Indications - To Test or Not?
-
When to Perform Genotype Assay:
- ART-Naïve: At entry into care to guide initial regimen selection.
- Virologic Failure: When viral load rebounds on ART (>200 copies/mL) to guide regimen changes.
- Pregnancy: Before starting ART or if virologic failure occurs.
- Suboptimal viral suppression after starting therapy.
-
When NOT to Test:
- Not reliable if viral load is <500 copies/mL, as amplification may fail.
- After discontinuing ART, as resistant strains may become undetectable.
⭐ Genotypic assays are the standard for routine testing. Phenotypic assays are reserved for complex cases with extensive resistance, often requiring expert consultation.
Assay Types - Genes vs. Phenes
| Feature | Genotypic Assay | Phenotypic Assay |
|---|---|---|
| Principle | Detects drug-resistance mutations in viral genes (e.g., pol for RT, PR, IN). | Measures viral replication in the presence of antiretroviral drugs. |
| Method | Sanger or Next-Generation Sequencing of viral RNA. | Recombinant virus culture with drug titration. |
| Turnaround | Faster: 1-2 weeks. | Slower: 2-4 weeks. |
| Cost | Lower. | Higher. |
| Sensitivity | Requires viral load >500-1000 copies/mL. Can detect minor variants. | Requires viral load >1000 copies/mL. Less sensitive to minor variants. |
| Utility | Standard for initial testing; infers resistance from known mutations. | "Gold standard" for complex cases; directly measures drug susceptibility. |
⭐ Exam Favorite: Genotypic testing is the preferred method for initial resistance assessment in treatment-naïve patients and for initial virologic failure due to its speed and lower cost. Phenotypic tests are reserved for complex cases with extensive treatment history.
Interpreting Reports - Decoding Mutations
-
Genotypic reports identify specific amino acid substitutions in viral genes (e.g., pol).
-
Mutation nomenclature: M184V = Methionine (M) at position 184 is replaced by Valine (V).
-
Interpretation relies on databases (e.g., Stanford HIVDB) to correlate mutations with drug resistance levels.
-
Key Resistance Mutations by Class:
- NRTIs: M184V (major 3TC/FTC resistance), K65R (broad NRTI resistance), Thymidine Analogue Mutations (TAMs).
- NNRTIs: K103N (class-wide resistance), Y181C, G190A.
- Protease Inhibitors (PIs): L90M, I84V (major mutations).
- Integrase Inhibitors (INSTIs): N155H, Q148H/R/K.
⭐ The M184V mutation, while conferring high-level resistance to Lamivudine/Emtricitabine, impairs viral replication and can paradoxically increase susceptibility to Tenofovir and Zidovudine.
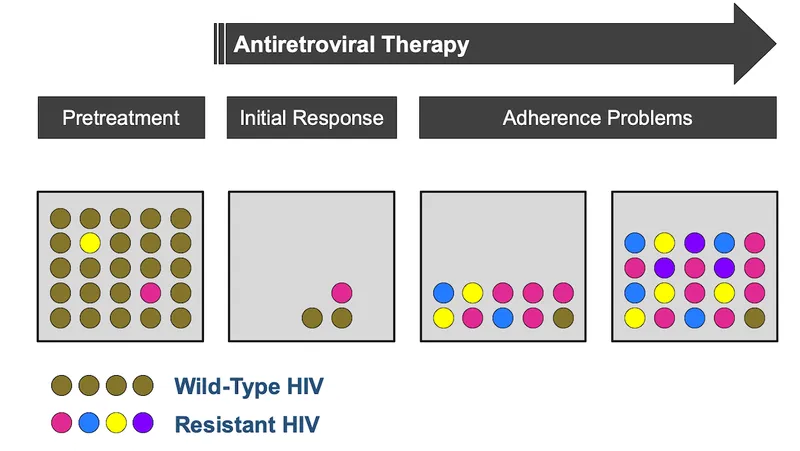
- HIV resistance testing is crucial at entry into care and upon virologic failure to guide antiretroviral therapy (ART).
- Genotypic assays, the standard initial test, detect resistance-conferring mutations in the pol gene (reverse transcriptase, protease, integrase).
- They are faster and cheaper than phenotypic tests but require expert interpretation.
- Phenotypic assays measure the concentration of a drug needed to inhibit viral growth, directly assessing resistance.
- Phenotyping is reserved for complex resistance patterns or after failure of multiple ART regimens.
- Tests may not detect minority resistance variants present at <20% of the viral population.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

