Biologics Overview - Targeted Takedown
- Monoclonal antibodies engineered to neutralize specific inflammatory mediators.
- Major classes for IBD:
- Anti-TNF (e.g., Infliximab)
- Anti-Integrin (e.g., Vedolizumab)
- Anti-IL-12/23 (e.g., Ustekinumab)
- Used for moderate-to-severe disease, often after conventional therapy fails. Requires pre-screening for infections.
⭐ Screen for latent TB before starting anti-TNF agents due to high risk of reactivation.
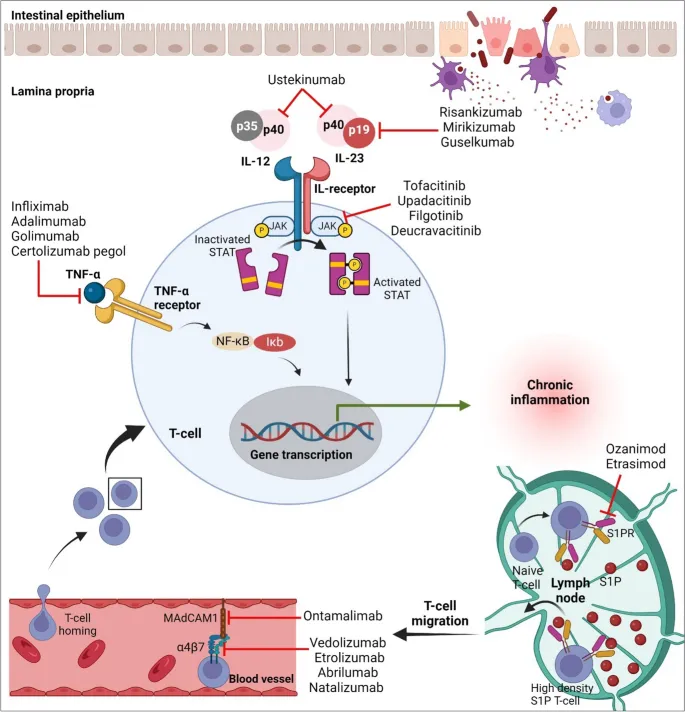
Anti-TNF-α Agents - The TNF Blockade
- Mechanism: Monoclonal antibodies that bind and neutralize TNF-α, preventing it from binding to its receptors and triggering pro-inflammatory signaling.
- Agents:
- Infliximab (chimeric)
- Adalimumab (human)
- Certolizumab pegol (humanized Fab' fragment; approved for Crohn's)
- Golimumab (human; approved for UC)
- Indications: Moderate-to-severe IBD (both UC and Crohn's) refractory to conventional therapy; fistulizing Crohn's disease.
- Key Adverse Effects:
- ↑ Infection risk (esp. reactivation of latent TB)
- Infusion/injection site reactions
- Demyelinating diseases
- Worsening heart failure
⭐ High-Yield: Always screen for latent TB (PPD or IGRA) and Hepatitis B virus before initiating anti-TNF-α therapy due to risk of reactivation.
Anti-Integrins - Gut-Specific Gatekeepers
- Mechanism: Selectively block gut-homing lymphocytes.
- Vedolizumab binds to the α4β7 integrin on T-cells.
- Prevents their interaction with MAdCAM-1 on gut endothelial cells.
- This inhibits lymphocyte trafficking into the GI tract lamina propria.
- Use: Moderate-to-severe Ulcerative Colitis & Crohn's Disease, especially after anti-TNF failure.
- Key Advantage: Gut-selective action minimizes systemic immunosuppression.
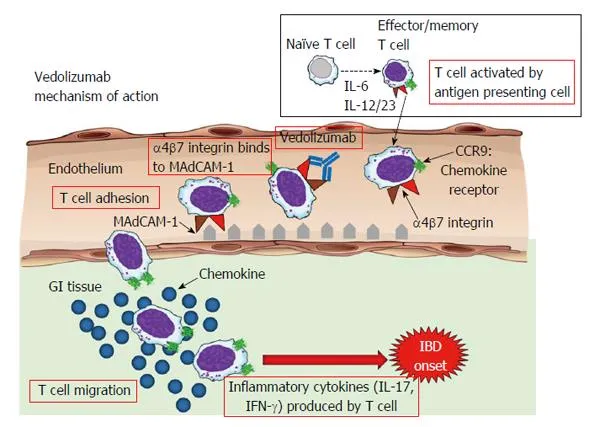
⭐ High-Yield: Unlike Natalizumab (α4β1/α4β7), Vedolizumab's gut-specificity (α4β7) spares CNS immune surveillance, virtually eliminating the risk of Progressive Multifocal Leukoencephalopathy (PML).
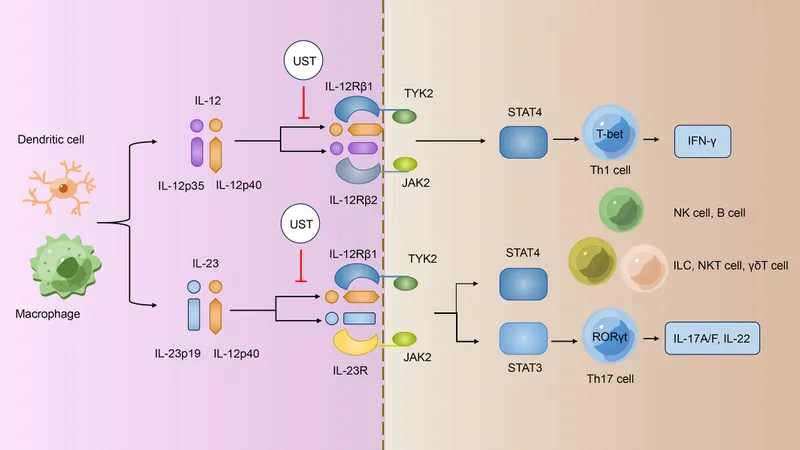
Anti-IL-12/23 Agents - Cytokine Control
- Ustekinumab: Human monoclonal antibody targeting the p40 subunit shared by Interleukin-12 (IL-12) and Interleukin-23 (IL-23).
- Mechanism:
- Blocks IL-12 → ↓ Th1 cell differentiation.
- Blocks IL-23 → ↓ Th17 cell pathway activation.
- Use: Moderate-to-severe Crohn's & Ulcerative Colitis, especially after anti-TNF failure.
- Risks: ↑ Infection risk (TB screening required), nasopharyngitis; rare reversible posterior leukoencephalopathy syndrome (RPLS).
⭐ By targeting the common p40 subunit, ustekinumab uniquely disrupts both the Th1 and Th17 inflammatory axes.
Pre-Biologic Screening - Safety First Protocol
- Core Principle: Identify and treat latent infections to prevent reactivation, especially with TNF-α inhibitors.
- Tuberculosis (TB) Screening: Mandatory.
- Interferon-Gamma Release Assay (IGRA) or Tuberculin Skin Test (PPD).
- Positive screen requires a chest X-ray to exclude active TB.
- Viral Hepatitis & HIV Screening:
- Hepatitis B panel: HBsAg, anti-HBs, anti-HBc.
- Hepatitis C antibody.
- HIV test.
- Vaccination Update:
- Ensure all vaccinations are current.
- ⚠️ Administer live vaccines ≥4 weeks BEFORE biologic initiation.
⭐ TNF-α inhibitors carry a high risk of reactivating latent TB. If screening is positive, treatment for latent TB (e.g., Isoniazid) must be initiated before starting the biologic.
High-Yield Points - ⚡ Biggest Takeaways
- Anti-TNF agents (e.g., infliximab) require screening for latent TB before initiation due to reactivation risk.
- They can also exacerbate congestive heart failure and increase risk for invasive fungal infections.
- Vedolizumab is a gut-specific anti-integrin (α4β7) with fewer systemic immunosuppressive effects than anti-TNF agents.
- Natalizumab carries a high risk of Progressive Multifocal Leukoencephalopathy (PML), limiting its use.
- Ustekinumab targets IL-12 and IL-23 and is effective for both Crohn's and ulcerative colitis.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

