethanol.
- $Pyruvate \rightarrow Acetaldehyde \rightarrow Ethanol$
⭐ Pyruvate decarboxylase requires Thiamine (B1). Its deficiency (e.g., in chronic alcoholism) impairs glucose breakdown, contributing to Wernicke-Korsakoff syndrome.
Anaplerosis & Gluconeogenesis - To Oxaloacetate
- Reaction: Pyruvate is irreversibly carboxylated to oxaloacetate (OAA).
- Enzyme: Pyruvate carboxylase.
- Location: Mitochondria.
- Equation: $Pyruvate + HCO_3^- + ATP \rightarrow OAA + ADP + P_i$
- Cofactor: Requires Biotin (Vitamin B7).
- 📌 Mnemonic: "ABC" for the enzyme's needs: ATP, Biotin, CO₂.
- Regulation:
- Allosterically activated by ↑ Acetyl-CoA. High Acetyl-CoA signals abundant energy, shunting pyruvate to gluconeogenesis or TCA replenishment instead of oxidation.
- Dual Roles:
- Anaplerosis: Replenishes OAA for the TCA cycle.
- Gluconeogenesis: First step for synthesizing glucose from pyruvate.
⭐ High-Yield Fact: Pyruvate carboxylase deficiency is an autosomal recessive disorder causing lactic acidosis and developmental delay due to impaired gluconeogenesis and TCA cycle function.
High‑Yield Points - ⚡ Biggest Takeaways
- Anaerobic glycolysis in muscle converts pyruvate to lactate via lactate dehydrogenase (LDH), regenerating NAD+.
- In yeast, pyruvate is converted to ethanol and CO₂ (alcoholic fermentation).
- Aerobic respiration converts pyruvate to acetyl-CoA via the pyruvate dehydrogenase (PDH) complex, linking glycolysis to the TCA cycle.
- Pyruvate can be carboxylated to oxaloacetate by pyruvate carboxylase for gluconeogenesis or to replenish TCA cycle intermediates.
- Transamination of pyruvate by alanine aminotransferase (ALT) yields alanine.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

