TDM Basics - The Balancing Act
- Immunosuppressants have a Narrow Therapeutic Index (NTI), where the therapeutic window between efficacy and toxicity is small, making precise dosing critical.
- Therapeutic Drug Monitoring (TDM) is used to maintain drug concentrations within this target range, individualizing therapy.
- The Balancing Act:
- Goal 1 (Efficacy): Prevent graft rejection by ensuring drug levels are not sub-therapeutic.
- Goal 2 (Safety): Avoid dose-related toxicities (e.g., nephrotoxicity, neurotoxicity) by ensuring levels are not supra-therapeutic.
⭐ For calcineurin inhibitors like Tacrolimus, trough concentrations ($C_0$) are the standard for TDM as they correlate well with total drug exposure ($AUC_{0-12}$).
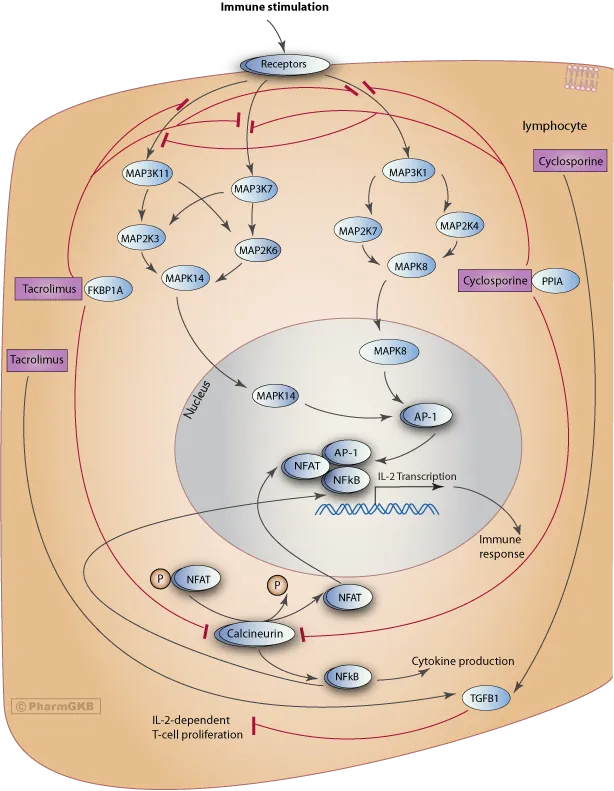
Calcineurin Inhibitors - Tacro & Cyclo
Calcineurin inhibitors (CNIs) are the backbone of most immunosuppressive regimens, preventing T-cell activation by blocking IL-2 transcription.
| Feature | Tacrolimus (FK-506) | Cyclosporine |
|---|---|---|
| Mechanism | Binds to FKBP-12 to inhibit calcineurin. | Binds to Cyclophilin to inhibit calcineurin. |
| Key Toxicities | More Neurotoxicity (tremor), Alopecia, New-onset Diabetes. | Hirsutism, Gingival Hyperplasia, Hyperlipidemia. |
| Shared Toxicity | Nephrotoxicity (vasoconstrictive), Hypertension, Hyperkalemia. | Nephrotoxicity, Hypertension, Hyperkalemia. |
| Monitoring | Trough (C0) levels. | Trough (C0) levels. |
| Target Range | Highly variable; typically 5-15 ng/mL. | Highly variable; typically 100-400 ng/mL. |
⭐ Exam Favorite: Both are metabolized by CYP3A4. Co-administration with inhibitors (e.g., azole antifungals, macrolides) can ↑ CNI levels and toxicity, while inducers (e.g., rifampin, phenytoin) can ↓ levels, risking rejection.
mTORi & Antimetabolites - Supporting Cast
A table comparing key non-calcineurin inhibitor agents used in immunosuppressive regimens.
| Class | Drug(s) | Key Toxicities |
|---|---|---|
| mTOR Inhibitors | Sirolimus (Rapamycin), Everolimus | Pancytopenia, poor wound healing, hyperlipidemia, insulin resistance, stomatitis. "Kidney-sparing" (less nephrotoxic than calcineurin inhibitors). |
| Antimetabolites | Mycophenolate Mofetil | GI distress (esp. diarrhea), bone marrow suppression. |
| Azathioprine | Pancreatitis, dose-related myelosuppression (leukopenia, thrombocytopenia). |
⭐ Allopurinol, a xanthine oxidase inhibitor, significantly increases Azathioprine toxicity by blocking its metabolism. Must reduce Azathioprine dose if co-administered.
Sampling Strategy - Peaks vs. Troughs
- Trough (C0): Lowest drug level, drawn immediately before the next dose. It assesses for overexposure and toxicity risk.
- Standard for Tacrolimus & Sirolimus.
- Peak (C2): Level drawn 2 hours post-dose.
- Sometimes used for Cyclosporine to better estimate total drug exposure (Area Under the Curve - AUC).
⭐ For Calcineurin inhibitors (Tacrolimus, Cyclosporine), trough levels are crucial. Consistently high troughs significantly increase the risk of long-term nephrotoxicity, a key cause of graft failure.
High‑Yield Points - ⚡ Biggest Takeaways
- Calcineurin inhibitors (Tacrolimus, Cyclosporine) and mTOR inhibitors have narrow therapeutic indices, demanding close monitoring.
- Trough levels (C0) are the standard for monitoring to minimize dose-related toxicities.
- Major toxicities include nephrotoxicity for CNIs and myelosuppression for mTOR inhibitors and antimetabolites.
- Mycophenolate (MMF) can cause significant leukopenia.
- Azathioprine requires TPMT testing to avoid severe myelosuppression.
- Watch for drug-drug interactions, particularly with CYP3A4 inhibitors/inducers, which alter drug levels.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

