Monoamine Hypothesis - The Chemical Imbalance
- Core Idea: Depression results from a functional deficiency of key monoamine neurotransmitters in the CNS.
- Primary Amines Implicated:
- Serotonin (5-HT): Regulates mood, sleep, and appetite.
- Norepinephrine (NE): Affects alertness, energy, and attention.
- Dopamine (DA): Modulates pleasure, reward, and motivation.
- Supporting Evidence: The mechanism of action of most antidepressants involves increasing monoamine levels (e.g., SSRIs, SNRIs, MAOIs).
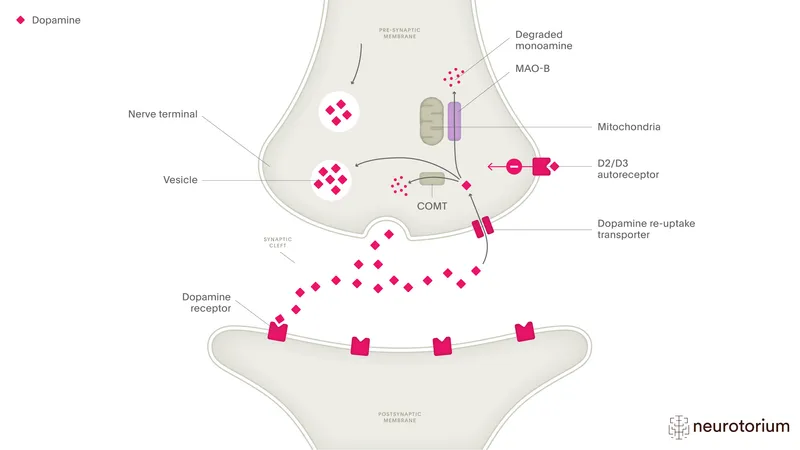
⭐ The 2-4 week therapeutic lag for antidepressants suggests that the acute increase in neurotransmitters is not the sole mechanism. The clinical effect likely involves downstream adaptations, such as changes in receptor sensitivity and gene expression.
Neurotrophic Hypothesis - Stunted Brain Growth
- Core Idea: Depression is linked to ↓ levels of Brain-Derived Neurotrophic Factor (BDNF), a key protein for neuron growth, survival, and neurogenesis.
- Pathophysiology:
- Chronic stress → ↑ cortisol → ↓ BDNF synthesis.
- Low BDNF leads to atrophy and reduced connectivity in the hippocampus and prefrontal cortex.
- This impairs mood regulation, memory, and executive function.
- Antidepressant Action:
- Most antidepressants ↑ BDNF levels over time.
- This stimulates neurogenesis and repairs neuronal connections.
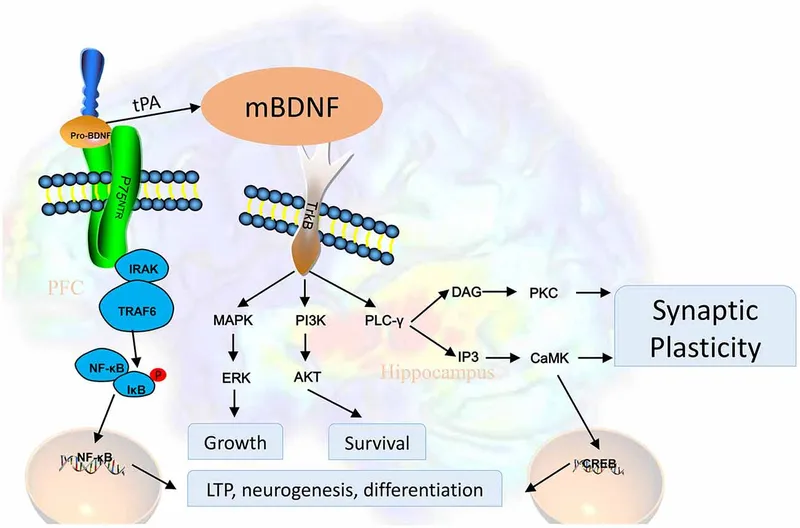
⭐ The delayed therapeutic effect of many antidepressants (weeks) is thought to reflect the time required for BDNF levels to rise and exert their neurogenic and synaptogenic effects.
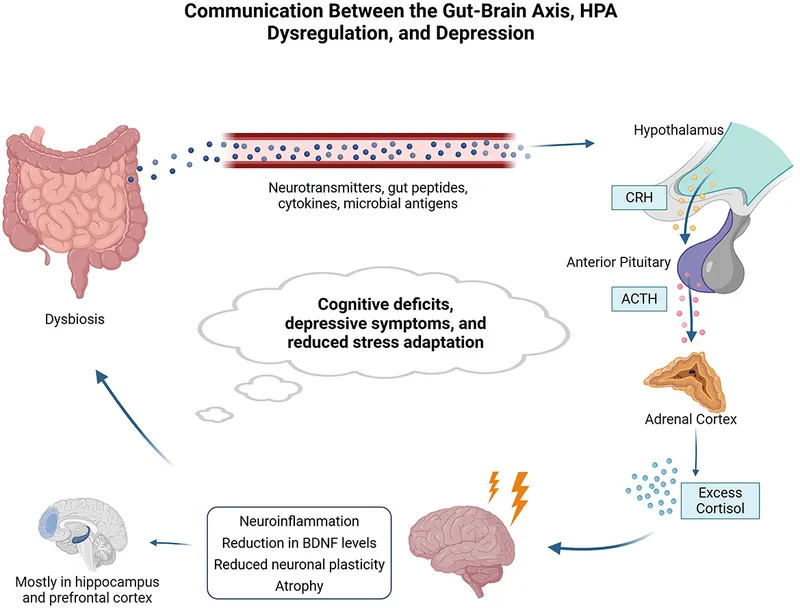
HPA Axis Dysregulation - The Stress Connection
- Chronic stress leads to a hyperactive Hypothalamic-Pituitary-Adrenal (HPA) axis, causing elevated cortisol levels (hypercortisolemia).
- Core Defect: Impaired negative feedback. Glucocorticoid receptors (GR) in the hippocampus & hypothalamus become desensitized to cortisol.
- This dysregulation leads to persistently high levels of CRH and ACTH, creating a vicious cycle.
- Sustained ↑ cortisol is neurotoxic, contributing to hippocampal atrophy and decreased neurogenesis seen in depression.
⭐ The Dexamethasone Suppression Test (DST) can reveal this faulty feedback loop. In many depressed patients, cortisol levels are not suppressed after a dose of dexamethasone, indicating HPA axis hyperactivity.
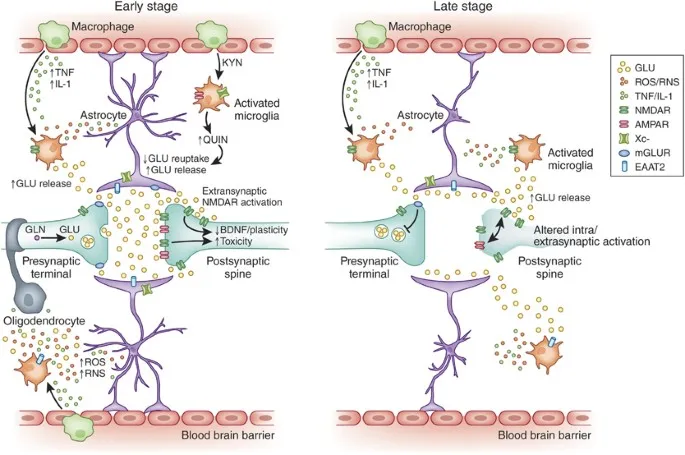
Neuroinflammation - A Brain on Fire
- Chronic stress and systemic inflammation ↑ pro-inflammatory cytokines (IL-6, TNF-α, IL-1β).
- These cytokines cross the blood-brain barrier, activating microglia (the brain's immune cells).
- Activated microglia drive neuroinflammation, which promotes depression by:
- Shifting tryptophan metabolism away from serotonin synthesis (↑ kynurenine pathway).
- ↓ Brain-Derived Neurotrophic Factor (BDNF), impairing neurogenesis.
- ↑ Oxidative stress, leading to neuronal damage.
⭐ Elevated C-Reactive Protein (CRP), a peripheral inflammatory marker, is associated with a poorer response to traditional SSRIs.
High-Yield Points - ⚡ Biggest Takeaways
- The monoamine hypothesis is central, postulating deficiencies in serotonin (5-HT), norepinephrine (NE), and dopamine (DA).
- Chronic depression leads to structural brain changes, including decreased hippocampal volume and reduced frontal lobe metabolic activity.
- HPA axis hyperactivity is common, resulting in elevated cortisol levels and impaired negative feedback.
- Inflammation plays a key role, with increased levels of pro-inflammatory cytokines.
- Brain-Derived Neurotrophic Factor (BDNF) levels are often decreased, impairing neuroplasticity and neurogenesis.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

