Conflicts of Interest - Money & Medicine
- Definition: Arises when a primary interest (e.g., patient welfare, research integrity) is potentially influenced by a secondary interest (e.g., financial gain).
- Common Sources:
- Industry-sponsored meals, gifts, travel
- Speaking & consulting fees (honoraria)
- Research grants
- Stock or equity interests
- Management: The primary ethical duty is transparency. Full disclosure to employers, IRBs, and patients is essential. May require recusal.
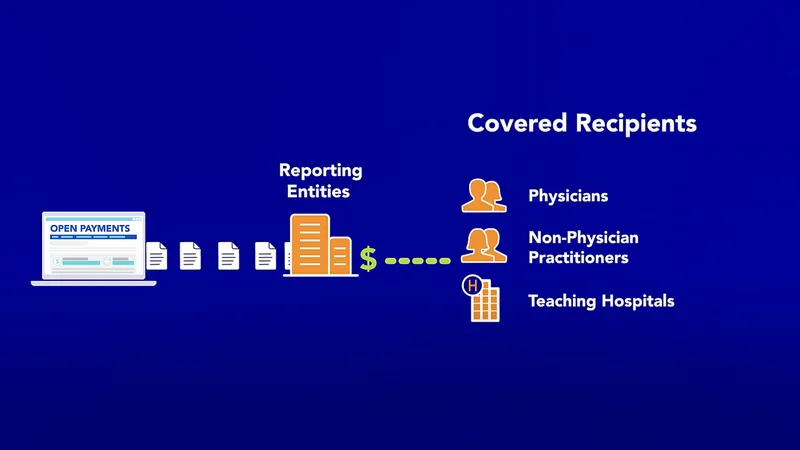
⭐ The Physician Payments Sunshine Act (Open Payments Program) requires manufacturers to report payments and transfers of value to physicians and teaching hospitals.
COI Flavors - More Than Just Cash
Conflicts of interest (COI) extend beyond direct financial gain, creating subtle biases that can compromise professional judgment in patient care, research, and education. Awareness of these non-financial influences is critical for maintaining trust and objectivity.
- Personal & Family Relationships: Bias from loyalty to friends, family, or close colleagues, potentially affecting referrals, hiring, or peer review.
- Professional Advancement: Desire for promotion, tenure, or prestigious appointments may influence research conduct or reporting.
- Intellectual COI: A researcher's strong commitment to their own theory can impair objectivity when evaluating competing ideas.
- Gifts & Perks (In-kind COI): Includes sponsored travel, meals, or entertainment. Even small gifts can create a sense of reciprocity.
⭐ The Physician Payments Sunshine Act requires public reporting of financial ties and transfers of value over $10 between manufacturers (drug/device) and physicians/teaching hospitals.

Taming the Beast - Handling COIs
- Core Principle: Transparency. The primary step in managing any potential conflict of interest is full disclosure to all relevant parties (institution, IRB, patients).
- Goal: To safeguard patient welfare and maintain the integrity of research and clinical judgment.
⭐ The Physician Payments Sunshine Act requires public reporting of financial relationships between drug/device manufacturers and physicians/teaching hospitals. This data is publicly available on the CMS "Open Payments" website.
High‑Yield Points - ⚡ Biggest Takeaways
- A conflict of interest arises when a primary interest (e.g., patient welfare) is potentially influenced by a secondary interest (e.g., financial gain).
- The primary goal is management and mitigation, not necessarily total elimination of the secondary interest.
- Full disclosure to patients, institutions, and journals is the most critical step in management.
- Even the perception of a conflict can erode patient trust as much as an actual conflict.
- Institutional Review Boards (IRBs) and specific committees oversee and manage these conflicts.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

