Fed State & Lipogenesis - Glucose-to-Fat Pipeline
- Driving Force: High insulin:glucagon ratio after a meal.
- Glycolysis: Glucose → Pyruvate. Also produces Dihydroxyacetone phosphate (DHAP) → Glycerol-3-Phosphate (the triglyceride backbone).
- Citrate Shuttle: Primary mechanism to move Acetyl-CoA from mitochondria to the cytosol for synthesis.
- High ATP/NADH levels inhibit isocitrate dehydrogenase, causing citrate to accumulate and exit the mitochondria.
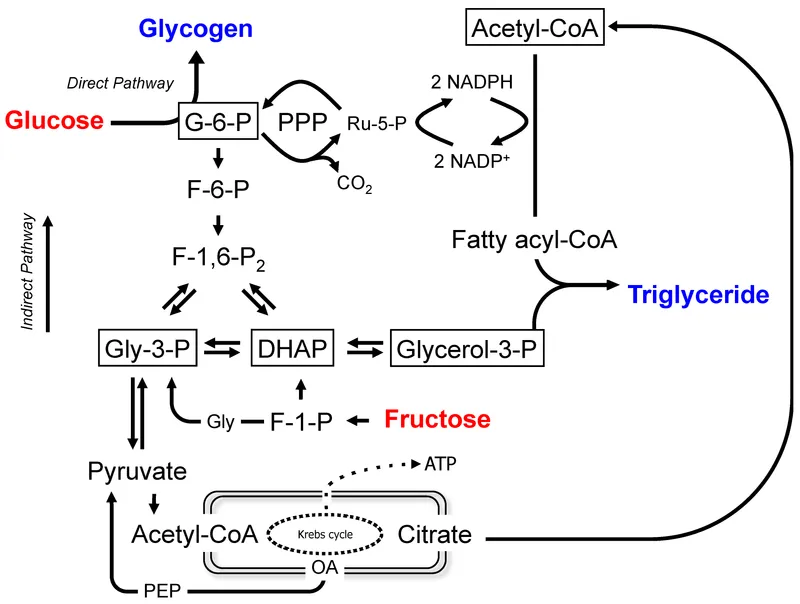
- Key Regulatory Steps:
- Acetyl-CoA Carboxylase (ACC): Irreversible, rate-limiting enzyme. Converts Acetyl-CoA → Malonyl-CoA.
- Activation: Insulin, Citrate.
- Inhibition: Glucagon, Palmitoyl-CoA.
- Fatty Acid Synthase: Synthesizes palmitate from Malonyl-CoA, requires NADPH from HMP Shunt.
- Acetyl-CoA Carboxylase (ACC): Irreversible, rate-limiting enzyme. Converts Acetyl-CoA → Malonyl-CoA.
⭐ Malonyl-CoA not only serves as a building block for fatty acids but also inhibits carnitine acyltransferase I (CAT-I), preventing newly synthesized fatty acids from entering the mitochondria for β-oxidation.
Fasting State & Ketogenesis - Fat-to-Fuel Switch
- Hormonal Trigger: ↓ Insulin and ↑ Glucagon activate Hormone-Sensitive Lipase (HSL) in adipose tissue, initiating lipolysis.
- Fatty Acid Influx: Free Fatty Acids (FFAs) are released from adipose tissue, travel bound to albumin, and are taken up by the liver.
- Hepatic β-Oxidation: In liver mitochondria, FFAs are oxidized into a large volume of Acetyl-CoA, which inhibits the Pyruvate Dehydrogenase Complex.
- Ketogenesis: Excess Acetyl-CoA is shunted into the ketogenesis pathway.
- Rate-Limiting Enzyme: Mitochondrial HMG-CoA synthase.
- Products: Acetoacetate and β-hydroxybutyrate (transportable energy) and acetone (exhaled).
⭐ The liver produces ketone bodies but cannot use them as fuel because it lacks the enzyme thiophorase (succinyl-CoA acetoacetate-CoA transferase).

Regulatory Crossroads - The On/Off Switches
- Acetyl-CoA Carboxylase (ACC): Rate-limiting enzyme for fatty acid synthesis.
- Activators: Insulin, Citrate. 📌 Insulin & Citrate Activate Carboxylase.
- Inhibitors: Glucagon, Palmitoyl-CoA (long-chain fatty acyl-CoA).
- Carnitine Palmitoyltransferase I (CPT-1): Rate-limiting for β-oxidation.
- Inhibitor: Malonyl-CoA (the product of ACC).
- Reciprocal Regulation: High insulin (well-fed state) → ↑ACC activity → ↑Malonyl-CoA → ↓CPT-1 activity, preventing futile cycling.
⭐ Malonyl-CoA is the pivotal molecule ensuring that fatty acid synthesis and oxidation do not occur simultaneously.
High‑Yield Points - ⚡ Biggest Takeaways
- Citrate shuttles acetyl-CoA from mitochondria for FA synthesis; it also inhibits glycolysis via PFK-1 inhibition.
- A high insulin/glucagon ratio (fed state) activates acetyl-CoA carboxylase (ACC), promoting lipid storage.
- Malonyl-CoA (from ACC) inhibits CAT-I, blocking newly synthesized FAs from mitochondrial entry and oxidation.
- The glycerol-3-P backbone for TG synthesis is derived from glycolytic intermediate DHAP.
- In fasting, hepatic β-oxidation drives ketone body formation from excess acetyl-CoA.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

