eConsent Tools - Pixels for Permission
- Definition: Use of digital media (tablets, videos, patient portals) to obtain and document informed consent.
- Advantages:
- ↑ Patient comprehension & engagement via interactive elements.
- Standardizes information delivery; reduces variability.
- Improves documentation, storage, and retrieval.
- Challenges:
- Digital Divide: May exclude patients with low digital literacy or no access.
- Requires robust data security (HIPAA compliance).
- Potential for software/hardware failure.
⭐ High-Yield Fact: Multimedia eConsent tools have been shown to significantly improve patient understanding and recall of procedural risks and benefits compared to traditional paper forms, which is a key factor in validating true informed consent.
eConsent Pros & Cons - A Digital Double-Edge
| Advantages (Pros) | Disadvantages (Cons) |
|---|---|
| * Improved Comprehension: - Interactive elements (videos, quizzes) can ↑ patient understanding over paper forms. | * Digital Divide: - Excludes patients with limited technology access or digital literacy, raising equity concerns. |
| * Standardization & Consistency: - Ensures all patients receive the same core information, reducing provider variability. | * Technical Barriers: - System crashes, software bugs, or poor user interface can disrupt the consent process. |
| * Enhanced Documentation: - Creates a clear, time-stamped digital record of the consent process, including what was viewed. | * Security & Privacy Risks: - Potential for HIPAA breaches if patient data is not properly encrypted and secured. |
| * Remote & Flexible Consenting: - Facilitates consent from off-site family members or in telemedicine settings. | * Impersonal Nature: - May reduce the crucial face-to-face dialogue between patient and provider, hindering rapport. |
Implementation & Compliance - Clicks with Conscience
- System Standards: Must comply with HIPAA for privacy and 21 CFR Part 11 for FDA-regulated trials, ensuring data integrity and security.
- Patient Verification: Robust identity confirmation is crucial.
- Methods: Two-factor authentication, security questions, or biometric data.
- Comprehension & Education:
- Tools must actively ensure patient understanding.
- Use plain language, interactive videos, and integrated "teach-back" quizzes.
- Secure Process:
- Digital signature must be securely captured and linked to the consent form.
- Provide the patient with a downloadable/printable copy.
⭐ The U.S. Electronic Signatures in Global and National Commerce (E-SIGN) Act gives e-signatures the same legal weight as handwritten ones, provided specific criteria are met.
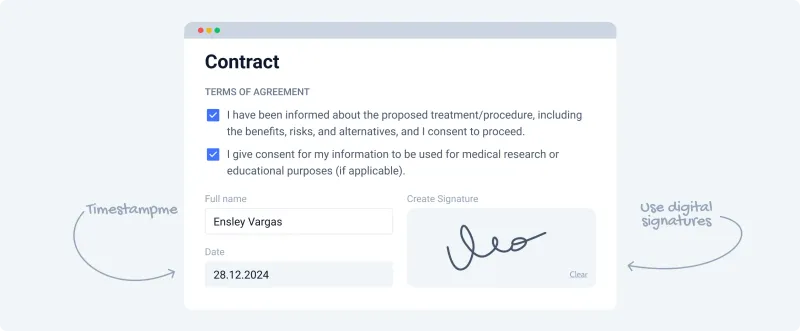
High‑Yield Points - ⚡ Biggest Takeaways
- Electronic consent is legally binding and can improve patient comprehension using videos or interactive modules.
- These tools standardize information delivery, reducing provider-to-provider variability and potential bias.
- They supplement, not replace, the core physician-patient discussion about risks, benefits, and alternatives.
- Clinicians must assess a patient's digital literacy and offer non-digital options when necessary.
- Proper documentation of the process is crucial for legal protection.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

