O2-Hb Dissociation Curve - Shifty Business
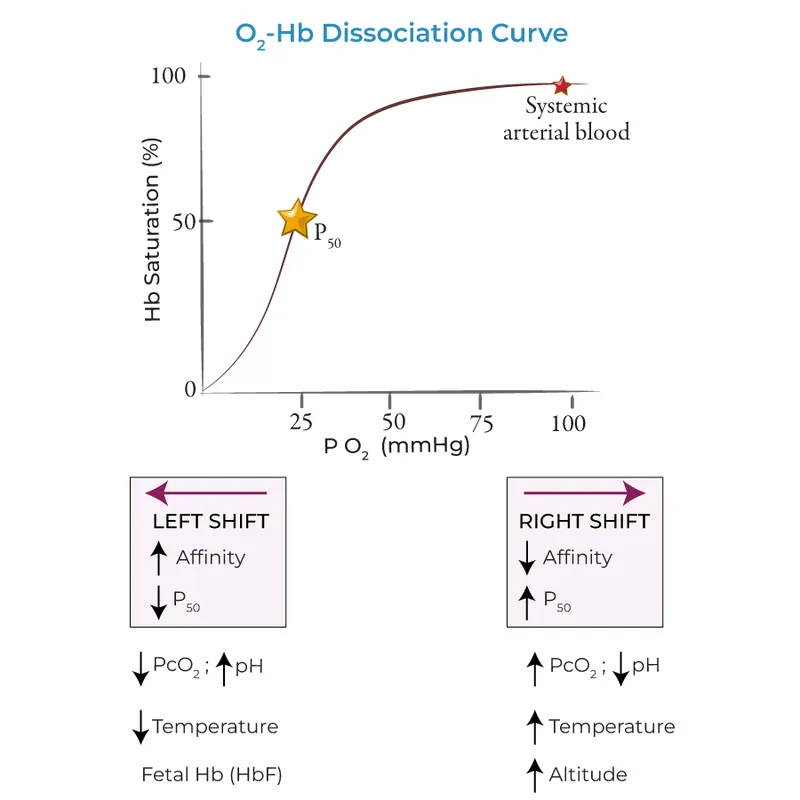
- Curve: Sigmoid shape, plots $SaO_2$ (Y-axis) vs. $PaO_2$ (X-axis), showing hemoglobin's affinity for oxygen.
- P50: The $PaO_2$ where hemoglobin is 50% saturated. Normal P50 ≈ 27 mmHg.
- Rightward Shift: Indicates ↓ O2 affinity, facilitating O2 unloading to tissues. A higher P50 means lower affinity.
- 📌 CADET, face Right! for factors causing a rightward shift:
- ↑ $CO_2$
- ↑ Acid (↓ pH)
- ↑ 2,3-DPG
- ↑ Exercise
- ↑ Temperature
⭐ The curve's sigmoid shape is a result of cooperative binding; as one O2 molecule binds to hemoglobin, the affinity for subsequent O2 molecules increases.
Bohr Effect - Letting Go of O2
Describes how ↑$PCO_2$ and ↑$H^+$ (↓pH) decrease hemoglobin's affinity for oxygen, enhancing $O_2$ release. This rightward shift of the oxygen-hemoglobin curve ensures oxygen delivery to metabolically active tissues.
- Mechanism: $H^+$ and $CO_2$ are allosteric inhibitors that stabilize the deoxygenated (T-state) of hemoglobin, promoting ↓Hb-$O_2$ affinity.
- Key Drivers in Tissues:
- ↑$H^+$ (acidosis)
- ↑$PCO_2$
- Chemical Basis: $H_2O + CO_2 \rightleftharpoons H_2CO_3 \rightleftharpoons H^+ + HCO_3^-$
- 📌 Mnemonic: Think CO₂ and Acid for the "C****ADET, face Right!" shift.
⭐ The Bohr effect is most prominent in the peripheral tissues, where high metabolic activity generates CO2 and acids, signaling a need for oxygen.
Rightward Shift Factors - CADET, face Right!
The oxygen-hemoglobin dissociation curve illustrates hemoglobin's affinity for O₂. A shift to the right indicates decreased affinity, facilitating O₂ release to tissues. A left shift signifies increased affinity, promoting O₂ uptake in the lungs. The P₅₀ is the partial pressure of O₂ at which hemoglobin is 50% saturated.
| Right Shift (↓ Affinity) | Left Shift (↑ Affinity) |
|---|---|
| ↑ P₅₀ | ↓ P₅₀ |
| ↑ CO₂ | ↓ CO₂ |
| ↓ pH (↑ Acid) | ↑ pH (↓ Acid) |
| ↑ 2,3-DPG | ↓ 2,3-DPG |
| ↑ Temperature | ↓ Temperature |
| ↑ Exercise | Fetal Hemoglobin (HbF) |
⭐ Fetal hemoglobin (HbF) has a low affinity for 2,3-DPG, resulting in a left-shifted curve compared to adult hemoglobin (HbA). This higher O₂ affinity facilitates O₂ transfer from the mother to the fetus across the placenta.
Haldane Effect - CO2's Ride Home
- In tissues, deoxygenated hemoglobin has a higher affinity for $CO_2$ and $H^+$, facilitating $CO_2$ uptake from tissues.
- Forms carbaminohemoglobin ($Hb-CO_2$).
- Buffers $H^+$, promoting conversion of $CO_2$ to bicarbonate ($HCO_3^-$) via the chloride shift.
- In the lungs, the binding of $O_2$ to hemoglobin decreases its affinity for $CO_2$, causing its release into the alveoli for expiration.
⭐ The Haldane effect is quantitatively more important for promoting $CO_2$ transport than the Bohr effect is for promoting $O_2$ transport.
📌 Haldane in the High-O2 Lungs helps release H+ from Hb.
High‑Yield Points - ⚡ Biggest Takeaways
- The Bohr effect is a rightward shift of the oxygen-hemoglobin curve, driven by ↑ PCO₂ or ↓ pH.
- This shift decreases hemoglobin's affinity for O₂, promoting oxygen unloading in metabolically active tissues.
- Key factors causing a rightward shift are ↑ CO₂, ↑ Acid, ↑ 2,3-DPG, ↑ Exercise, and ↑ Temperature.
- Protons (H⁺) and CO₂ act as allosteric inhibitors, stabilizing the taut (T) form of hemoglobin.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

