Spermatogenesis - The Sperm Factory
- Location: Seminiferous tubules of testes.
- Duration: Full cycle takes approx. 74 days.
- Process: Diploid spermatogonia → (Mitosis) → Primary spermatocytes → (Meiosis I) → Haploid secondary spermatocytes → (Meiosis II) → Spermatids → (Spermiogenesis) → Spermatozoa.
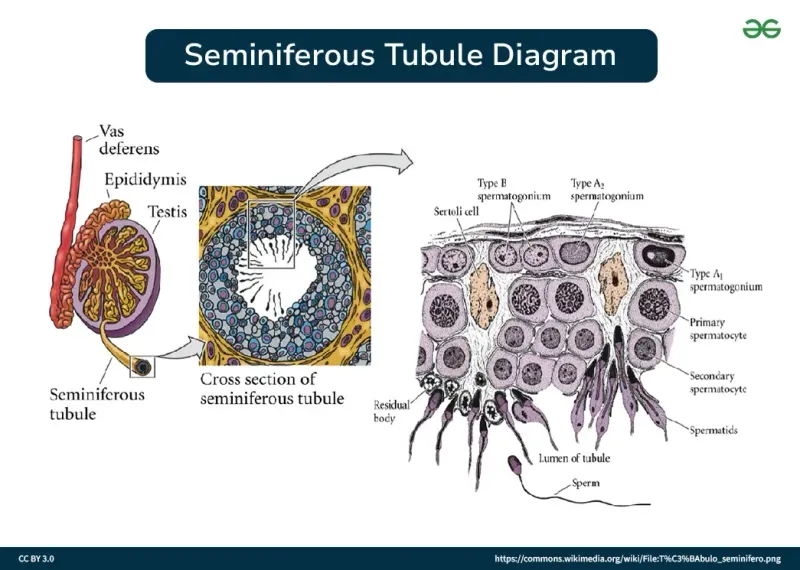
- Key Cells:
- Sertoli cells: Support and nourish developing sperm; form blood-testis barrier. Stimulated by FSH.
- Leydig cells: Secrete testosterone. Stimulated by LH.
⭐ High-Yield Fact: Spermatogenesis requires a temperature 2-3°C below core body temperature, maintained by the pampiniform plexus and scrotum positioning.
The Assembly Line - From Stem Cell to Sperm
-
Location: Seminiferous tubules of the testes, beginning at puberty.
-
Duration: The entire process takes approximately 64 days.
-
Key Players:
- Sertoli cells (nurse cells): Support developing sperm, form the blood-testis barrier, and secrete Inhibin B.
- Leydig cells: Produce testosterone in response to LH.
-
Spermiogenesis: The final stage where spermatids mature into spermatozoa. This involves shedding excess cytoplasm and developing a head (with acrosome) and tail.
⭐ Spermatogenesis is temperature-sensitive and occurs optimally below core body temperature (~34-35°C). Conditions like cryptorchidism or varicoceles that increase testicular temperature can impair sperm production and lead to infertility.
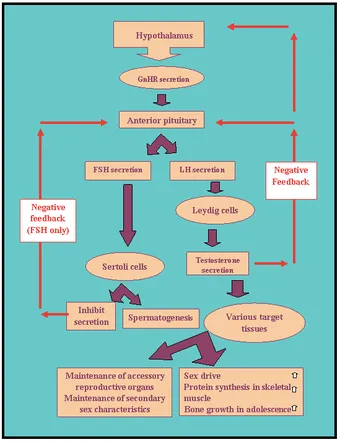
Hormonal Bosses - The Endocrine Control
- HPG Axis (Hypothalamic-Pituitary-Gonadal): The primary driver, initiated by pulsatile GnRH from the hypothalamus.
-
Key Hormones & Cells:
- LH → Leydig Cells: Produce Testosterone.
- FSH → Sertoli Cells: Support spermatogenesis, produce Inhibin B & Androgen-Binding Protein (ABP) which keeps local testosterone levels high.
-
Feedback Loops:
- Testosterone negatively feeds back on the hypothalamus (↓ GnRH) and pituitary (↓ LH).
- Inhibin B selectively inhibits FSH secretion from the pituitary.
⭐ Continuous (non-pulsatile) GnRH administration shuts down the axis by downregulating pituitary receptors, a principle used in treating prostate cancer.
The Final Product - Sperm Anatomy & Journey
- Anatomy: A mature spermatozoon consists of:
- Head: Contains the haploid nucleus (23,X or 23,Y). Capped by the acrosome, which holds enzymes (e.g., hyaluronidase, acrosin) to penetrate the ovum.
- Midpiece: Concentrated with mitochondria to produce ATP for motility.
- Tail (Flagellum): Propels the sperm.

- Journey (Ejaculation Pathway):
- 📌 SEVEN UP: Seminiferous tubules → Epididymis → Vas deferens → Ejaculatory duct → (Nothing) → Urethra → Penis.
⭐ Capacitation: The final step of sperm maturation, occurring within the female reproductive tract. It is required for the sperm to become competent for fertilization.
High‑Yield Points - ⚡ Biggest Takeaways
- Spermatogenesis transforms spermatogonia into mature spermatozoa within the seminiferous tubules, taking about 64 days.
- Sertoli cells ("nurse cells") support sperm development, form the blood-testis barrier, and are stimulated by FSH.
- Leydig cells, stimulated by LH, produce testosterone, the primary driver of spermatogenesis.
- Spermiogenesis is the final maturation step where a round spermatid becomes a motile spermatozoon.
- Key products include the acrosome, containing hydrolytic enzymes for fertilization.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

