Pathophysiology - Autoimmune Ambush
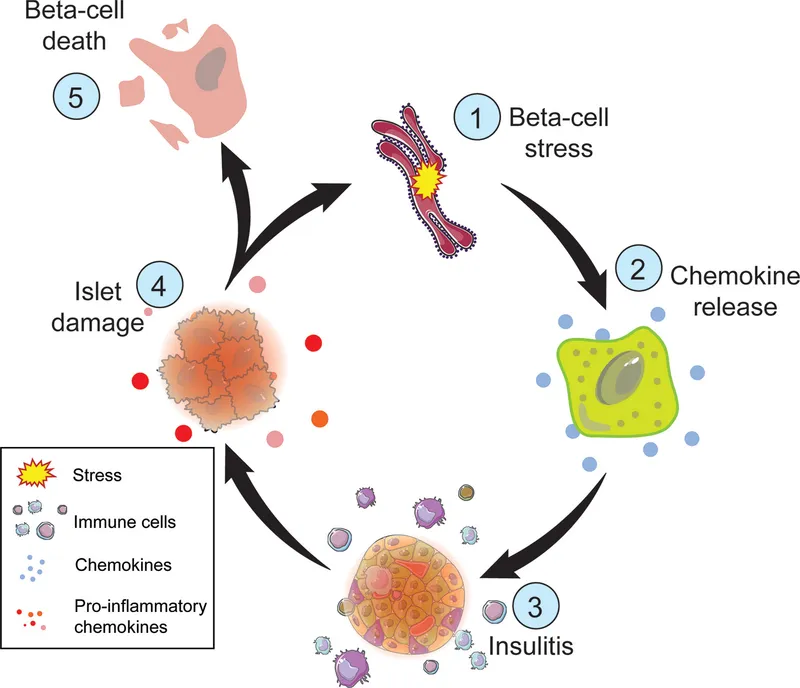
- Genetic Predisposition: Strong link to specific HLA genes, particularly HLA-DR3 and HLA-DR4.
- Mechanism: A trigger (e.g., viral infection) in a susceptible individual initiates a T-cell-mediated autoimmune assault on pancreatic β-cells.
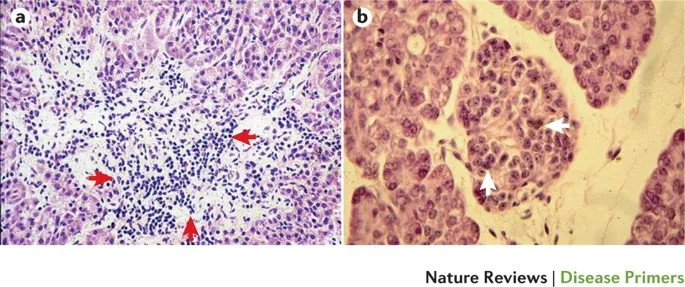
- CD8+ cytotoxic T-lymphocytes infiltrate islets (insulitis), leading to β-cell destruction.
- Results in absolute insulin deficiency.
- Key Markers: Autoantibodies like anti-GAD65, ICA, and IAA serve as markers of autoimmunity.
⭐ Over 90% of individuals with Type 1 Diabetes possess either HLA-DR3 or HLA-DR4 haplotypes.
Clinical Presentation - The Sweet Symptoms
- Classic Triad (The "3 Ps"):
- Polyuria: Osmotic diuresis (renal glucose threshold > 180 mg/dL).
- Polydipsia: Intense thirst from dehydration.
- Polyphagia: Hunger from cellular starvation.
- Weight Loss: Unexplained, due to catabolism (muscle, fat breakdown).
- Diabetic Ketoacidosis (DKA): Often the first sign, with nausea, vomiting, abdominal pain, & Kussmaul respirations.
⭐ High-Yield: About 25-30% of children with new-onset T1D first present in Diabetic Ketoacidosis (DKA).

Diagnosis - Cracking the Code
-
Core Criteria: Diagnosis requires one of the following:
- Fasting Plasma Glucose (FPG) ≥ 126 mg/dL
- 2-hour OGTT plasma glucose ≥ 200 mg/dL
- HbA1c ≥ 6.5%
- Random plasma glucose ≥ 200 mg/dL with classic hyperglycemia symptoms.
-
Antibody Panel: To confirm autoimmune etiology vs. T2DM.
- Most common: GAD65, Islet antigen-2 (IA-2).
⭐ Low or undetectable C-peptide levels are a hallmark, confirming scant endogenous insulin production, unlike in early T2DM where it can be normal or high.
Management - The Insulin Imperative
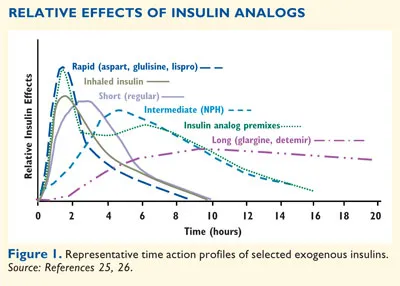
- Lifelong insulin replacement is mandatory. The goal is to mimic physiological insulin secretion.
- Basal-Bolus Regimen (Gold Standard):
- Basal: Once/twice daily long-acting insulin (Glargine, Detemir) or continuous subcutaneous infusion (insulin pump). Covers ~50% of daily needs.
- Bolus: Rapid-acting insulin (Lispro, Aspart) before each meal to control postprandial glucose. Covers the remaining ~50%.
- Initial Dosing: Typically 0.4-1.0 units/kg/day.
- Monitoring:
- Self-monitoring of blood glucose (SMBG) or continuous glucose monitoring (CGM).
- HbA1c target: <7.0%.
⭐ Dawn Phenomenon: Early morning hyperglycemia (around 3 AM to 8 AM) due to nocturnal growth hormone spikes. Manage by increasing the evening basal insulin dose or adjusting pump settings.
Complications - The Highs and Lows
-
Hyperglycemia: Diabetic Ketoacidosis (DKA)
- Triad: Glucose >250 mg/dL, pH <7.3 (anion gap metabolic acidosis), + ketones.
- Triggers: Infection, ischemia, infarction, insulin deficit.
- Symptoms: Fruity breath, Kussmaul respirations, abdominal pain.
-
Hypoglycemia
- Blood glucose <70 mg/dL.
- Autonomic symptoms (sweating, tremor) precede neuroglycopenic (confusion, coma).
- Treatment: Conscious → 15g carbs. Unconscious → IV Dextrose or IM Glucagon.
⭐ Dawn Phenomenon vs. Somogyi Effect: To differentiate morning hyperglycemia, check glucose at 3 AM. A low reading suggests Somogyi (rebound), while a normal/high reading indicates Dawn (hormonal surge).
High‑Yield Points - ⚡ Biggest Takeaways
- Results from T-cell mediated autoimmune destruction of pancreatic β-cells, causing absolute insulin deficiency.
- Strongest genetic risk is associated with HLA-DR3 and HLA-DR4.
- Classic triad: polyuria, polydipsia, polyphagia, often with abrupt weight loss.
- Diabetic Ketoacidosis (DKA) is a frequent and life-threatening initial presentation.
- Key labs: positive autoantibodies (anti-GAD65, anti-islet cell) and low/absent C-peptide.
- Management requires lifelong insulin therapy; cannot be managed with oral agents.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

