HIPAA & Research - The Privacy Rulebook
Patient authorization is the default for using Protected Health Information (PHI) in research. Key exceptions allow access while protecting privacy.
- Authorization Exceptions:
- De-identified Data: Not PHI if all 18 identifiers are removed. HIPAA's Privacy Rule does not apply.
- Limited Data Set (LDS): PHI with direct identifiers removed. Requires a Data Use Agreement (DUA).
- IRB/Privacy Board Waiver: Granted if research poses minimal privacy risk and obtaining authorization is impracticable.
⭐ Research preparatory activities (e.g., assessing study feasibility) and research on decedents' PHI are also permitted without individual authorization under specific HIPAA provisions.
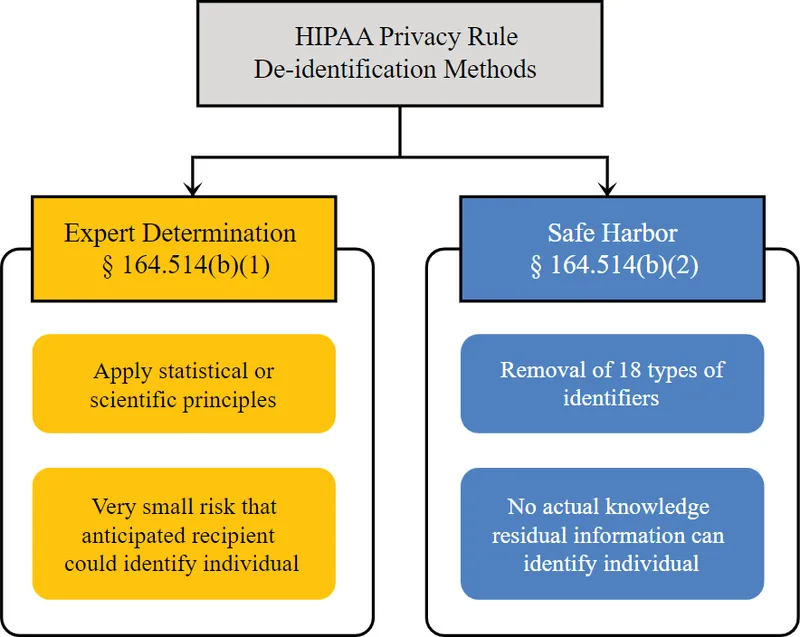
De-identification Methods - Anonymizing PHI
-
De-identified health information is not considered Protected Health Information (PHI) and is exempt from the HIPAA Privacy Rule. It is achieved via two pathways:
-
1. Safe Harbor Method:
- Mandates the removal of 18 specific personal identifiers.
- Key removals include names, all geographic units smaller than a state, all date elements (except year), medical record numbers, and biometric data.
- The provider must have no actual knowledge that the remaining information could identify the subject.
-
2. Expert Determination Method:
- A statistical expert applies methods to determine that the risk of re-identification is "very small."
- Requires formal documentation of the statistical analysis.
⭐ High-Yield: Under Safe Harbor, the first 3 digits of a zip code can be kept if the corresponding geographic area contains >20,000 people. All other geographic data smaller than a state must be removed.
Authorization & Waivers - Getting Permission
-
Authorization: The default method. Researchers must obtain signed, patient-specific permission to use or disclose Protected Health Information (PHI) for research.
- Core Elements: Plain language, specific purpose, expiration date, and right to revoke.
-
Waiver of Authorization: An Institutional Review Board (IRB) or Privacy Board can waive authorization if:
- Research poses minimal risk to privacy.
- Research is not practicable without the waiver.
- Waiver won't adversely affect patient rights.
⭐ Research is designed to create generalizable knowledge. Quality Improvement (QI) is for internal processes. This distinction determines if IRB oversight/waiver is needed.
-
Other Data Types:
- Limited Data Set: PHI with direct identifiers removed. Requires a Data Use Agreement (DUA).
- De-identified Data: All 18 HIPAA identifiers removed. No longer PHI; HIPAA rules do not apply.
-
Decision Flow:
High-Yield Points - ⚡ Biggest Takeaways
- De-identified health information is not PHI and can be used for research without authorization.
- Research with PHI requires either written patient authorization or a waiver of authorization from an IRB/Privacy Board.
- A Limited Data Set (LDS), which excludes direct identifiers, can be used for research with a Data Use Agreement (DUA).
- Authorization forms for research must be specific and are separate from the informed consent.
- Recruitment can involve PHI but is strictly regulated.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

