Hemoglobin Structure - The Oxygen Taxi
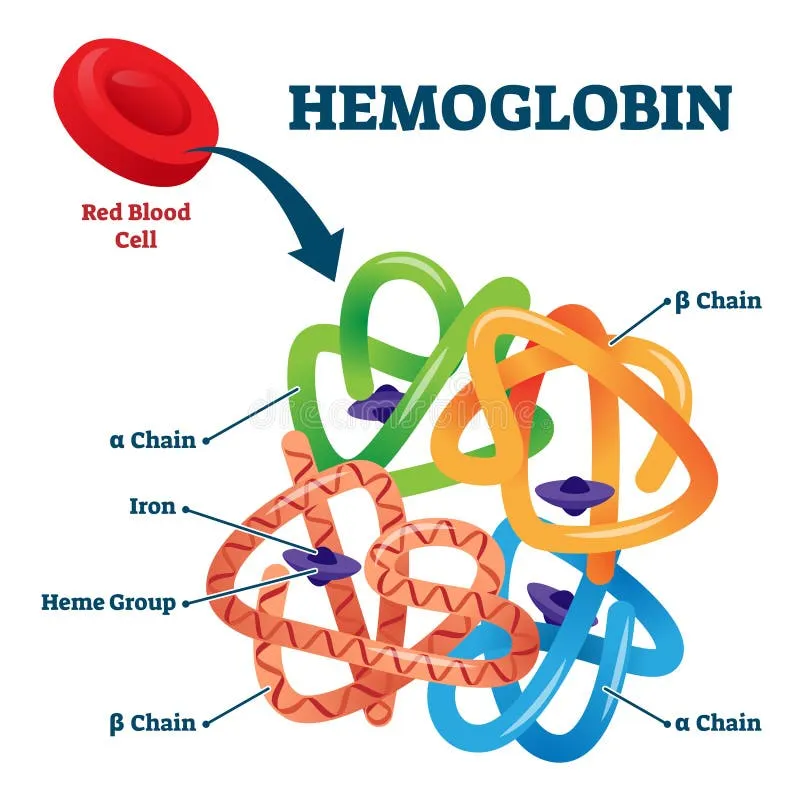
- Composition: Adult hemoglobin (HbA) is a tetramer ($α_2β_2$). Each of the four globin chains contains a heme group (iron-porphyrin) that binds one $O_2$ molecule. Total capacity: four $O_2$ per Hb.
- Key Forms:
- Taut (T) form: Deoxygenated state with low $O_2$ affinity. Stabilized by ↑$H^+$, ↑$CO_2$, and ↑2,3-BPG.
- Relaxed (R) form: Oxygenated state with high $O_2$ affinity. Binding $O_2$ triggers this conformational shift.
⭐ Fetal hemoglobin (HbF, $α_2γ_2$) binds 2,3-BPG poorly, giving it a higher oxygen affinity than HbA to facilitate placental $O_2$ transfer.
Oxy-Hgb Dissociation Curve - The Great Let-Go
- Sigmoid curve showing hemoglobin's (Hb) dynamic affinity for oxygen ($O_2$).
- Cooperative Binding: Binding of one $O_2$ increases Hb's affinity for the next.
- P50: The $PO_2$ where Hb is 50% saturated. Normal ≈ 27 mmHg. ↑P50 = ↓affinity.
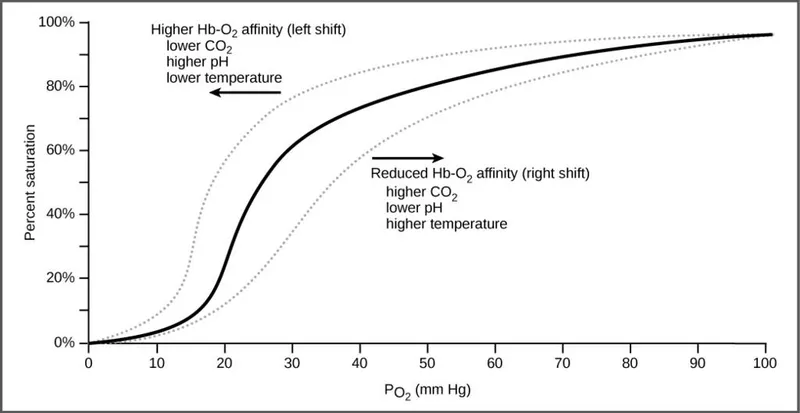
- Right Shift: Hb "lets go" of $O_2$ to tissues.
- 📌 CADET, face Right!: ↑ CO₂, Acid, 2,3-DPG, Exercise, Temperature.
- Left Shift: Hb holds on to $O_2$.
- Caused by opposite factors, plus Fetal Hb (HbF) and CO.
⭐ Fetal hemoglobin (HbF) has a low affinity for 2,3-DPG, creating a left-shifted curve. This higher $O_2$ affinity is crucial for pulling oxygen from the maternal circulation.
Curve Shifters - Right & Left Hooks
Factors that alter hemoglobin's affinity for $O_2$, shifting the dissociation curve.
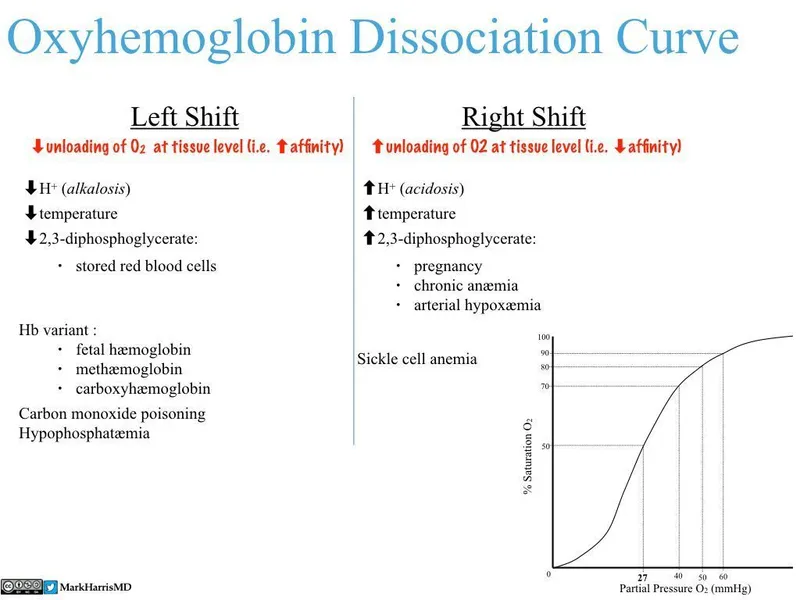
| Right Shift (↓ $O_2$ Affinity) | Left Shift (↑ $O_2$ Affinity) |
|---|---|
| * C$O_2$ (↑) | * $CO_2$ (↓) |
| * Acid (↓ pH) | * Alkali (↑ pH) |
| * DPG (2,3-DPG) (↑) | * DPG (2,3-DPG) (↓) |
| * Exercise | * Temperature (↓) |
| * Temperature (↑) | * Fetal Hemoglobin (HbF) |
| 📌 CADET, face Right! | * Carbon Monoxide (CO) |
| (for factors that shift right) | * Methemoglobin |
Binding Blunders - When O2 Can't Ride
- Methemoglobinemia
- Iron oxidized ($Fe^{2+} o Fe^{3+}$), can't bind $O_2$. Causes functional anemia.
- Causes: Nitrites, dapsone, anesthetics (benzocaine).
- Findings: Chocolate-colored blood, cyanosis. $SpO_2$ often reads near 85%.
- Tx: Methylene blue, Vitamin C.
- Carbon Monoxide (CO) Poisoning
- CO has >200x affinity for Hb than $O_2$, ↓ $O_2$ saturation & content.
- Causes left-shift, impairing $O_2$ unloading.
- Tx: 100% $O_2$; hyperbaric $O_2$.
⭐ In CO poisoning, $PaO_2$ (dissolved $O_2$) is normal, but $SaO_2$ (Hb-bound $O_2$) is low.
High‑Yield Points - ⚡ Biggest Takeaways
- Hemoglobin exhibits positive cooperativity, resulting in a sigmoidal O₂-dissociation curve.
- A right shift (↓ O₂ affinity) is caused by ↑ CO₂, ↑ Acid/H⁺, ↑ 2,3-BPG, ↑ Exercise, and ↑ Temperature.
- Think "CADET, face Right" for factors that promote O₂ unloading to tissues.
- A left shift (↑ O₂ affinity) is caused by the opposite conditions, plus fetal hemoglobin (HbF) and CO poisoning.
- Bohr effect: In tissues, ↑ H⁺ and ↑ CO₂ promote O₂ release from hemoglobin.
- Haldane effect: In lungs, O₂ binding promotes the release of H⁺ and CO₂ from hemoglobin.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

