Aplastic Anemia - Empty Marrow Misery
- Pathogenesis: Autoimmune T-cell destruction of multipotent hematopoietic stem cells → pancytopenia with a hypocellular, fatty marrow ("empty marrow").
- Etiologies: Idiopathic (~70%), drugs (chloramphenicol, carbamazepine, NSAIDs), toxins (benzene), viral (Hepatitis, Parvovirus B19, EBV), radiation.
- Clinical & Labs: Pancytopenia symptoms (fatigue, infections, bleeding). ↓ Reticulocytes, ↑ EPO. No hepatosplenomegaly.
- Diagnosis: Gold standard is bone marrow biopsy → hypocellular marrow (<25%) replaced by fat.
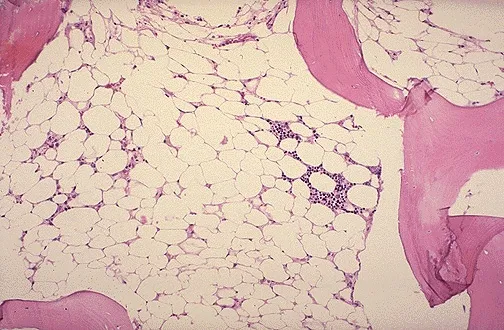
- Treatment: Supportive care (transfusions). Definitive: Allogeneic HSCT for young (<50 yrs), immunosuppression (ATG, cyclosporine) for older/unfit.
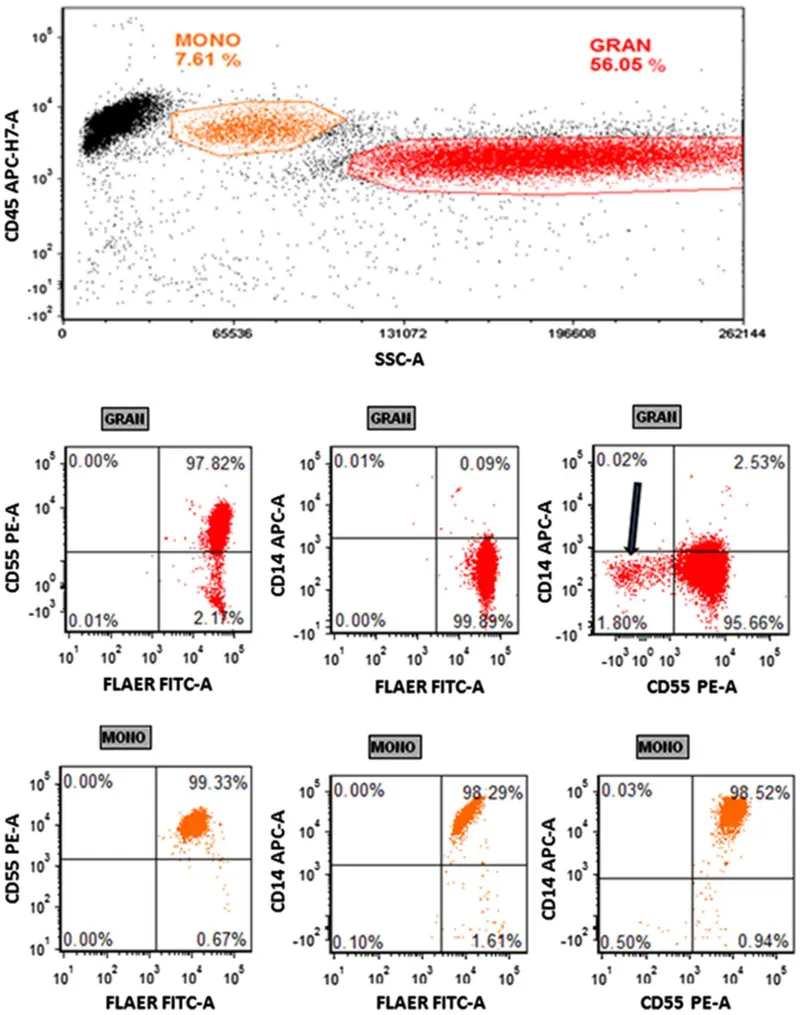
⭐ Up to 50% of aplastic anemia patients may have a co-existing Paroxysmal Nocturnal Hemoglobinuria (PNH) clone, detectable by flow cytometry for CD55/CD59.
PNH - Defective Anchor Drama
- Acquired somatic mutation in the PIGA gene of a hematopoietic stem cell, leading to an inability to synthesize glycosylphosphatidylinositol (GPI) anchors.
- Cells lack GPI-anchored proteins like CD55 (Decay Accelerating Factor) & CD59 (MAC Inhibitory Protein), making them susceptible to complement-mediated destruction.
- Classic Triad: Intravascular hemolysis (dark urine, esp. in AM), pancytopenia, and venous thrombosis (often in atypical sites).
- Diagnosis: Flow cytometry showing absent CD55/CD59 on RBCs and granulocytes.
⭐ A major cause of mortality in PNH is thrombosis, particularly in unusual locations like hepatic (Budd-Chiari syndrome), portal, or cerebral veins.
- Treatment: C5 inhibitors (Eculizumab, Ravulizumab).
Myelodysplastic Syndromes - Dysfunctional Factory
Clonal hematopoietic stem cell disorders with ineffective hematopoiesis, leading to peripheral cytopenias despite a hypercellular bone marrow. It's a "factory" producing faulty cells.
- Hallmark: Dysplasia in ≥1 cell line.
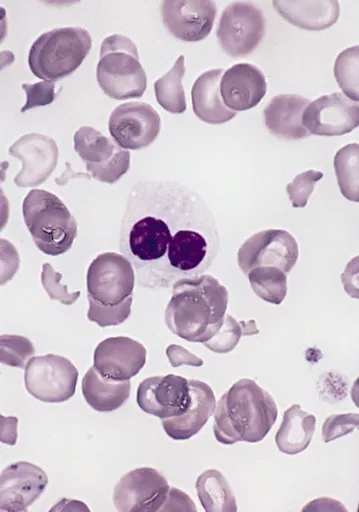
- Myeloid: Pseudo-Pelger-Huët cells (bilobed neutrophils).
- Erythroid: Ring sideroblasts.
- Megakaryocytic: Small, separated nuclei (pawn-ball megakaryocytes).
- Diagnosis: Bone marrow biopsy shows hypercellularity, dysplasia, and < 20% blasts.
- Risk: Transformation to AML (defined by ≥ 20% blasts).
⭐ 5q- Syndrome: A specific MDS subtype, often in older women, with macrocytic anemia, normal/↑ platelets, and a favorable prognosis.
Inherited Syndromes - Faulty Blueprints
-
Fanconi Anemia (FA)
- Defect in DNA crosslink repair (e.g., BRCA genes).
- Presents with progressive pancytopenia, macrocytic anemia.
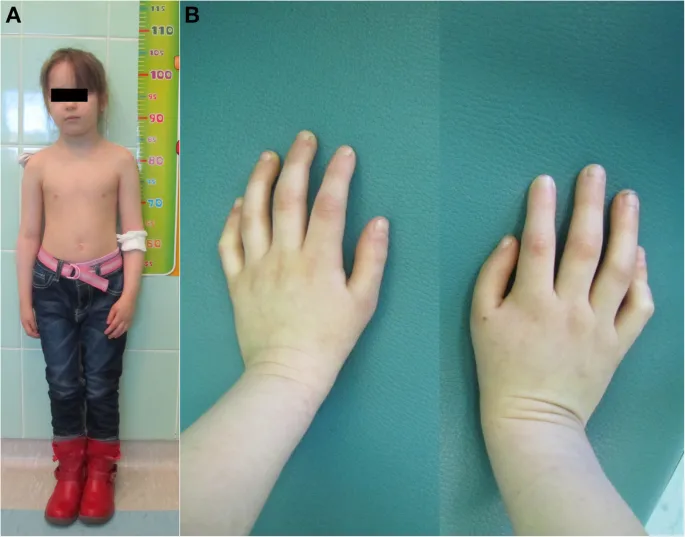
- Associated with café-au-lait spots, short stature, and thumb/radial defects.
- ↑ risk of AML, MDS, and solid tumors.
⭐ Chromosomal breakage analysis after exposure to DNA cross-linking agents (e.g., diepoxybutane) is diagnostic.
-
Diamond-Blackfan Anemia (DBA)
- Congenital pure red cell aplasia due to ribosomal protein gene mutations.
- Presents in infancy with macrocytic anemia, reticulocytopenia.
- Craniofacial and thumb abnormalities (triphalangeal thumb).
-
Dyskeratosis Congenita (DC)
- Defective telomere maintenance (TERC, TERT genes).
- Classic triad: abnormal skin pigmentation, nail dystrophy, oral leukoplakia.
- 📌 Mnemonic: CAN of worms: Cancer, Aplastic anemia, Nail/skin changes.
High‑Yield Points - ⚡ Biggest Takeaways
- Aplastic anemia presents with pancytopenia and a hypocellular bone marrow; key causes include drugs, viruses, and autoimmune damage.
- Fanconi anemia (AR) combines pancytopenia with café-au-lait spots and thumb/radial defects; diagnosed by chromosomal breakage tests.
- Diamond-Blackfan anemia is a congenital pure red cell aplasia with macrocytic anemia, low reticulocytes, and physical anomalies.
- PNH (PIGA mutation) causes hemolysis, thrombosis, and pancytopenia from CD55/CD59 deficiency.
- Myelodysplastic syndromes (MDS) feature cytopenias despite a hypercellular marrow; carries a high risk of AML transformation.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

