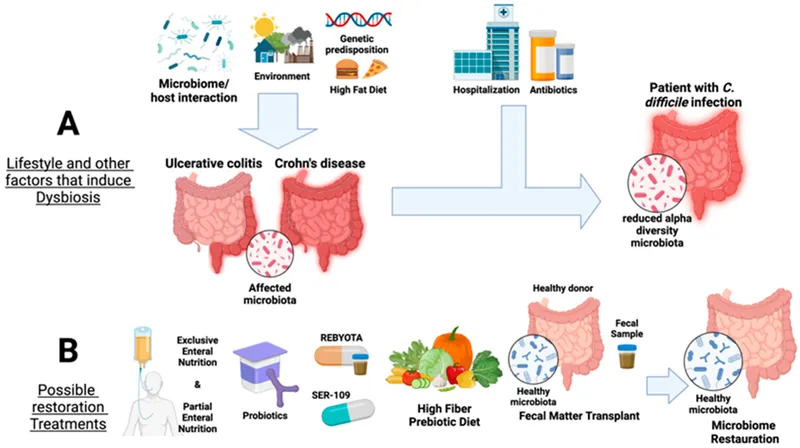
Normal Gut Flora - The Body's Inner Ecosystem
- Composition: A dense, diverse community of primarily anaerobic bacteria (Bacteroides, Firmicutes) that create a barrier against pathogens.
- Key Functions:
- Metabolic: Synthesizes Vitamin K, biotin; ferments complex carbs into Short-Chain Fatty Acids (SCFAs) like butyrate (colonocyte fuel).
- Protective: Prevents pathogen colonization through competition for space and nutrients.
- Immunologic: Modulates development of Gut-Associated Lymphoid Tissue (GALT).
⭐ Broad-spectrum antibiotics like Clindamycin, Ampicillin, and Cephalosporins are most frequently implicated in causing pseudomembranous colitis.
Pathophysiology - When Antibiotics Attack
- Antibiotic Action: Broad-spectrum antibiotics (e.g., clindamycin, cephalosporins, fluoroquinolones) eradicate susceptible gut commensals.
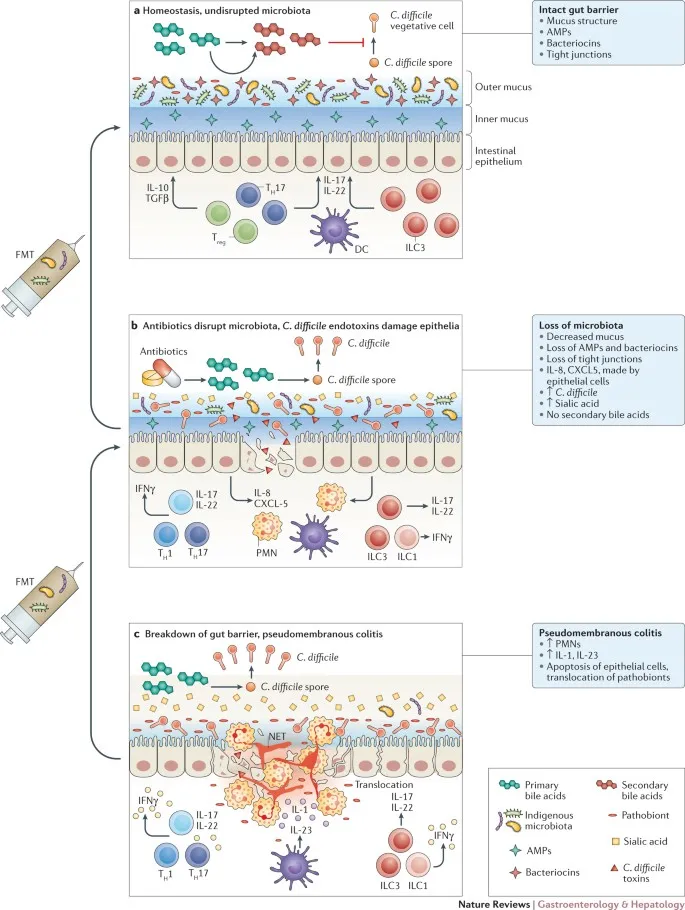
- Dysbiosis: This disrupts the normal microbiome, reducing metabolic competition and protective bacteriocins.
- Leads to ↓ loss of colonization resistance.
- Allows antibiotic-resistant organisms like Clostridioides difficile to proliferate from a spore state.
- C. difficile Pathogenesis:
- Vegetative cells produce exotoxins.
- Toxin A (enterotoxin): Causes inflammation and fluid secretion.
- Toxin B (cytotoxin): More potent; disrupts cytoskeleton leading to cell death.
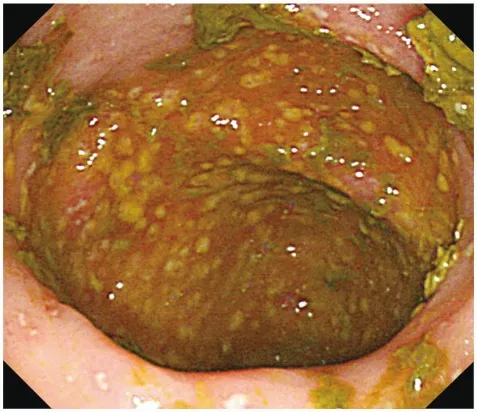
- Toxins cause mucosal injury, neutrophil infiltration, and formation of characteristic pseudomembranes.
- Vegetative cells produce exotoxins.
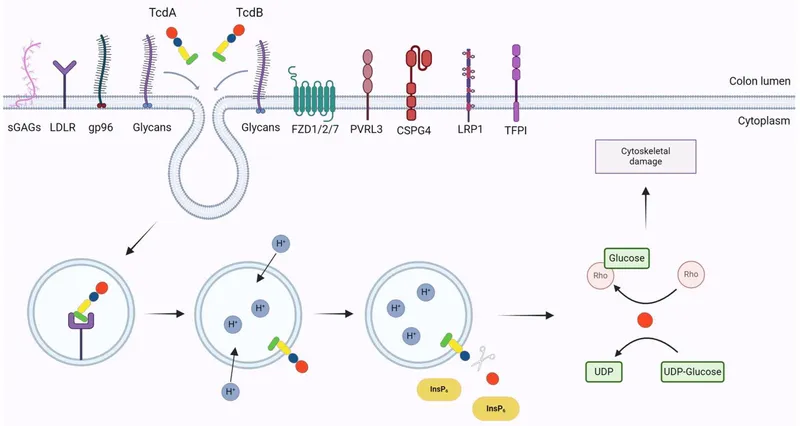
⭐ C. difficile toxins glucosylate and inactivate Rho family GTPases, leading to actin cytoskeleton disruption, epithelial cell death, and loss of intestinal barrier function.
C. difficile - The Main Offender
- Pathogen: Gram-positive, spore-forming, obligate anaerobe rod.
- Pathogenesis: Antibiotic use (esp. clindamycin, cephalosporins, fluoroquinolones) disrupts normal colonic flora, allowing C. difficile overgrowth and toxin production.
- Toxin A (Enterotoxin): Binds to the brush border, causing inflammation, fluid secretion, and watery diarrhea.
- Toxin B (Cytotoxin): More potent; causes cytoskeletal disruption, leading to cell death and the formation of characteristic pseudomembranes (yellow-white plaques of fibrin, inflammatory cells, and necrotic debris).
- Complications: Pseudomembranous colitis, toxic megacolon, bowel perforation.
⭐ Diagnosis: Nucleic Acid Amplification Tests (NAATs) are the preferred method for detecting C. difficile toxin genes in stool samples due to their high sensitivity and specificity.
Diagnosis & Management - The Counter-Attack
-
Diagnosis:
- Primary: Stool Nucleic Acid Amplification Test (NAAT) for C. difficile toxin genes.
- Enzyme immunoassay (EIA) for toxins A/B is less sensitive but highly specific.
- 💡 Avoid repeat testing within 7 days during the same episode.
-
Management Algorithm:
- Advanced/Refractory Cases:
- Fecal Microbiota Transplant (FMT): Restores gut microbial diversity to prevent spore germination.
- Bezlotoxumab: Monoclonal antibody against Toxin B, used adjunctively.
⭐ For multiple recurrences, Fecal Microbiota Transplant (FMT) shows superior efficacy (>80% cure rate) compared to standard antibiotic tapers by re-establishing a resilient microbiome.
High‑Yield Points - ⚡ Biggest Takeaways
- Broad-spectrum antibiotics disrupt the protective gut microbiota, creating an opportunity for pathogen overgrowth.
- Clostridioides difficile, a spore-forming anaerobe, is the most common cause.
- Toxins A (enterotoxin) and B (cytotoxin) mediate colonic mucosal injury, leading to inflammation and diarrhea.
- The classic presentation is pseudomembranous colitis.
- Diagnosis is confirmed via stool toxin assays or nucleic acid amplification tests (NAAT).
- Treatment mainstays are oral vancomycin or fidaxomicin.
- Fecal microbiota transplantation (FMT) is highly effective for recurrent cases.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

