Non-Inferiority Trials - Not Worse, Just Different
- Goal: To show a new treatment is not unacceptably worse than the standard. Used when new options offer other benefits (e.g., ↑safety, ↓cost).
- Non-Inferiority Margin (Δ): The pre-specified, largest clinically acceptable difference to still be considered "good enough."
- Hypotheses:
- H₀ (Null): The new treatment is inferior (Difference > Δ).
- H₁ (Alternative): The new treatment is non-inferior (Difference ≤ Δ).
- Sample Size: Influenced by α, β (power), variance, and Δ. A smaller, stricter margin (↓Δ) requires a ↑ sample size.
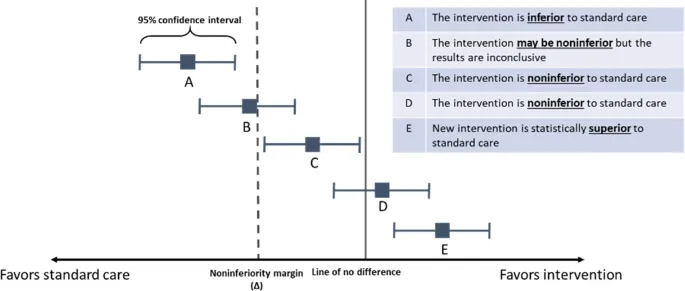
⭐ For non-inferiority to be claimed, the entire confidence interval for the treatment effect difference must be less than the non-inferiority margin (Δ).
NI Sample Size - The Secret Sauce
-
Goal: Prove a new treatment is not unacceptably worse than the standard. The sample size hinges on the non-inferiority margin (δ).
-
Core Formula (per group):
- $n = \frac{(Z_{\alpha} + Z_{\beta})^2 \times (2\sigma^2)}{(\Delta - \delta)^2}$
- $\Delta$: Assumed true difference between treatments.
- $\delta$: The pre-defined non-inferiority margin.
-
Key Relationship: The required sample size is highly sensitive to the gap between the true effect ($\Delta$) and the NI margin ($\delta$).
⭐ Exam Pearl: Counterintuitively, non-inferiority trials often require a larger sample size than superiority trials, especially if the new drug's efficacy is expected to be very similar to the standard (i.e., Δ is small).
The Formula - Cranking the Numbers
- Calculates subjects needed to prove a new treatment is not unacceptably worse than standard treatment.
- Formula for continuous outcomes (per group):
$$ n = \frac{2 \sigma^2 (Z_{\alpha} + Z_{\beta})^2}{(\Delta - \delta)^2} $$
- Key Inputs:
- $Z_{\alpha}$: Significance level (e.g., 1.96 for α=0.025)
- $Z_{\beta}$: Statistical power (e.g., 0.84 for 80% power)
- $\sigma^2$: Data variability (variance)
- $\delta$: The non-inferiority margin (critical value)
- $\Delta$: Expected difference in effect (often assumed to be 0)
- Key Inputs:
- Sample Size Drivers:
- Sample size ↑ as power ↑, significance ↑ (α ↓), or variance ↑.
- Crucially, sample size ↑ dramatically as the margin (δ) ↓ (becomes stricter).
⭐ The non-inferiority margin (δ) is the most critical choice. It must be smaller than the active control's established benefit over placebo, ensuring the new drug preserves a clinically meaningful effect.
Sample Size Levers - Dialing It In
- Non-Inferiority Margin (δ): The most critical lever.
- Smaller (stricter) margin → ↑ sample size.
- Larger (lenient) margin → ↓ sample size.
- Power (1-β):
- Higher power (e.g., 90% vs 80%) → ↑ sample size. Reduces Type II error risk.
- Significance Level (α):
- Lower α (e.g., 0.01) → ↑ sample size. Reduces Type I error risk.
- Outcome Variability (σ²):
- Higher data variability → ↑ sample size for precise estimates.
⭐ The non-inferiority margin (δ) isn't arbitrary. It's set based on historical data of the active control's effect over a placebo, ensuring the new drug preserves a clinically meaningful effect.
High‑Yield Points - ⚡ Biggest Takeaways
- The goal is to show a new treatment is not unacceptably worse than the standard one.
- A pre-specified non-inferiority margin (δ) sets the boundary of acceptable difference.
- Success requires the entire confidence interval of the effect to be above -δ.
- Sample size is driven by the margin (δ), power (1-β), and significance (α).
- A smaller (stricter) margin demands a larger sample size to achieve adequate power.
- If the CI crosses -δ, the result is inconclusive, not a confirmation of inferiority.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

