RCT Fundamentals - The Gold Standard
- Definition: An experimental study where participants are randomly assigned to receive either an intervention (treatment) or a placebo/standard care (control).
- Purpose: Considered the gold standard for establishing causality between an intervention and an outcome.
- Key Feature: Randomization. This process minimizes selection bias and balances known and unknown confounding variables between groups.
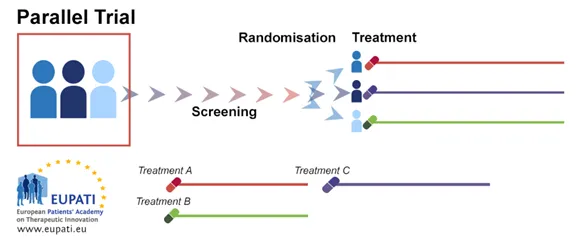
⭐ The primary strength of an RCT is its ability to minimize confounding by randomly distributing potential confounders between the study groups, thereby isolating the intervention's true effect.
Controls & Blinding - Seeing Clearly
- Control Group: The benchmark for comparison in an experiment. Essential for isolating the effects of the intervention.
- Placebo Control: An inert substance/procedure given to the control group. Helps differentiate the true therapeutic effect from the psychological placebo effect.
- Standard of Care: Active control using the currently accepted treatment. Used when withholding treatment is unethical.
- Blinding (Masking): Concealing treatment allocation from individuals involved in the study to prevent bias.
- Single-Blind: Patients are unaware of their group allocation.
- Double-Blind: Both patients and investigators are unaware of the allocation. The gold standard for minimizing observer and subject bias.
⭐ Double-blinding is the most effective method to reduce both patient expectancy effects and observer bias from the research team.
Analysis & Interpretation - The Numbers Game
- Primary Analysis: Intention-to-Treat (ITT)
- Analyzes all randomized patients in their original assigned groups, regardless of adherence or protocol deviations.
- Preserves randomization benefits and reflects real-world effectiveness.
- Key Efficacy Metrics:
- Absolute Risk Reduction (ARR): $\text{Control Event Rate} - \text{Experimental Event Rate}$
- Relative Risk Reduction (RRR): $1 - \text{Relative Risk}$
- Number Needed to Treat (NNT): $\frac{1}{\text{ARR}}$
⭐ A lower NNT indicates a more effective intervention. An NNT of 5 means you treat 5 people to prevent one additional bad outcome.
- Statistical Significance (p < 0.05):
- The 95% Confidence Interval (CI) for Relative Risk (RR) or Odds Ratio (OR) must not include 1.0.
- The 95% CI for a mean difference must not include 0.
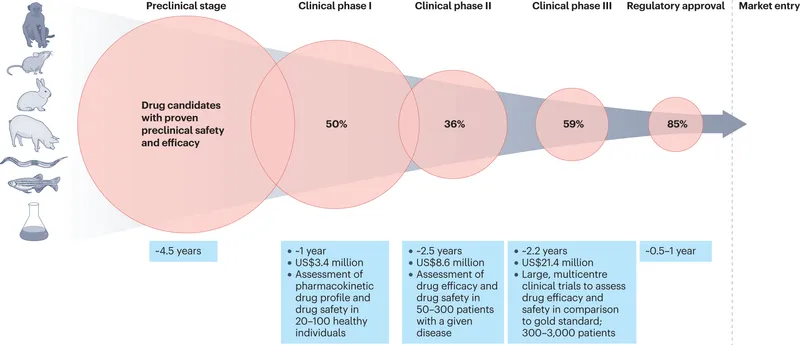
Clinical Trial Phases - The Long Road
- Phase I: Safety & Dosage.
- Assesses safety, toxicity, pharmacokinetics, and pharmacodynamics.
- Small number of healthy volunteers (~20-80).
- Phase II: Efficacy & Dosing.
- Assesses treatment efficacy, optimal dosing, and adverse effects.
- Moderate number of patients with the disease (~100-300).
- Phase III: Comparison & Effectiveness.
- Compares the new treatment to the current standard of care.
- Large, randomized controlled trials (~1,000-3,000+).
- Basis for FDA approval.
- Phase IV: Post-Marketing Surveillance.
- Monitors long-term effectiveness and adverse effects after approval.
- Detects rare or long-term side effects.
📌 Mnemonic: Safety, Works?, Improvement?, Market? (SWIM)
⭐ Only about 1 in 10 drugs that enter Phase I trials are ultimately approved by the FDA.
High‑Yield Points - ⚡ Biggest Takeaways
- The gold standard for determining causality.
- Relies on random assignment to minimize selection bias and balance known and unknown confounders.
- Blinding (single, double, triple) is a key feature to reduce observer and subject bias.
- Intention-to-treat analysis preserves randomization by analyzing participants in their originally assigned groups.
- Loss to follow-up is a major threat, potentially introducing attrition bias, a type of selection bias.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

