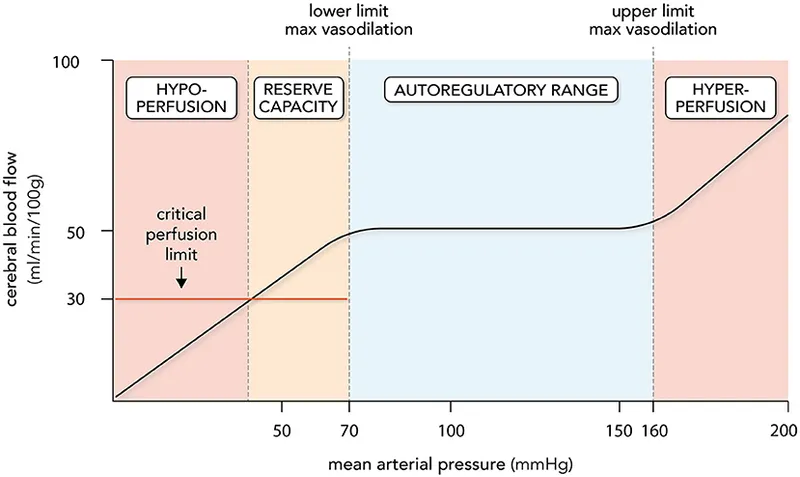
Autoregulation - Keeping Flow Steady
Intrinsic ability of an organ to maintain constant blood flow despite changes in perfusion pressure. The primary goal is to match blood flow to metabolic demand.
- Governing Equation: $Flow = ΔP / R$
- Organs alter vascular resistance (R) to maintain flow when perfusion pressure (ΔP) changes.
⭐ Coronary, cerebral, and renal circulations exhibit excellent autoregulation.
Myogenic Mechanism - The Muscle Flex
- Core Principle: An intrinsic property of vascular smooth muscle to contract in response to stretch. This is also known as the Bayliss effect.
- Mechanism:
- ↑ Arterial pressure → stretches the arteriolar wall.
- Stretch opens mechanosensitive Ca²⁺ channels in the smooth muscle membrane.
- ↑ Ca²⁺ influx → depolarization → smooth muscle contraction (vasoconstriction).
- Function: Maintains constant blood flow despite changes in systemic pressure. It's independent of neural or hormonal control.
- Crucial in: Organs with critical flow needs like the kidney (afferent arteriole) and brain.
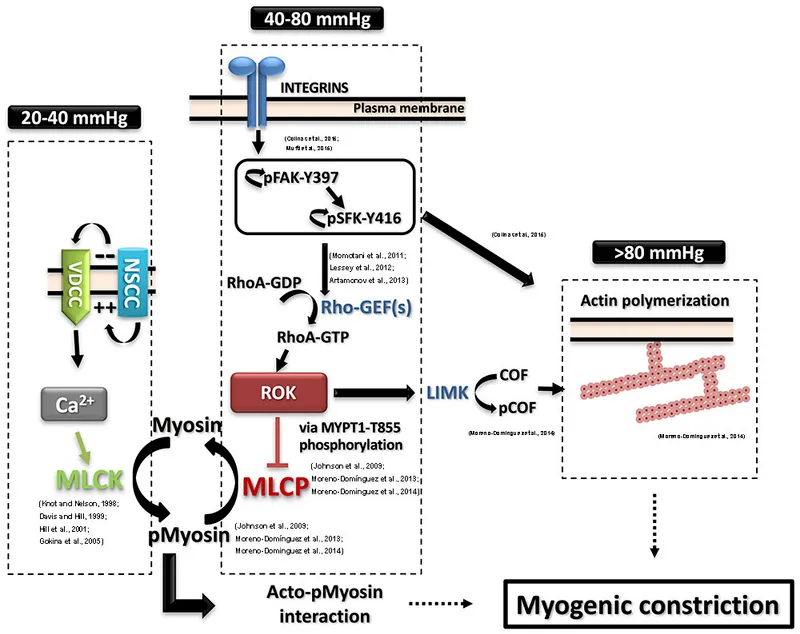
⭐ The myogenic response is vital for protecting fragile capillaries from damage caused by sudden surges in arterial pressure.
Metabolic Control - Chemical Signals
Increased tissue metabolism or decreased O₂ supply triggers the release of vasoactive metabolites, directly coupling blood flow to metabolic demand.
- Key Vasodilators
- ↑ $CO_2$, ↑ $H^+$ (↓ pH)
- ↑ Adenosine (especially in heart)
- ↑ $K^+$, ↑ Lactate
- Key Vasoconstrictors
- Endothelin: Potent, locally released peptide.
- Oxygen: Causes vasoconstriction in systemic arterioles but vasodilation in pulmonary circulation.
- Active Hyperemia: ↑ blood flow due to ↑ metabolic activity.
- Reactive Hyperemia: ↑ blood flow following a period of ischemia.
⭐ Adenosine is the primary mediator of coronary blood flow regulation, linking myocardial oxygen consumption directly to coronary perfusion.
Organ Specifics - Location, Location, Location
Autoregulation varies significantly between organs, tailored to their unique metabolic demands and functions. The brain, heart, and kidneys are classic examples of tissues with robust intrinsic control over their own blood supply.
| Organ | Primary Mechanism(s) | Key Mediators | Clinical Pearl |
|---|---|---|---|
| Brain | Metabolic (dominant) | Arterial $pCO_2$ is the most potent cerebral vasodilator. | Chronic hypertension shifts the autoregulatory curve to the right. |
| Heart | Local Metabolic | Adenosine, $NO$, $CO_2$, ↓$O_2$. Most sensitive to metabolic changes. | Coronary steal: vasodilators can shunt blood away from ischemic zones. |
| Kidney | Myogenic & Tubuloglomerular Feedback (TGF) | Stretch (myogenic); ATP, Adenosine (TGF at macula densa). | ACE inhibitors preferentially dilate the efferent arteriole, reducing intraglomerular pressure. |
High‑Yield Points - ⚡ Biggest Takeaways
- Autoregulation maintains constant blood flow to vital organs despite fluctuating arterial pressure.
- It primarily involves myogenic and metabolic mechanisms.
- The myogenic response causes vasoconstriction when arteriolar smooth muscle is stretched by ↑ pressure.
- Metabolic control matches blood flow to tissue activity; key vasodilators include adenosine, NO, CO₂, and K⁺.
- Most prominent in the heart, brain, and kidneys.
- Failure can lead to organ damage from hypoperfusion or hyperperfusion.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

