Beta-Lactamase Basics - The Ring Breakers
- Core Function: Bacterial enzymes that confer resistance by breaking the central beta-lactam ring of antibiotics like penicillins and cephalosporins.
- Mechanism: They perform hydrolysis of the amide bond within the ring, rendering the antibiotic molecule inactive and unable to bind to its target (penicillin-binding proteins).
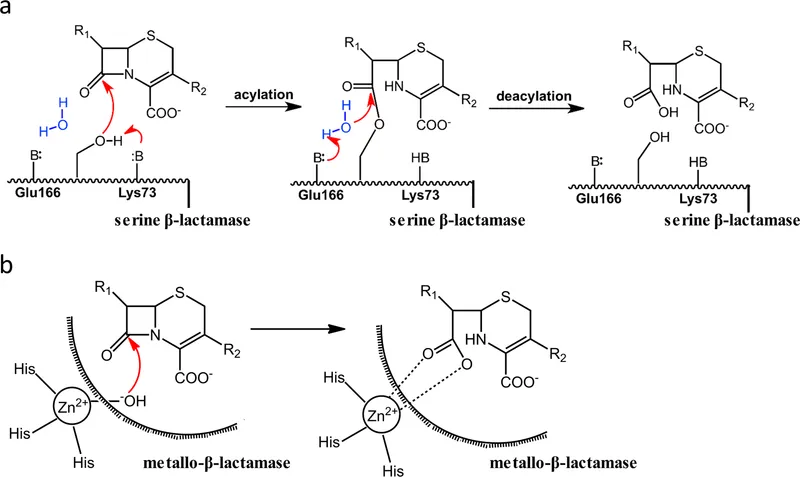
⭐ Some bacteria export beta-lactamases into the periplasmic space, inactivating antibiotics before they can reach their targets in the cell wall.
Ambler Classification - A, B, C, D of Destruction
- Categorizes β-lactamases by amino acid sequence. Classes A, C, and D are serine hydrolases; Class B are metallo-enzymes.
| Class | Mechanism | Key Examples & Notes | Inhibited By |
|---|---|---|---|
| A | Serine-based | TEM, SHV, CTX-M (ESBLs), KPC | Clavulanate, Avibactam |
| B | Metallo (Zinc) | NDM-1, VIM, IMP (MBLs) | None (Aztreonam stable) |
| C | Serine-based | AmpC (inducible cephalosporinases) | Avibactam |
| D | Serine-based | OXA-type (in Acinetobacter) | Variable |
⭐ Class B Metallo-β-lactamases (MBLs) require zinc cofactors and are not inhibited by common β-lactamase inhibitors like clavulanate or tazobactam, making them a significant resistance threat.
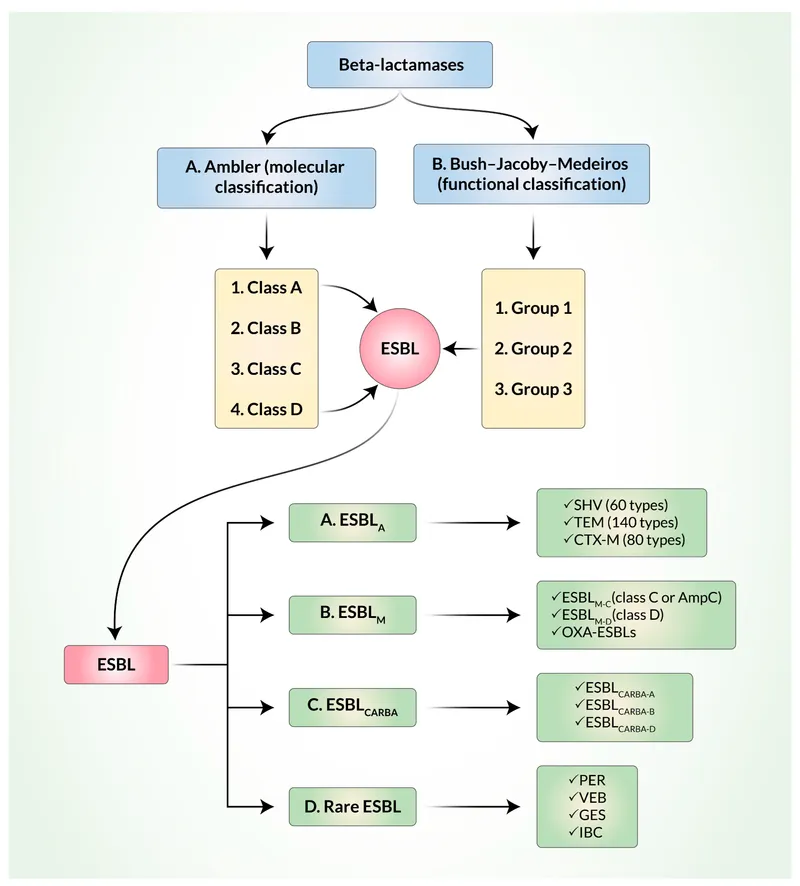
Clinically Key Players - The Usual Suspects
-
Simple Penicillinases: Common, often plasmid-mediated, conferring resistance to basic penicillins.
- Staphylococcus aureus (penicillinase)
- Haemophilus influenzae
- Neisseria gonorrhoeae
- Enterobacteriaceae (e.g., E. coli, carrying TEM-1)
-
Extended-Spectrum β-Lactamases (ESBLs): Hydrolyze most penicillins, cephalosporins (1st, 2nd, 3rd gen), and monobactams.
- Primarily in Klebsiella pneumoniae and E. coli.
- 📌 Mnemonic (ESBL): "Escape Some Big Lactams".
-
Carbapenemases (e.g., KPC, NDM, OXA): The most versatile; hydrolyze carbapenems and nearly all other β-lactams.
- Defines Carbapenem-Resistant Enterobacteriaceae (CRE): Klebsiella, E. coli.
- Also key in multi-drug resistant Pseudomonas aeruginosa and Acinetobacter baumannii.
⭐ ESBL infections are a major clinical challenge; they are resistant to most cephalosporins and aztreonam. Carbapenems are the typical treatment, but carbapenemase-producing strains (CRE) are an urgent threat.
Inhibitor Counter-Attack - The Suicide Squad
- 📌 CAST of common inhibitors: Clavulanic Acid, Avibactam, Sulbactam, Tazobactam.
- Mechanism: Structurally resemble β-lactam molecules, acting as "suicide" substrates.
- They irreversibly bind to the β-lactamase enzyme's active site.
- This sacrificial inactivation protects the co-administered antibiotic from destruction.
⭐ Avibactam is a notable exception; it's a non-β-lactam inhibitor that binds reversibly. It has broader activity, covering Klebsiella pneumoniae carbapenemase (KPC) and AmpC-type enzymes.
High‑Yield Points - ⚡ Biggest Takeaways
- β-lactamases are the primary mechanism of bacterial resistance to β-lactam antibiotics.
- Penicillinases and cephalosporinases are simple β-lactamases; Extended-Spectrum β-Lactamases (ESBLs) inactivate most penicillins and cephalosporins.
- ESBLs are commonly plasmid-encoded in Klebsiella pneumoniae and E. coli.
- Carbapenemases (e.g., KPC, NDM) confer resistance to nearly all β-lactams, including carbapenems.
- β-lactamase inhibitors (e.g., clavulanate, tazobactam) can overcome many forms of this resistance.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

