DM Diagnosis - The Sweet Numbers
-
Definition: A state of chronic hyperglycemia due to defects in insulin secretion, action, or both.
-
Diagnostic Criteria: Any of the following. Confirmation often requires a repeat test.
- HbA1c: ≥6.5%
- Fasting Plasma Glucose (FPG): ≥126 mg/dL (no caloric intake for ≥8 hrs)
- 2-hr Oral Glucose Tolerance Test (OGTT): ≥200 mg/dL (post 75g glucose load)
- Random Plasma Glucose (RPG): ≥200 mg/dL (with classic symptoms like polyuria, polydipsia)
⭐ HbA1c is advantageous as it doesn't require fasting and reflects long-term glycemic control (2-3 months).

Type 1 DM Pathophysiology - Beta Cell Betrayal
- Autoimmune Attack: A chronic, T-cell mediated (Type IV hypersensitivity) assault on pancreatic β-cells.
- Genetic Susceptibility: Strongly linked to specific HLA genes, notably HLA-DR3 and HLA-DR4.
- 📌 DR 3 & 4 are the doors to T1D.
- Key Markers: Autoantibodies are present in >85% of newly diagnosed patients.
- Anti-glutamic acid decarboxylase (anti-GAD65)
- Islet cell autoantibodies (ICA)
- Histopathology: Characterized by "insulitis"-a lymphocytic infiltrate within pancreatic islets.
- End Result: Absolute deficiency of insulin.
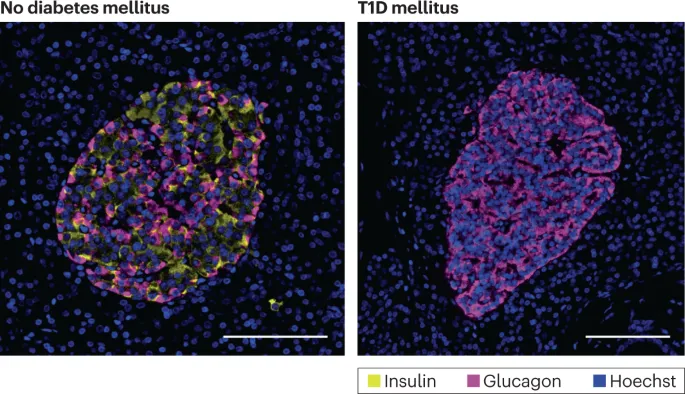
⭐ Exam Favorite: Autoantibodies (e.g., GAD65) are often detectable years before hyperglycemia manifests, marking a preclinical phase of active, ongoing β-cell destruction.
Type 2 DM Pathophysiology - Resistance is Futile
-
Insulin Resistance: The cornerstone defect where peripheral tissues (muscle, liver, adipose) show a diminished response to normal insulin levels.
- Key Drivers: Central obesity, metabolic syndrome, and strong genetic predisposition are major risk factors.
- Adipokine Role: Dysregulated secretion from visceral fat; ↓ adiponectin (sensitizer) and ↑ resistin (antagonist) worsen resistance.
-
Relative Insulin Deficiency: Occurs as pancreatic β-cells fail to meet the demand.
- β-cell Exhaustion: Initially, hyperinsulinemia compensates for resistance. Over time, β-cells become exhausted, leading to dysfunction and ↓ insulin secretion.
- Amyloid Deposition: Islet Amyloid Polypeptide (IAPP) co-secreted with insulin deposits in islets, further impairing β-cell function.
⭐ Glucagon levels are often paradoxically elevated in T2DM, exacerbating hyperglycemia by driving unopposed hepatic glucose production.
Chronic Complications - The Sugar Fallout
-
Microvascular Damage
- Retinopathy: Non-proliferative (hemorrhages, exudates) & proliferative (neovascularization).
- Nephropathy: Nodular glomerulosclerosis (Kimmelstiel-Wilson lesions).
- Neuropathy: Peripheral "stocking-glove" sensory loss; autonomic dysfunction (gastroparesis).
-
Macrovascular Damage
- Accelerated atherosclerosis leading to:
- Coronary Artery Disease (CAD) → Myocardial Infarction
- Peripheral Vascular Disease (PVD) → Limb ischemia
- Cerebrovascular Accident (CVA) → Stroke
- Accelerated atherosclerosis leading to:

⭐ HbA1c reflects average blood glucose over the prior ~3 months because the glycation of hemoglobin is irreversible, mirroring the red blood cell lifespan.
High‑Yield Points - ⚡ Biggest Takeaways
- Type 1 DM is an autoimmune process destroying pancreatic β-cells, strongly associated with HLA-DR3/DR4.
- Type 2 DM is defined by insulin resistance and later, relative insulin deficiency, with characteristic islet amyloid deposition.
- Chronic complications are driven by non-enzymatic glycosylation (AGEs) and the polyol pathway.
- Kimmelstiel-Wilson lesions (nodular glomerulosclerosis) are pathognomonic for diabetic nephropathy.
- Accelerated atherosclerosis is the major cause of morbidity/mortality.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

