Mechanism of Action - Taking the Brakes Off
T-cell activation requires two signals to attack. Cancer cells exploit inhibitory checkpoint pathways to apply the "brakes" on this process and evade the immune system.
- Signal 1 (Accelerator): T-cell receptor (TCR) on the T-cell binds to the Major Histocompatibility Complex (MHC) on an antigen-presenting cell (APC) or tumor cell.
- Signal 2 (Accelerator): Co-stimulatory protein B7 (CD80/86) on the APC binds to CD28 on the T-cell.
Inhibitory Checkpoints (Brakes):
- CTLA-4: On the T-cell surface, it binds to B7 with higher affinity than CD28, preventing co-stimulation.
- PD-1: On the T-cell surface, it binds to PD-L1 on tumor cells, inducing T-cell "exhaustion" and apoptosis.
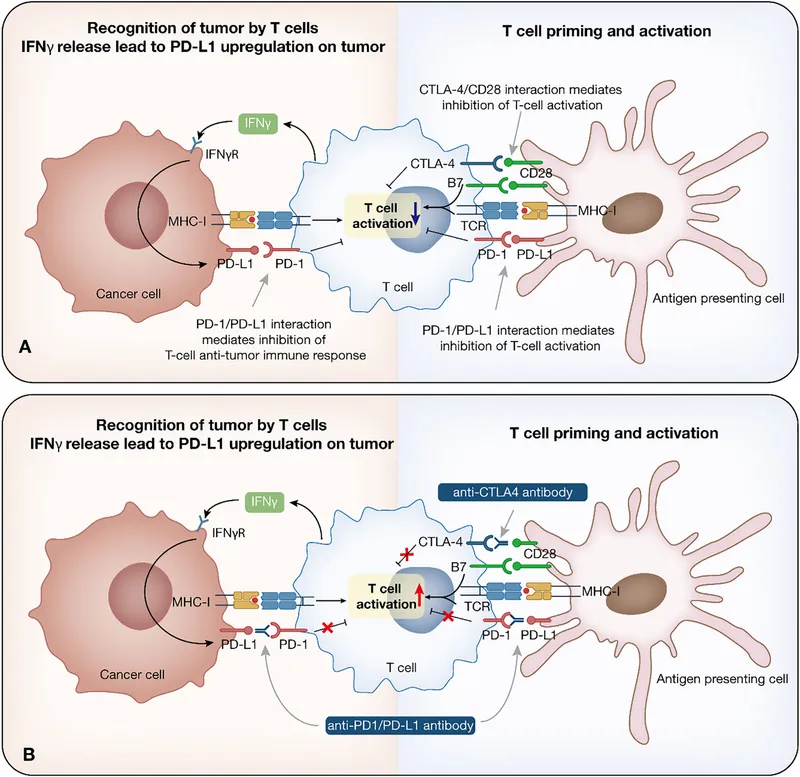
⭐ Cancers with high mutational burdens (e.g., melanoma, non-small cell lung cancer) often respond better to checkpoint inhibitors as they create more neoantigens for T-cells to recognize.
Checkpoint Inhibitors - The Major Players
Checkpoint inhibitors enhance the body's own immune system to fight cancer by blocking signals that suppress T-cell activity. Management of resulting immune-related adverse events (irAEs) typically involves corticosteroids.
| Class | Drug(s) | Key Indications | Unique/High-Yield Toxicities |
|---|---|---|---|
| CTLA-4 Ab | Ipilimumab | Melanoma, RCC | High risk of irAEs: severe colitis, dermatitis, hypophysitis, hepatitis. |
| PD-1 Ab | Nivolumab, Pembrolizumab | Melanoma, NSCLC, RCC, Hodgkin Lymphoma | irAEs are common but often less severe. Pneumonitis, thyroiditis, myocarditis are notable. |
| PD-L1 Ab | Atezolizumab, Durvalumab | Urothelial Carcinoma, NSCLC | Similar profile to PD-1 inhibitors; risk of infusion-related reactions. |
⭐ Pembrolizumab has a "tumor-agnostic" approval for any solid tumor with mismatch repair deficiency (dMMR) or high microsatellite instability (MSI-H).
Immune-Related AEs - When the Body Attacks
Checkpoint inhibitors can cause widespread inflammation by unleashing T-cells. Onset varies from weeks to months after starting therapy. Common manifestations include:
- Skin: Dermatitis, rash, pruritus (most common)
- GI: Colitis, diarrhea (can be severe, bloody)
- Liver: Hepatitis (asymptomatic ↑LFTs)
- Lung: Pneumonitis (cough, dyspnea, hypoxia)
- Endocrine: Hypophysitis, thyroiditis, adrenal insufficiency
Management Algorithm
⭐ Hypophysitis is a classic irAE of CTLA-4 inhibitors (e.g., Ipilimumab), presenting with headache and pituitary dysfunction.
High‑Yield Points - ⚡ Biggest Takeaways
- Checkpoint inhibitors restore anti-tumor immunity by blocking signals like PD-1, PD-L1, and CTLA-4.
- Key examples: Pembrolizumab (anti-PD-1), Ipilimumab (anti-CTLA-4), and Atezolizumab (anti-PD-L1).
- Major side effects are immune-related adverse events (irAEs) like colitis, hepatitis, and pneumonitis, managed with corticosteroids.
- CAR-T cell therapy engineers a patient's T-cells to attack cancer cells.
- Watch for Cytokine Release Syndrome (CRS) and neurotoxicity with CAR-T; treat CRS with Tocilizumab (IL-6 inhibitor).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

