Glycogen Structure - The Body's Sugar Stash
- Function: Main storage form of glucose in animals, found primarily in liver and skeletal muscle.
- Structure: Large, branched polymer of glucose residues.
- Linear chains: Glucose units linked by $α-1,4$ glycosidic bonds.
- Branch points: Occur every 8-12 residues, created by $α-1,6$ glycosidic bonds.

- Advantage: Branching creates many non-reducing ends, allowing for rapid synthesis and degradation to maintain glucose homeostasis.
⭐ Glycogenolysis and synthesis occur at the non-reducing ends, allowing for rapid glucose release from multiple points simultaneously.
Glycogenesis - Building the Branchies
- Activation: Glucose is converted to Glucose-1-Phosphate (G1P) and then activated by UTP to form UDP-Glucose.
- Enzyme: UDP-glucose pyrophosphorylase.
- Elongation (Rate-Limiting): Glycogen synthase adds UDP-glucose to the non-reducing end of a glycogen primer, forming linear $α-1,4$ glycosidic bonds.
- Branching: Branching enzyme (amylo-$α-1,4$ → $α-1,6$-transglucosidase) transfers a segment of a linear chain to create an $α-1,6$ branch point.
- This increases glycogen solubility and the number of terminal residues for rapid synthesis or degradation.
⭐ Insulin stimulates glycogenesis by activating protein phosphatase, which dephosphorylates and activates glycogen synthase.
Glycogenolysis - Tapping the Reserve
- Primary Goal: Maintain blood glucose homeostasis (liver) & provide immediate fuel for glycolysis (muscle).
- Key Enzymes:
- Glycogen Phosphorylase: Rate-limiting enzyme. Cleaves α-1,4 glycosidic bonds using inorganic phosphate ($P_i$) to release Glucose-1-Phosphate. Requires Vitamin B6 (PLP) as a cofactor.
- Debranching Enzyme: Possesses two distinct catalytic activities:
- 4-α-D-glucanotransferase: Transfers 3 glucose residues from a limit branch to the non-reducing end of another chain.
- α-1,6-glucosidase: Hydrolyzes the single remaining α-1,6 bond to release free glucose.
- Regulation:
- Activation: Glucagon (liver), Epinephrine (liver & muscle) via ↑cAMP.
- Inhibition: Insulin via protein phosphatase activation.
⭐ Muscle lacks Glucose-6-Phosphatase. Thus, muscle glycogen provides ATP for the muscle itself and cannot directly contribute to blood glucose levels.
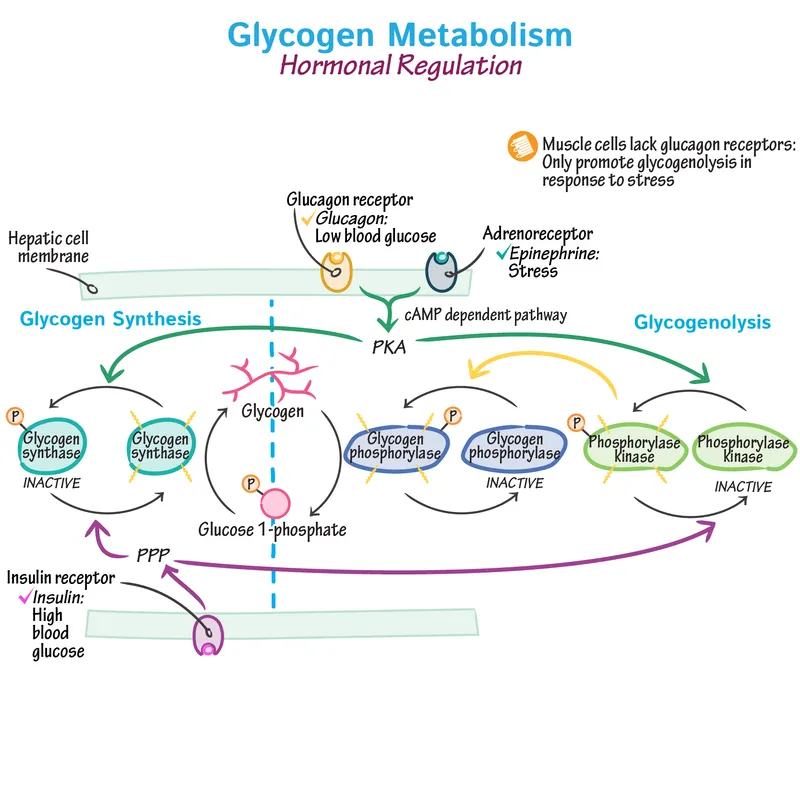
Hormonal Regulation - Metabolic Master Control
- Insulin (Well-fed state): High insulin/glucagon ratio promotes glycogen storage (dephosphorylation).
- Activates protein phosphatase, which dephosphorylates key enzymes.
- ↑ Glycogen Synthase activity (active when dephosphorylated).
- ↓ Glycogen Phosphorylase activity (inactive when dephosphorylated).
- Glucagon & Epinephrine (Fasting/Stress): Low insulin/glucagon ratio promotes glycogenolysis (phosphorylation).
- Activate PKA via cAMP (Glucagon, β-receptors) or Ca²⁺/PKC (α₁-receptors).
- ↓ Glycogen Synthase activity.
- ↑ Glycogen Phosphorylase activity.
⭐ Muscle lacks glucagon receptors; its glycogenolysis responds to epinephrine but not glucagon. Muscle glycogen is for local use, not blood glucose maintenance.
High‑Yield Points - ⚡ Biggest Takeaways
- Glycogen, the primary storage form of glucose, resides mainly in the liver and skeletal muscle.
- Liver glycogen maintains blood glucose homeostasis; muscle glycogen serves as a local energy reserve.
- Structurally, it has α-1,4 glycosidic bonds (linear) and α-1,6 glycosidic bonds (branch points).
- Key enzymes: Glycogen synthase (synthesis) and glycogen phosphorylase (degradation).
- Insulin stimulates synthesis; glucagon and epinephrine stimulate breakdown.
- Branching increases solubility and creates numerous non-reducing ends for rapid glucose mobilization.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

