DDR Signaling - Damage Control Central
- Core Function: Senses DNA damage, halts the cell cycle to allow time for repair, or triggers apoptosis if damage is irreparable.
- Key Sensors (Kinases):
- ATM (Ataxia-Telangiectasia Mutated): Activated primarily by Double-Strand Breaks (DSBs).
- ATR (AT and Rad3-related): Responds to Single-Strand Breaks (SSBs) & stalled replication forks.
- Key Effector: p53 ("Guardian of the Genome") is stabilized, leading to transcription of cell cycle inhibitors (e.g., p21).
⭐ Li-Fraumeni syndrome, an autosomal dominant disorder with high cancer predisposition, is caused by germline mutations in the TP53 gene.
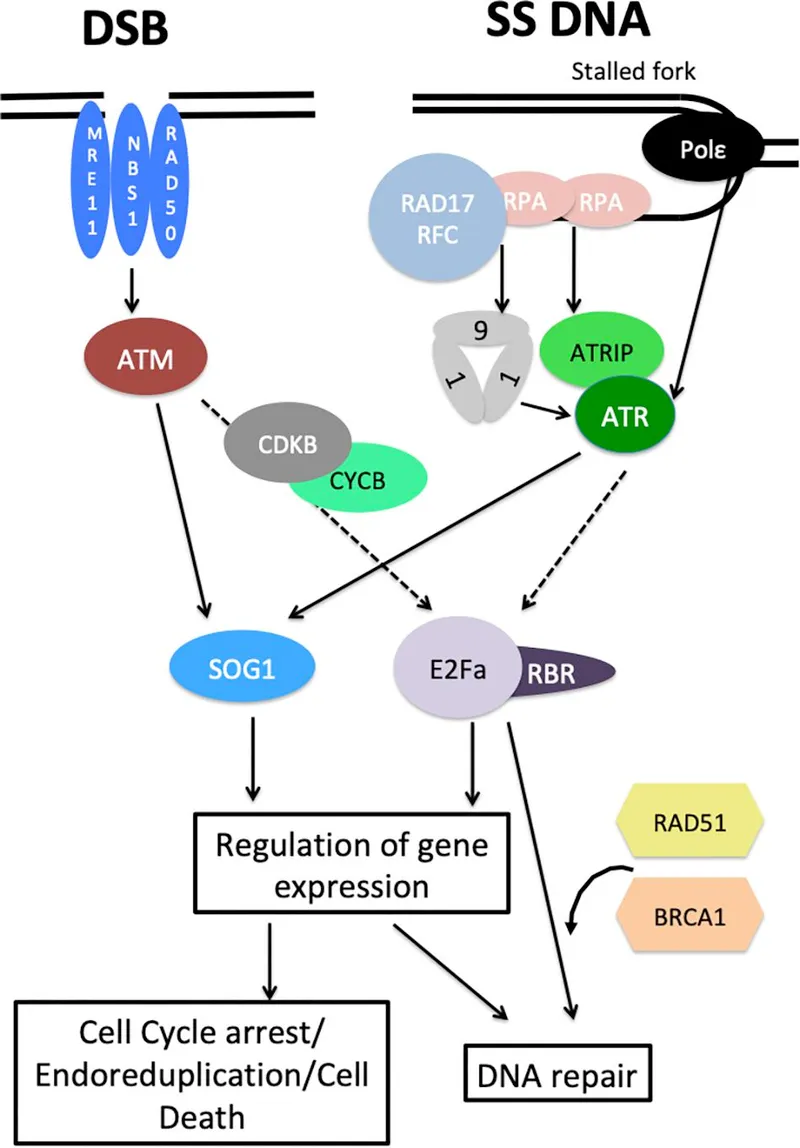
Sensors & Transducers - First Responders
- Sensors: Proteins that directly recognize DNA lesions or altered chromatin structures.
- Double-Strand Breaks (DSBs): Detected by the MRN complex (MRE11-RAD50-NBS1).
- Single-Strand Breaks (SSBs) & Stalled Forks: Recognized by RPA (Replication Protein A) and the 9-1-1 checkpoint clamp.
- Transducers: Key kinases that amplify the damage signal.
- ATM (Ataxia-Telangiectasia Mutated): Recruited and activated by the MRN complex at DSBs.
- ATR (ATM and Rad3-related): Recruited to RPA-coated ssDNA.
- 📌 Mnemonic: ATM for MRN/DSBs; ATR for RPA/SSBs.
⭐ Ataxia-Telangiectasia results from ATM gene mutations. It presents with cerebellar ataxia, telangiectasias, immunodeficiency, and extreme sensitivity to ionizing radiation due to failed DSB repair signaling.
Effector Proteins - The Cleanup Crew
Once activated, the DDR signaling cascade triggers downstream effector proteins to manage the damage. The cell's fate-repair, arrest, or death-hangs in the balance, largely orchestrated by the master regulator, p53.
- Cell Cycle Arrest: Halts progression to prevent replication of damaged DNA.
- p53 induces p21, which inhibits Cyclin-Dependent Kinases (CDKs).
- Prevents G1/S and G2/M transitions.
- Apoptosis: Initiated when damage is too extensive for repair.
- p53 upregulates pro-apoptotic proteins like BAX.
- DNA Repair: Activates and recruits the specific molecular machinery.
- e.g., BRCA1, BRCA2 for homologous recombination.
⭐ Li-Fraumeni Syndrome: A rare, autosomal dominant disorder caused by a germline mutation in the TP53 gene. It leads to a significantly increased risk of developing multiple types of cancer at a young age.
Clinical Correlates - When Signals Cross
-
Ataxia-Telangiectasia (AT)
- Gene Defect: Autosomal recessive mutation in the ATM gene.
- Pathophysiology: Defective dsDNA break sensing, leading to failed cell cycle arrest.
- Clinical Triad: Cerebellar ataxia, oculocutaneous telangiectasias, and severe immunodeficiency.
- Labs: ↑ alpha-fetoprotein (AFP), IgA deficiency.
- ⚠️ Warning: Extreme sensitivity to ionizing radiation.
-
Li-Fraumeni Syndrome
- Gene Defect: Autosomal dominant mutation in the TP53 gene.
- Pathophysiology: Loss of the "guardian of the genome" impairs apoptosis and cell cycle control.
⭐ Exam Favorite: TP53 mutations are found in over 50% of all human cancers, making it a critical tumor suppressor gene.
High‑Yield Points - ⚡ Biggest Takeaways
- ATM and ATR are the primary sensors; ATM for double-strand breaks and ATR for single-strand breaks.
- They activate the p53 tumor suppressor, the "guardian of the genome," via phosphorylation.
- p53 induces p21, which inhibits CDKs, leading to cell cycle arrest at G1/S or G2/M checkpoints, allowing time for repair.
- If damage is irreparable, p53 triggers apoptosis by upregulating proteins like BAX.
- Germline TP53 mutations cause Li-Fraumeni syndrome, predisposing to various cancers.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

