Mechanism of Action - Taming the Cytokine
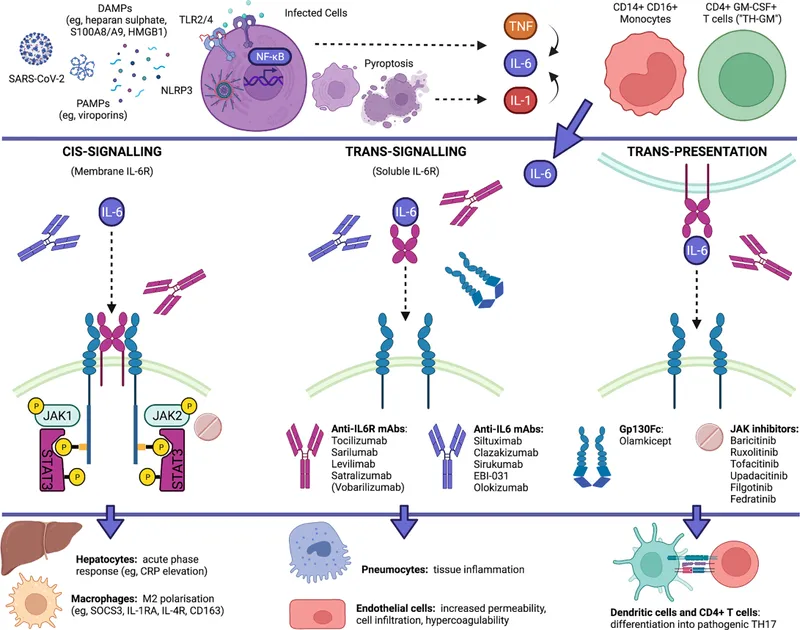
- Blocks IL-6 Signaling: Interleukin-6 (IL-6) is a key pro-inflammatory cytokine. These drugs are monoclonal antibodies that interrupt its signaling cascade, primarily through the JAK-STAT pathway.
- Two Main Targets:
- IL-6 Receptor (IL-6R): Tocilizumab & Sarilumab bind to membrane-bound and soluble IL-6R, preventing IL-6 from docking.
- IL-6 Cytokine: Siltuximab binds directly to the circulating IL-6 molecule itself.
- Downstream Effect: Both methods block JAK-STAT activation, leading to ↓ transcription of inflammatory genes and a rapid ↓ in acute-phase reactants (e.g., CRP, fibrinogen).
⭐ A notable risk with IL-6 inhibitors is an increased incidence of gastrointestinal perforation. The mechanism may involve impaired mucosal healing or masking of peritoneal irritation symptoms due to potent anti-inflammatory effects.
The 'mabs' & PK - Meet the Inhibitors
-
Tocilizumab (Actemra) & Sarilumab (Kevzara)
- Humanized monoclonal antibodies that bind to and inhibit IL-6 receptors (both soluble and membrane-bound).
- 📌 The "-li-" infix signifies an immunomodulatory agent.
-
Pharmacokinetics (PK) Profile
- Route: Tocilizumab is available as IV and SC injections. Sarilumab is SC only.
- Half-life: Long and concentration-dependent due to target-mediated drug disposition.
- Tocilizumab: ~11-13 days
- Sarilumab: ~8-10 days
- Metabolism: Degraded into small peptides and amino acids via catabolic pathways, similar to endogenous IgG. Not cleared by liver or kidneys.
⭐ Exam Pearl: Tocilizumab carries a notable black box warning for GI perforation, a risk potentially increased with concurrent use of NSAIDs or corticosteroids.
Clinical Uses - Quelling the Fire
-
Rheumatoid Arthritis (RA):
- For moderate to severe RA, particularly in patients with an inadequate response to DMARDs like TNF-α inhibitors.
- Can be used as monotherapy or with methotrexate.
-
Giant Cell Arteritis (GCA):
- Induces and maintains remission, acting as a crucial glucocorticoid-sparing agent.
- Significantly reduces cumulative steroid dose and toxicity.
-
Juvenile Idiopathic Arthritis (JIA):
- Approved for both Systemic JIA (sJIA) and Polyarticular JIA (pJIA).
-
Castleman Disease:
- Effective for multicentric Castleman disease (MCD).
⭐ Cytokine Release Syndrome (CRS): A first-line treatment for severe or life-threatening CRS, a major complication of CAR-T cell therapy. Tocilizumab is the indicated drug.
Adverse Effects & Monitoring - Watchful Waiting
- Infections: ↑ risk of serious infections.
- Upper respiratory tract infections are common.
- Screen for latent TB before starting therapy.
- Gastrointestinal:
- ⚠️ GI perforation: Risk is ↑ in patients with a history of diverticulitis.
- Nausea, diarrhea, or abdominal pain.
- Laboratory Abnormalities & Monitoring:
- Neutropenia: Monitor ANC (Absolute Neutrophil Count).
- Thrombocytopenia: Monitor platelet count.
- Elevated LFTs: Monitor ALT/AST; risk of hepatotoxicity.
- Dyslipidemia: ↑ LDL, HDL, and triglycerides. Monitor lipid panel 4-8 weeks after initiation.
⭐ Exam Favorite: Be cautious with IL-6 inhibitors in patients with a history of diverticulitis due to the heightened risk of bowel perforation.
High‑Yield Points - ⚡ Biggest Takeaways
- Tocilizumab and Sarilumab are key IL-6 receptor antagonists, blocking pro-inflammatory cytokine signaling.
- Key indications include rheumatoid arthritis, systemic juvenile idiopathic arthritis, and cytokine release syndrome (CRS).
- Major risk is immunosuppression, leading to serious infections; screen for latent TB before therapy.
- Monitor blood counts for neutropenia and thrombocytopenia, and LFTs for elevated liver enzymes.
- A rare but serious adverse effect is GI perforation.
- Can also cause dyslipidemia (elevated cholesterol and triglycerides).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

