Cardiology

On this page
🎯 Cardiac Imaging Mastery: Your Diagnostic Vision Arsenal
Master cardiac imaging, and you unlock the blueprint of every structural defect, functional abnormality, and hemodynamic crisis. Each modality reveals distinct anatomical territories and physiological parameters-echocardiography captures real-time valve dynamics, nuclear imaging quantifies perfusion deficits, CT reconstructs coronary anatomy with submillimeter precision, and MRI characterizes tissue composition. Understanding when to deploy each tool transforms diagnostic uncertainty into clinical clarity, enabling you to visualize pathology before it becomes irreversible.
🔊 Echocardiography: The Real-Time Cardiac Theater
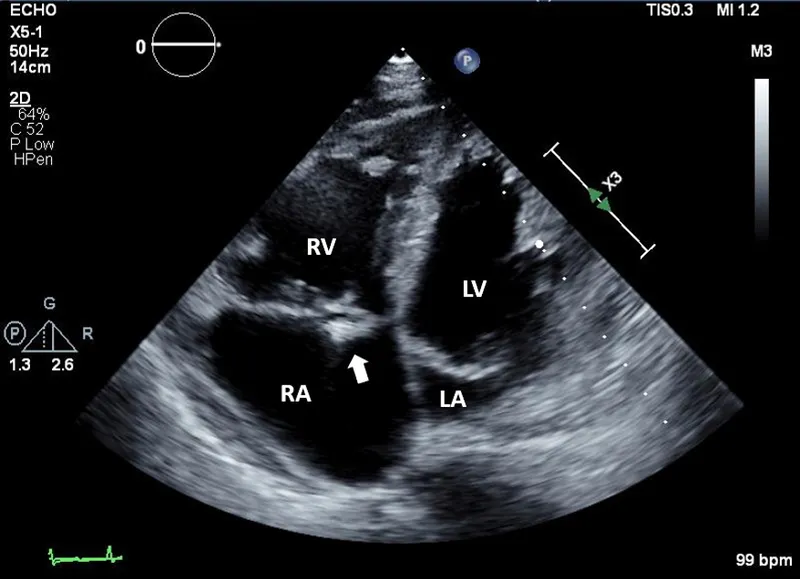
Echocardiography stands as the first-line imaging workhorse, providing real-time visualization of cardiac structure and function without radiation exposure. Transthoracic echocardiography (TTE) achieves diagnostic accuracy exceeding 85% for valvular pathology and 90% for regional wall motion abnormalities, while transesophageal echocardiography (TEE) elevates sensitivity to >95% for posterior structures, vegetations, and atrial appendage thrombi.
Doppler Physics: Velocity Becomes Pressure
Understanding Doppler principles transforms velocity measurements into hemodynamic pressures. The modified Bernoulli equation-Pressure gradient = 4 × (velocity)²-enables non-invasive pressure quantification across stenotic valves. A peak velocity of 4 m/s across the aortic valve translates to a 64 mmHg gradient, defining severe aortic stenosis when valve area falls below 1.0 cm².
- Continuous-wave Doppler: Captures peak velocities along entire beam path
- Essential for severe stenosis (velocities >4 m/s)
- Cannot localize exact depth of signal origin
- Ideal for aortic stenosis, mitral regurgitation severity
- Pulsed-wave Doppler: Samples specific cardiac locations
- Limited by Nyquist limit (aliasing at velocities >1.5-2.0 m/s)
- Precise localization enables diastolic function assessment
- Critical for E/A ratio calculation (normal 1.0-2.0)
- Tissue Doppler Imaging: Measures myocardial velocities
- E' velocity <8 cm/s indicates diastolic dysfunction
- E/E' ratio >14 suggests elevated filling pressures (>15 mmHg)
- Distinguishes constrictive pericarditis from restrictive cardiomyopathy
📌 Remember: PLEA for Doppler types-Pulsed-wave for Localization, E' for diastolic function, Aliasing limits pulsed-wave, Continuous-wave captures All velocities along beam path without depth discrimination.
Strain Imaging: Detecting Subclinical Dysfunction
Global longitudinal strain (GLS) quantifies myocardial deformation, detecting contractile abnormalities before ejection fraction declines. Normal GLS measures -18% to -22% (negative values indicate shortening). GLS values worse than -16% predict adverse outcomes even when ejection fraction remains preserved at ≥50%, identifying subclinical cardiotoxicity in 30-40% of chemotherapy patients before conventional measures decline.
⭐ Clinical Pearl: In suspected cardiac amyloidosis, the "apical sparing" pattern-preserved apical strain with reduced basal and mid-ventricular strain-demonstrates 93% sensitivity and 82% specificity when combined with increased wall thickness (≥12 mm) and low voltage on ECG.

TEE Indications: When Surface Views Fail
Transesophageal echocardiography overcomes TTE limitations through proximity to posterior cardiac structures, eliminating interference from lung, bone, and adipose tissue. TEE becomes essential when TTE image quality proves inadequate or when superior spatial resolution alters management.
| Clinical Scenario | TEE Advantage | Detection Rate TTE | Detection Rate TEE | Critical Threshold |
|---|---|---|---|---|
| Endocarditis vegetations | Proximity to valves | 60-70% | >95% | Vegetations >2 mm |
| Left atrial appendage thrombus | Direct visualization | 40-50% | 95-98% | Before cardioversion |
| Aortic dissection | Ascending aorta imaging | 60-80% | 95-99% | Intimal flap detection |
| Prosthetic valve dysfunction | Eliminates acoustic shadowing | 40-60% | 85-95% | Paravalvular leak >3 mm |
| Mitral regurgitation mechanism | Surgical planning precision | 70-80% | 90-98% | Prolapse vs. restriction |
💡 Master This: TEE transforms endocarditis management-detecting vegetations <5 mm that TTE misses, identifying abscess formation in 85% of perivalvular cases, and guiding surgical timing when vegetation size exceeds 10 mm with embolic risk approaching 50% in the first week.
Stress Echocardiography: Unmasking Ischemia
Stress echocardiography combines exercise or pharmacologic stress with imaging to detect inducible wall motion abnormalities, achieving 85% sensitivity and 80% specificity for obstructive coronary disease. Dobutamine stress echo (DSE) incrementally increases heart rate and contractility using 10-40 mcg/kg/min dosing, targeting 85% of maximum predicted heart rate (220 - age).
- Wall motion scoring system: Quantifies ischemic burden
- 1 = Normal (thickening >30%)
- 2 = Hypokinesis (thickening 10-30%)
- 3 = Akinesis (thickening <10%)
- 4 = Dyskinesis (paradoxical outward motion)
- 5 = Aneurysmal (diastolic deformity)
- Interpretation thresholds: Define ischemia severity
- New wall motion abnormality in ≥2 contiguous segments indicates significant stenosis (>70% luminal narrowing)
- Ischemia at low workload (<5 METs or heart rate <120 bpm) predicts high-risk disease
- Biphasic response (initial augmentation then deterioration) suggests viability in hibernating myocardium
⭐ Clinical Pearl: DSE identifies viable myocardium in 70-80% of akinetic segments showing low-dose augmentation (5-10 mcg/kg/min), predicting functional recovery post-revascularization with 80% positive predictive value when contractile reserve demonstrates improvement of ≥1 wall motion score grade.
Connect these foundational echo principles through nuclear imaging to understand how perfusion deficits correlate with wall motion abnormalities in ischemic heart disease.
🔊 Echocardiography: The Real-Time Cardiac Theater
☢️ Nuclear Cardiology: The Perfusion Detectives
Nuclear imaging quantifies myocardial perfusion and viability through radiotracer distribution, detecting ischemia with 85-90% sensitivity and 70-75% specificity for obstructive coronary disease. Single-photon emission computed tomography (SPECT) and positron emission tomography (PET) reveal perfusion-metabolism mismatches that distinguish viable from scarred myocardium, guiding revascularization decisions when echocardiography remains equivocal.
SPECT Perfusion Imaging: Stress-Rest Comparison
SPECT myocardial perfusion imaging (MPI) employs technetium-99m-based radiotracers-sestamibi or tetrofosmine-that distribute proportionally to coronary blood flow. The two-day stress-rest protocol administers 25-30 mCi at peak stress and 25-30 mCi at rest, with imaging performed 30-60 minutes post-injection to allow hepatic clearance.
- Perfusion defect interpretation: Distinguishes ischemia from infarction
- Reversible defect: Stress-induced hypoperfusion normalizing at rest indicates ischemia requiring revascularization
- Fixed defect: Persistent hypoperfusion suggests transmural scar, though 10-30% represent hibernating myocardium
- Partially reversible: Mixed ischemia and scar predicts intermediate benefit from revascularization
- Reverse redistribution: Paradoxical worsening at rest suggests non-transmural infarction with peri-infarct ischemia
- Quantitative parameters: Define high-risk anatomy
- Summed stress score >13 indicates severe ischemia (ischemic burden >10% of left ventricle)
- Transient ischemic dilation ratio >1.2 suggests severe three-vessel or left main disease
- Post-stress ejection fraction drop >5% indicates stress-induced dysfunction
- Increased lung uptake (heart-to-lung ratio >0.5) reflects elevated filling pressures
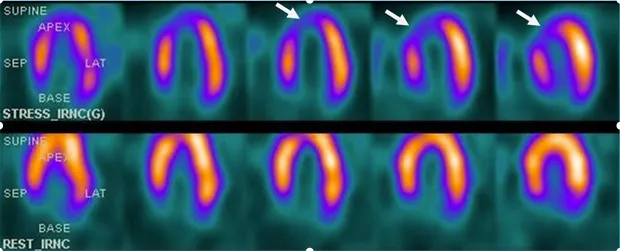
📌 Remember: RIPS for SPECT defect patterns-Reversible means ischemia, Irreversible suggests scar, Partial reversal shows mixed pathology, Severe when involving >10% myocardium requiring intervention.
PET Imaging: Superior Resolution and Viability
PET imaging delivers superior spatial resolution (4-5 mm vs. 10-15 mm for SPECT) and quantitative blood flow measurement, detecting coronary disease with 90-95% sensitivity. Rubidium-82 perfusion imaging combined with F-18 fluorodeoxyglucose (FDG) metabolism imaging identifies viable myocardium through perfusion-metabolism mismatch patterns.
| Perfusion | Metabolism | Interpretation | Viability | Revascularization Benefit |
|---|---|---|---|---|
| Normal | Normal | Normal myocardium | 100% viable | No benefit |
| Reduced | Normal/Increased | Hibernating myocardium | 80-90% viable | High benefit |
| Reduced | Reduced (matched) | Transmural scar | <50% viable | Low benefit |
| Normal | Reduced | Stunning (post-ischemic) | 90-100% viable | Moderate benefit |
| Reduced | Absent | Complete infarction | <10% viable | No benefit |
⭐ Clinical Pearl: PET viability imaging predicts functional recovery with 85% accuracy when perfusion-metabolism mismatch involves ≥10% of left ventricular mass, guiding revascularization in patients with severe LV dysfunction (ejection fraction <35%) where hibernating myocardium comprises 25-40% of dysfunctional segments.
Radiotracers: Matching Agent to Question
Different radiotracers provide complementary physiological information based on their cellular uptake mechanisms and kinetics. Understanding tracer properties enables selection of optimal imaging protocols for specific clinical scenarios.
- Technetium-99m agents (Sestamibi, Tetrofosmine)
- Half-life 6 hours enables same-day or two-day protocols
- Myocardial extraction fraction 60-70% (flow-dependent)
- Hepatic uptake requires 30-60 minute delay before imaging
- Gated acquisition provides simultaneous function assessment (ejection fraction ±5% accuracy)
- Thallium-201: The redistribution specialist
- Potassium analog with 85% first-pass extraction
- Redistribution begins 10-15 minutes post-injection, complete by 4 hours
- Late imaging (24 hours) distinguishes hibernation from scar in 70% of fixed defects
- Higher radiation dose (3-4 times Tc-99m) limits use
- Rubidium-82: The PET perfusion tracer
- Ultra-short half-life (75 seconds) enables rapid stress-rest protocols (<30 minutes total)
- Generator-produced (no cyclotron required)
- Quantitative flow measurement (mL/min/g) detects balanced ischemia
- Coronary flow reserve <2.0 indicates microvascular dysfunction
💡 Master This: When SPECT shows fixed defects but viability remains uncertain, PET FDG imaging resolves the question-perfusion-metabolism mismatch in ≥5 segments predicts 80% probability of functional improvement post-revascularization, while matched perfusion-metabolism defects indicate scar with <20% recovery likelihood.
High-Risk Features: Nuclear Red Flags
Specific scintigraphic patterns identify patients requiring aggressive management regardless of symptom severity. These high-risk markers predict annual cardiac event rates exceeding 5%, warranting coronary angiography and consideration for revascularization.
- Extensive ischemia: Summed difference score >10 (ischemic burden >10% LV)
- Transient ischemic dilation: LV cavity appears larger during stress than rest (ratio >1.2)
- Stress-induced LV dysfunction: Ejection fraction drops ≥10% from rest to stress
- Increased lung uptake: Thallium-201 lung-to-heart ratio >0.5 indicates pulmonary congestion
- Right ventricular visualization: RV uptake suggests RV ischemia or pressure overload
- Multiple vascular territory defects: Involvement of ≥2 coronary distributions
⭐ Clinical Pearl: Transient ischemic dilation demonstrates 85% specificity for severe three-vessel or left main disease, elevating annual cardiac event risk to 8-12% even when ischemic burden appears moderate (5-10% of myocardium), mandating invasive evaluation regardless of stress test functional capacity.
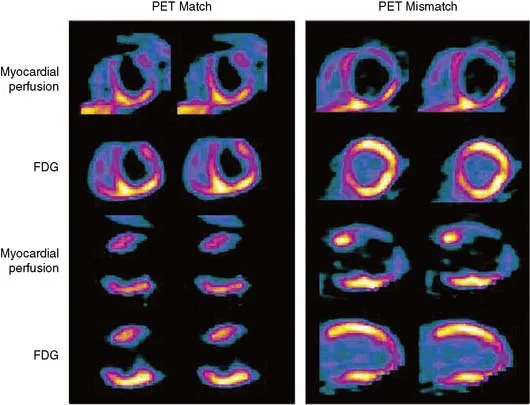
These nuclear perfusion patterns integrate with anatomical coronary imaging through CT angiography to correlate functional ischemia with anatomical stenosis severity.
☢️ Nuclear Cardiology: The Perfusion Detectives
🔬 Cardiac CT: The Anatomical Cartographer
Cardiac computed tomography delivers submillimeter spatial resolution (0.5-0.625 mm) for coronary anatomy visualization, achieving 95-99% negative predictive value for excluding obstructive disease. Modern scanners complete acquisition in <5 heartbeats using <5 mSv radiation, making CT coronary angiography (CTCA) the preferred non-invasive test for intermediate-risk patients with 10-90% pre-test probability of coronary disease.
Coronary CT Angiography: Non-Invasive Luminography
CTCA visualizes coronary lumen and wall, detecting stenosis, plaque composition, and anatomical variants with diagnostic accuracy rivaling invasive angiography. The technique requires heart rate control (<65 bpm optimal) using beta-blockers and sublingual nitroglycerin (0.4-0.8 mg) for coronary vasodilation, with iodinated contrast administration (60-100 mL at 5-6 mL/s) timed to peak arterial enhancement.
- Stenosis grading: Quantifies luminal narrowing
- Minimal: <25% diameter stenosis (no hemodynamic significance)
- Mild: 25-49% stenosis (rarely flow-limiting)
- Moderate: 50-69% stenosis (may cause ischemia with high demand)
- Severe: ≥70% stenosis (usually flow-limiting at rest or stress)
- Total occlusion: 100% (chronic occlusions show collaterals)
- Plaque characterization: Predicts rupture risk
- Calcified plaque: High attenuation (>130 HU), stable morphology
- Non-calcified plaque: Low attenuation (<30 HU), lipid-rich vulnerable plaques
- Mixed plaque: Combination pattern with 30-130 HU components
- Positive remodeling: Outward vessel expansion (remodeling index >1.1) indicates high-risk plaque
- Napkin-ring sign: Low-attenuation core with peripheral rim enhancement predicts acute coronary syndrome

📌 Remember: PLANS for CTCA plaque types-Positive remodeling high-risk, Low attenuation lipid-rich, Attenuation <30 HU vulnerable, Napkin-ring sign predicts rupture, Stable calcified plaques >130 HU.
Coronary Calcium Scoring: Risk Stratification Without Contrast
Non-contrast CT quantifies coronary artery calcium (CAC) using the Agatston score, providing powerful risk stratification independent of traditional risk factors. CAC scoring reclassifies 25-30% of intermediate-risk patients, guiding statin therapy initiation when clinical risk calculators prove equivocal.
| CAC Score | Risk Category | 10-Year Event Rate | Annual Event Rate | Management Implication |
|---|---|---|---|---|
| 0 | Very low | <1% | <0.1% | No statin unless FH |
| 1-99 | Low | 1-5% | 0.5-1.0% | Consider statin if >75th percentile |
| 100-399 | Moderate | 5-10% | 1.5-2.5% | Statin indicated |
| 400-999 | High | 10-20% | 3-5% | High-intensity statin + aspirin |
| ≥1000 | Very high | >20% | >5% | Maximum therapy + imaging |
⭐ Clinical Pearl: A CAC score of zero confers <1% risk of major adverse cardiac events over 5 years in asymptomatic patients, effectively ruling out obstructive disease with 95-99% negative predictive value and potentially deferring statin therapy in low-to-intermediate risk patients without diabetes or family history.
FFR-CT: Functional Assessment from Anatomy
Fractional flow reserve derived from CT (FFR-CT) applies computational fluid dynamics to CTCA datasets, estimating lesion-specific ischemia without additional imaging or stress testing. FFR-CT values ≤0.80 indicate hemodynamically significant stenosis, achieving 85% sensitivity and 80% specificity compared to invasive FFR measurement.
💡 Master This: FFR-CT transforms CTCA from pure anatomy to functional assessment, reducing unnecessary catheterization by 60-70% in patients with moderate stenosis (50-70%) where anatomical severity alone proves indeterminate, identifying the 40-50% of intermediate stenoses that cause ischemia requiring revascularization.
Structural and Functional CT Applications
Beyond coronary imaging, cardiac CT excels at evaluating cardiac masses, pericardial disease, and congenital anomalies with spatial resolution surpassing echocardiography and MRI for specific applications.
- Cardiac masses: Differentiates thrombus from tumor
- Thrombus: No enhancement post-contrast (<20 HU increase)
- Myxoma: Heterogeneous enhancement (40-80 HU increase)
- Lipoma: Fat attenuation (-50 to -100 HU)
- Metastases: Avid enhancement with pericardial involvement
- Pericardial thickness: Quantifies constriction
- Normal pericardium <2 mm thick
- Constrictive pericarditis shows thickening >4 mm in 80%
- Calcification present in 30-50% of constrictive cases
- Tube-shaped ventricles with biatrial enlargement
- Pulmonary vein anatomy: Pre-ablation mapping
- Identifies anatomical variants in 40% (common ostia, accessory veins)
- Measures ostial diameters (15-20 mm typical)
- Detects post-ablation stenosis >50% requiring intervention
⭐ Clinical Pearl: Pre-procedural CT for transcatheter aortic valve replacement (TAVR) proves essential-measuring aortic annulus dimensions (20-29 mm typical), assessing coronary height above annulus (≥10 mm safe), and identifying vascular access routes, reducing paravalvular leak incidence from 15-20% to <5% with CT-guided sizing.
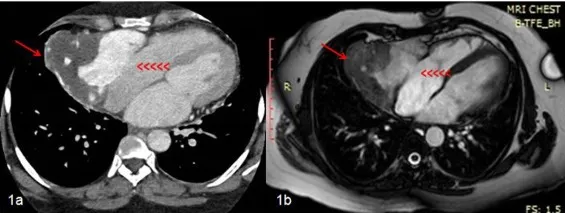
These CT anatomical insights complement tissue characterization capabilities through cardiac MRI to provide comprehensive structural and functional assessment.
🔬 Cardiac CT: The Anatomical Cartographer
🧲 Cardiac MRI: The Tissue Characterization Virtuoso
Cardiac magnetic resonance imaging (CMR) delivers unparalleled soft tissue contrast and functional assessment without ionizing radiation, achieving gold-standard accuracy for ventricular volumes (±5 mL), ejection fraction (±3%), and mass quantification (±10 g). Late gadolinium enhancement (LGE) detects myocardial scar with 95% sensitivity, distinguishing ischemic from non-ischemic cardiomyopathy patterns and guiding implantable cardioverter-defibrillator (ICD) placement when scar burden predicts arrhythmic risk.
Cine Imaging: The Motion Picture
Steady-state free precession (SSFP) cine sequences capture cardiac motion throughout the cycle with temporal resolution 30-50 ms, enabling precise volumetric quantification and wall motion assessment. Standard short-axis stack coverage from base to apex using 8-12 slices with 8 mm thickness provides comprehensive evaluation.
- Volumetric quantification: Gold-standard measurements
- End-diastolic volume (EDV): Normal 130-160 mL (men), 100-120 mL (women)
- End-systolic volume (ESV): Normal 40-60 mL (men), 30-45 mL (women)
- Stroke volume (SV = EDV - ESV): Normal 70-90 mL
- Ejection fraction (EF = SV/EDV × 100): Normal ≥55%
- Cardiac output (CO = SV × HR): Normal 4-8 L/min
- Regional wall motion: Seventeen-segment model
- Basal segments (1-6): Anterior, anteroseptal, inferoseptal, inferior, inferolateral, anterolateral
- Mid segments (7-12): Same distribution as basal
- Apical segments (13-16): Anterior, septal, inferior, lateral
- True apex (17): Apical cap
- Wall thickening >40% systolic indicates normal function

📌 Remember: SAVED for CMR volume measurements-Stroke volume EDV minus ESV, Accurate within ±5 mL, Ventricular function gold standard, Ejection fraction ≥55% normal, Dilated when EDV exceeds 200 mL.
Late Gadolinium Enhancement: The Scar Map
LGE imaging performed 10-20 minutes post-gadolinium administration (0.1-0.2 mmol/kg) identifies myocardial scar through delayed washout from expanded extracellular space. The transmural extent of LGE predicts functional recovery post-revascularization-segments with <50% transmural enhancement demonstrate 80% recovery probability, while >75% transmural scar shows <10% recovery likelihood.
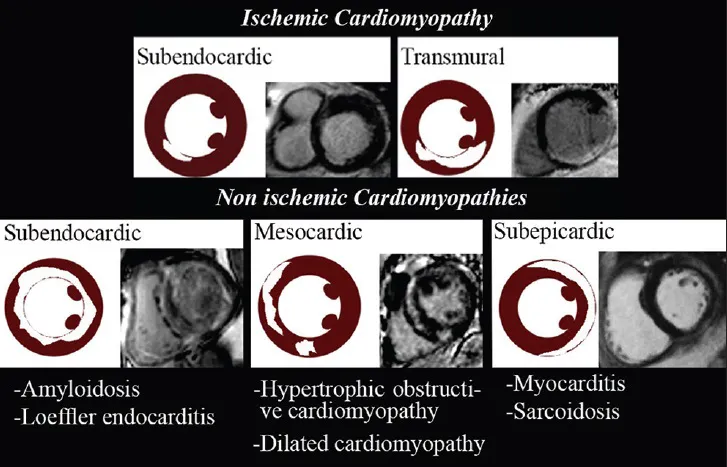
| LGE Pattern | Distribution | Diagnosis | Transmurality | Clinical Significance |
|---|---|---|---|---|
| Subendocardial | Coronary territory | Ischemic scar | 25-50% wall | Partial recovery possible |
| Transmural | Coronary territory | Ischemic scar | >75% wall | No recovery expected |
| Mid-wall | Septum, free wall | Non-ischemic CMP | 25-75% wall | Arrhythmia risk marker |
| Subepicardial | Circumferential | Myocarditis | <25% wall | Usually recovers |
| Global diffuse | Entire myocardium | Amyloidosis | Interstitial | Poor prognosis |
⭐ Clinical Pearl: Mid-wall fibrosis detected by LGE in dilated cardiomyopathy increases sudden cardiac death risk 5-8 fold, identifying 40-50% of patients with ejection fraction 30-35% who benefit from primary prevention ICD despite falling above traditional ≤35% cutoff for implantation guidelines.
T1 and T2 Mapping: Quantitative Tissue Signatures
Parametric mapping techniques measure tissue T1 and T2 relaxation times, detecting diffuse myocardial processes that escape visual detection on conventional sequences. Native T1 mapping identifies edema and infiltration, while post-contrast T1 enables extracellular volume (ECV) calculation-a quantitative fibrosis biomarker.
- Native T1 values (1.5T field strength)
- Normal myocardium: 950-1050 ms
- Acute myocarditis: 1100-1200 ms (↑ edema and inflammation)
- Amyloidosis: 1050-1150 ms (↑ protein deposition)
- Iron overload: <850 ms (↓ from paramagnetic iron)
- Anderson-Fabry disease: <900 ms (↓ from sphingolipid accumulation)
- Extracellular volume fraction: Quantifies diffuse fibrosis
- Normal ECV: 25-30%
- Mild fibrosis: 30-35% (early heart failure)
- Moderate fibrosis: 35-40% (symptomatic cardiomyopathy)
- Severe fibrosis: >40% (advanced disease, poor prognosis)
- Amyloidosis: 40-60% (massive expansion)
💡 Master This: ECV measurement transforms CMR from qualitative to quantitative fibrosis assessment, detecting diffuse interstitial expansion in 80-90% of heart failure patients with preserved ejection fraction (HFpEF) who show normal LGE, predicting hospitalization risk when ECV exceeds 30% even with normal ejection fraction.
Stress Perfusion: Ischemia Without Radiation
CMR stress perfusion imaging using vasodilator stress (adenosine 140 mcg/kg/min or regadenoson 0.4 mg bolus) combined with gadolinium first-pass imaging detects subendocardial ischemia with 90% sensitivity and 80% specificity. Perfusion defects appear as dark subendocardial regions during peak hyperemia that normalize on rest imaging.
⭐ Clinical Pearl: CMR stress perfusion combined with LGE provides comprehensive ischemia and viability assessment in a single examination-detecting ischemia in 85-90% of patients with obstructive disease while simultaneously quantifying scar burden to predict revascularization benefit, reducing need for multiple imaging tests by 60-70%.
Advanced Applications: Beyond Standard Protocols
CMR uniquely characterizes complex pathology through specialized sequences targeting specific tissue properties, enabling definitive diagnosis when other modalities remain inconclusive.
- T2-weighted imaging: Detects myocardial edema
- Acute myocardial infarction shows edema 2-3 times infarct size
- Myocarditis demonstrates patchy hyperintensity in 80%
- Takotsubo cardiomyopathy shows apical edema without LGE
- Edema resolves over 4-8 weeks following acute injury
- T2 mapping*: Quantifies myocardial iron
- Normal T2*: >20 ms at 1.5T
- Mild iron overload: 15-20 ms
- Moderate overload: 10-15 ms (cardiac dysfunction risk)
- Severe overload: <10 ms (heart failure develops)
- Guides chelation therapy intensity and monitoring
- Phase-contrast imaging: Measures blood flow velocity
- Quantifies valvular regurgitation (regurgitant fraction >40% severe)
- Calculates shunt ratios (Qp:Qs >1.5 significant)
- Assesses aortic stiffness (pulse wave velocity >10 m/s abnormal)
CMR tissue characterization integrates with other modalities through multimodality imaging protocols to provide comprehensive diagnostic assessment in complex cases.
🧲 Cardiac MRI: The Tissue Characterization Virtuoso
🔀 Multimodality Integration: The Imaging Orchestration
Optimal diagnostic accuracy emerges from strategic modality integration rather than isolated test interpretation. Understanding each modality's strengths, limitations, and complementary relationships enables construction of efficient imaging pathways that maximize diagnostic yield while minimizing cost, radiation, and redundancy. Master clinicians select imaging sequences that answer specific clinical questions with >90% diagnostic confidence.
Test Selection Framework: Matching Question to Modality
Clinical scenarios dictate optimal imaging choices based on information requirements, patient factors, and resource availability. The following decision matrix guides efficient test selection for common diagnostic dilemmas.
- Acute chest pain with elevated troponin
- First-line: Echocardiography for wall motion abnormalities (sensitivity 85%)
- If echo non-diagnostic: Cardiac MRI with LGE (sensitivity 95%, detects 2-3 g scar)
- Alternative: CT coronary angiography if MRI contraindicated
- Avoid: Nuclear imaging acutely (redistribution patterns unreliable)
- Suspected coronary disease, intermediate risk
- First-line: CT coronary angiography (NPV 95-99%)
- If stenosis 50-70%: FFR-CT or stress imaging for functional significance
- Alternative: Stress echocardiography if contrast allergy
- Reserve: Invasive angiography for high-risk features or positive non-invasive test
- Cardiomyopathy evaluation
- First-line: Echocardiography for structure and function
- Second-line: Cardiac MRI with LGE and T1 mapping for tissue characterization
- If infiltrative disease suspected: Nuclear imaging (PET for cardiac sarcoid, Tc-99m-PYP for amyloid)
- Role of CT: Exclude coronary disease if ischemic etiology uncertain
📌 Remember: MATCH for modality selection-MRI for tissue characterization, Angiography (CT) for anatomy, Transthoracic echo for first-line function, Cath for intervention, Hybrid approaches when single modality insufficient.
Radiation Considerations: ALARA in Practice
Radiation exposure from cardiac imaging accumulates over lifetime, with effective doses varying 100-fold between modalities. Strategic test selection minimizes cumulative exposure while maintaining diagnostic accuracy, particularly important in young patients requiring serial imaging.
| Modality | Effective Dose (mSv) | Equivalent CXRs | Lifetime Cancer Risk | Optimal Use Case |
|---|---|---|---|---|
| Echocardiography | 0 | 0 | None | First-line, serial monitoring |
| Cardiac MRI | 0 | 0 | None | Tissue characterization, young patients |
| SPECT MPI | 10-15 | 500-750 | 1 in 1000-2000 | Functional ischemia assessment |
| PET MPI | 2-4 | 100-200 | 1 in 5000-10000 | Viability, sarcoidosis, infection |
| CT calcium score | 1-3 | 50-150 | 1 in 10000+ | Risk stratification, no contrast |
| CT coronary angiography | 3-8 | 150-400 | 1 in 2000-5000 | Anatomical stenosis evaluation |
| Invasive angiography | 5-15 | 250-750 | 1 in 1000-3000 | Intervention, CT contraindication |
⭐ Clinical Pearl: A 40-year-old woman undergoing serial SPECT imaging every 2 years for 20 years accumulates 100-150 mSv total exposure, increasing lifetime cancer risk by 0.5-1.0%-consider substituting stress echocardiography or cardiac MRI for follow-up studies to eliminate radiation while maintaining 80-85% diagnostic accuracy.
Contraindications and Limitations: Knowing When Not to Image
Every modality carries contraindications and technical limitations that may render studies non-diagnostic or unsafe. Anticipating these barriers enables appropriate alternative selection before scheduling suboptimal examinations.
- Echocardiography limitations
- Poor acoustic windows in 15-20% (obesity, COPD, chest wall deformity)
- Prosthetic valve shadowing obscures evaluation
- Operator-dependent quality variation
- Limited tissue characterization capability
- Nuclear imaging contraindications
- Pregnancy (relative contraindication, risk-benefit assessment required)
- Recent myocardial infarction <48 hours (redistribution patterns unreliable)
- Severe bronchospasm (adenosine/regadenoson contraindicated)
- Cumulative radiation concerns with serial studies
- Cardiac CT limitations
- Heart rate >75 bpm degrades image quality
- Severe calcification obscures lumen assessment
- Contrast allergy or renal dysfunction (eGFR <30 mL/min)
- Radiation exposure in young patients
- Arrhythmias cause motion artifact
- Cardiac MRI contraindications
- Non-MRI-conditional devices (older pacemakers, ICDs, metallic fragments)
- Severe claustrophobia (5-10% of patients)
- eGFR <30 mL/min (gadolinium nephrogenic systemic fibrosis risk)
- Inability to breath-hold 10-15 seconds
💡 Master This: When transthoracic echo proves non-diagnostic due to poor windows, proceed directly to cardiac MRI rather than TEE if no acute intervention planned-MRI provides superior functional quantification, tissue characterization, and ischemia assessment in a single comprehensive examination without sedation or esophageal intubation risks.
Cost-Effectiveness: Value-Based Imaging
Healthcare resource stewardship requires balancing diagnostic accuracy against examination costs, considering downstream testing and management implications. The most expensive test proves cost-effective when it definitively answers the clinical question and guides appropriate therapy.
- Cost hierarchy (approximate Medicare reimbursement)
- Echocardiography: $200-400
- CT calcium score: $75-150
- Stress echocardiography: $400-600
- SPECT MPI: $1000-1500
- CT coronary angiography: $500-800
- Cardiac MRI: $1000-1500
- PET MPI: $2000-3000
- Invasive angiography: $2500-5000
- Value considerations
- High NPV tests (CT, MRI) avoid downstream catheterization in 60-70%
- Comprehensive single examinations reduce need for multiple studies
- Radiation-free modalities enable serial monitoring without cumulative risk
- Tissue characterization by MRI guides specific therapies in 40-50% of cardiomyopathy
⭐ Clinical Pearl: In intermediate-risk chest pain patients, CT coronary angiography proves most cost-effective despite higher upfront cost-95-99% negative predictive value safely discharges 50-60% of emergency department patients without further testing, reducing total episode costs by 30-40% compared to stress testing strategies requiring additional imaging.

These multimodality integration principles synthesize into practical clinical algorithms through evidence-based imaging pathways that optimize diagnostic accuracy while minimizing resource utilization.
🔀 Multimodality Integration: The Imaging Orchestration
🎯 Imaging Mastery Arsenal: Your Diagnostic Command Center
Strategic cardiac imaging mastery transforms clinical uncertainty into diagnostic clarity through systematic modality deployment. The following frameworks synthesize key principles into immediately applicable clinical tools, enabling rapi
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for USMLE prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app




















