AEFI Classification - The Reaction Spectrum
- Vaccine product-related: Caused or precipitated by a vaccine's inherent properties.
- Vaccine quality defect-related: Due to one or more quality defects of the vaccine product.
- Immunization error-related: Caused by inappropriate handling, prescribing, or administration.
- Immunization anxiety-related: Arising from anxiety about the immunization.
- Coincidental event: Caused by something other than the vaccine, error, or anxiety.
⭐ Programmatic errors (immunization error-related reactions) are the most common and most preventable cause of AEFIs.
Common AEFIs - Fever, Owies & Lumps
- Fever
- Most common systemic AEFI, esp. after DTP & Measles vaccines.
- Management: Paracetamol (10-15 mg/kg/dose).
- Injection Site Reactions (Pain, Swelling, Redness)
- Most common local AEFI.
- Management: Cold compress. Usually resolves in 1-3 days.
- Sterile Abscess: Common with DPT, DT, TT. Indurated, painless swelling. No I&D needed.
- BCG Lymphadenitis: Non-suppurative resolves spontaneously.
⭐ Suppurative BCG lymphadenitis should NOT be excised; needle aspiration is preferred. Antitubercular therapy is generally reserved for extra-axillary or disseminated disease.
Severe AEFIs - Rare But Real Risks
- Anaphylaxis: Acute, multi-system allergic reaction. Onset within 30 minutes.
- Management: IM Adrenaline (0.01 mg/kg, 1:1000 solution), max 0.5 mg.
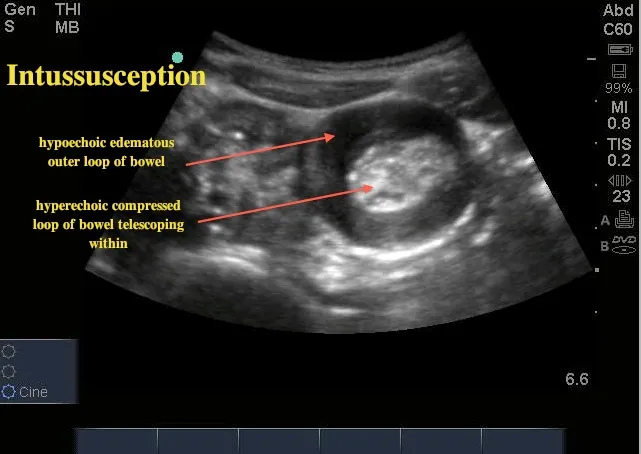
- Intussusception: Telescoping of the bowel, causing obstruction.
- Small ↑ risk with Rotavirus vaccines (RV1, RV5).
- Presents as colicky pain, vomiting, and red currant jelly stool.
- Thrombocytopenic Purpura (ITP):
- Associated with MMR vaccine. Onset 15-35 days post-vaccination.
- Hypotonic-Hyporesponsive Episode (HHE):
- Linked to Pertussis vaccine (DTwP > DTaP).
- Sudden pallor, limpness, ↓ responsiveness. Self-resolving.
- Vaccine-Associated Paralytic Poliomyelitis (VAPP):
- Caused by live Oral Polio Vaccine (OPV). Risk: ~1 in 2.7 million doses.
- Reason for global shift to Inactivated Polio Vaccine (IPV).
- Disseminated BCG Infection (BCG-osis):
- Occurs in infants with underlying immunodeficiency (e.g., SCID).
⭐ The highest risk of intussusception after the rotavirus vaccine is within the first 7 days following the first dose.
AEFI Surveillance in India - Reporting for Duty
- AEFI: Adverse Event Following Immunization. Any untoward medical occurrence post-immunization, which does not necessarily have a causal relationship with the vaccine.
- Reporting: Mandatory for all healthcare workers. Done via SAFEVAC (Surveillance and Action for Events Following Vaccination) portal.
⭐ All serious AEFIs (resulting in death, hospitalization, disability, or cluster) must be reported to the District Immunization Officer (DIO) within 24 hours.
High‑Yield Points - ⚡ Biggest Takeaways
- Most common AEFI is pain at the injection site; most common systemic symptom is fever.
- Fever is most common after the whole-cell Pertussis (wP) vaccine.
- Seizures are most linked to the wP component of the DPT vaccine.
- Anaphylaxis, the most severe reaction, occurs within 30 minutes of vaccination.
- Brachial neuritis is classically associated with the Tetanus toxoid.
- MMR vaccine is linked to thrombocytopenia and aseptic meningitis.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

