PTM Basics - Protein Finishing School
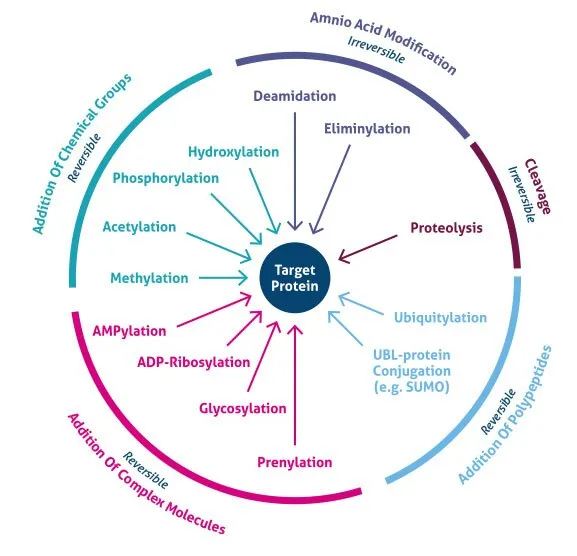
- What? Covalent modification of proteins after translation, diversifying function & regulating activity.
- Where? Endoplasmic Reticulum, Golgi apparatus, cytoplasm.
- Key Modifications:
- Phosphorylation: On Ser, Thr, Tyr residues via kinases. Reversible.
- Glycosylation: N-linked (Asn, in RER) & O-linked (Ser/Thr, in Golgi).
- Ubiquitination: Targets proteins for proteasomal degradation.
- Acetylation/Methylation: Modifies histones, regulating gene access.
- Hydroxylation: Proline/Lysine in collagen; requires Vitamin C.
⭐ I-cell disease results from failed mannose-6-phosphate tagging of lysosomal proteins, causing them to be secreted. Features include coarse facies & skeletal defects.
Key Modifications - Molecular Tagging
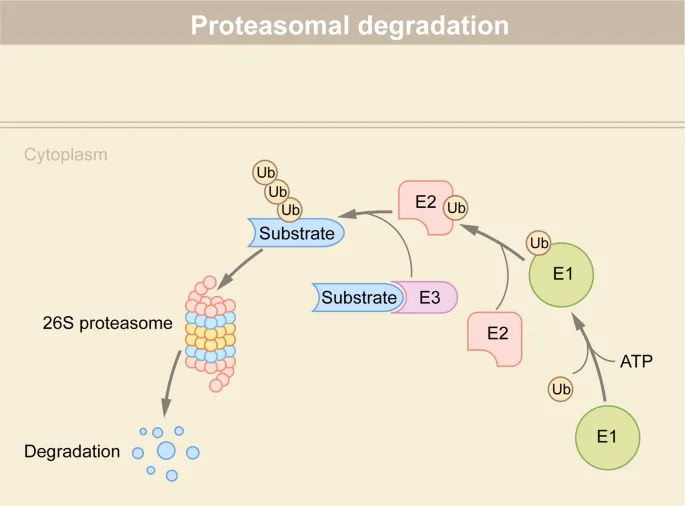
- Ubiquitination: Covalent attachment of ubiquitin (a 76-amino acid polypeptide) to target proteins, primarily marking them for degradation.
- Function: Acts as a molecular "tag" for destruction by the proteasome. Also involved in DNA repair and signal transduction.
- Mechanism: A three-enzyme cascade:
- E1 (Activating Enzyme): Activates ubiquitin via ATP.
- E2 (Conjugating Enzyme): Carries the activated ubiquitin.
- E3 (Ligase): Recognizes the specific protein substrate and transfers ubiquitin to it. Determines substrate specificity.
⭐ Proteasome inhibitors (e.g., Bortezomib, Carfilzomib) are key drugs for multiple myeloma. By blocking the proteasome, they cause an accumulation of toxic, misfolded proteins within malignant plasma cells, triggering apoptosis.
- SUMOylation: Attachment of a Small Ubiquitin-like Modifier (SUMO).
- Function: Alters protein function, localization, and protein-protein interactions. It does not tag for degradation.
Clinical Focus - When PTMs Go Wrong
-
Defective PTMs disrupt protein structure, function, and localization, leading to numerous diseases.
-
Glycosylation Errors
- I-Cell Disease (Mucolipidosis II): Failure of Golgi to add mannose-6-phosphate tag to lysosomal enzymes. Proteins are secreted extracellularly instead of trafficked to the lysosome.
- Presents with coarse facial features, skeletal abnormalities, clouded corneas.
- I-Cell Disease (Mucolipidosis II): Failure of Golgi to add mannose-6-phosphate tag to lysosomal enzymes. Proteins are secreted extracellularly instead of trafficked to the lysosome.
-
Hydroxylation Defects
- Scurvy: Vitamin C (ascorbate) deficiency impairs prolyl & lysyl hydroxylases, leading to defective collagen synthesis.
- Ehlers-Danlos Syndrome (some types): Lysyl hydroxylase deficiency causes unstable collagen, leading to joint hypermobility.
-
Abnormal Phosphorylation
- Alzheimer's Disease: Hyperphosphorylation of tau protein leads to neurofibrillary tangles and neuronal death.
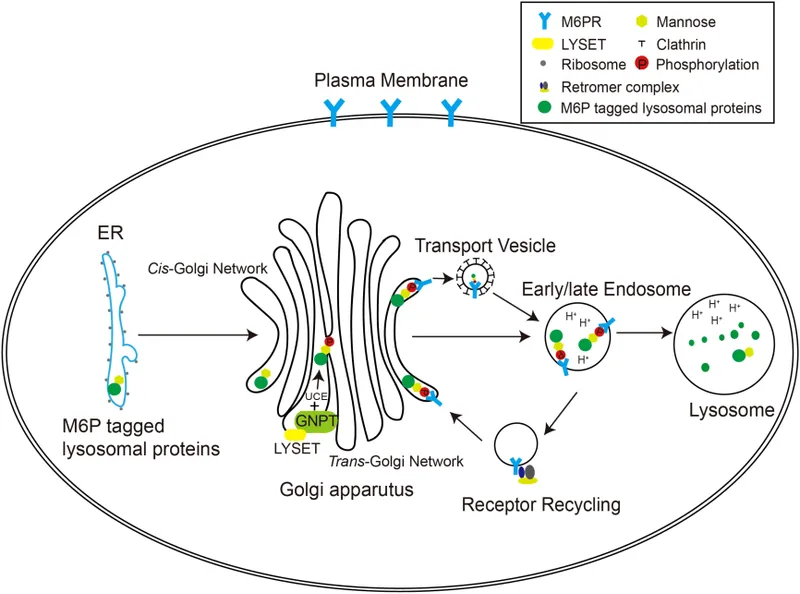
⭐ In I-cell disease, the diagnostic hallmark is high plasma levels of lysosomal enzymes, which are secreted from the cell due to the failed mannose-6-phosphate targeting signal.
High‑Yield Points - ⚡ Biggest Takeaways
- Trimming of propeptides is crucial for activating zymogens (e.g., trypsinogen).
- Phosphorylation by kinases and dephosphorylation by phosphatases are key regulatory switches.
- Glycosylation (N-linked in the ER, O-linked in the Golgi) is vital for protein targeting and stability.
- Hydroxylation of proline and lysine in collagen synthesis requires vitamin C.
- Ubiquitination tags proteins for degradation by the proteasome.
- Disulfide bonds between cysteine residues stabilize extracellular proteins like insulin.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

