Rheumatology & Haematology

On this page
Foundations: Defining Inflammatory and Haematological Disorders
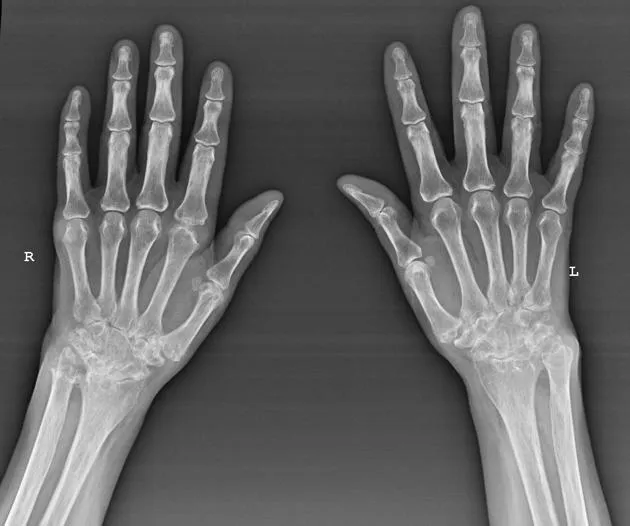
A 52-year-old woman presents with bilateral hand pain and morning stiffness lasting 90 minutes. Her metacarpophalangeal joints are swollen and tender. This clinical picture immediately raises suspicion for inflammatory arthritis, specifically , where early recognition transforms outcomes. Meanwhile, in the haematology clinic, a 68-year-old man's routine bloods reveal haemoglobin of 92 g/L with microcytosis-triggering a systematic approach to . These scenarios exemplify the precision required in rheumatological and haematological diagnosis.
Essential Classifications:
-
Inflammatory arthropathies:
- Rheumatoid arthritis (RA): Symmetrical polyarthritis, RF/anti-CCP positive in 70-80%
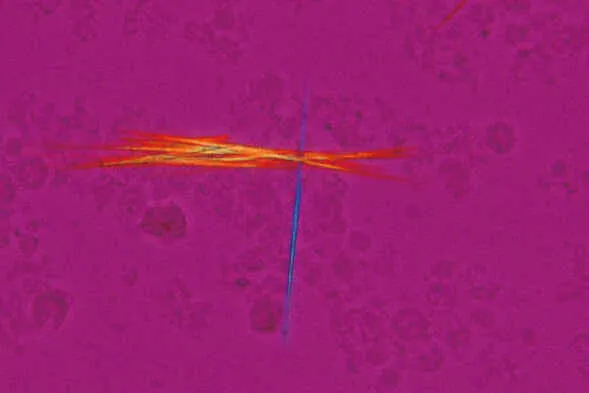
- Crystal arthropathies: Monosodium urate (gout) vs calcium pyrophosphate (pseudogout)
- Prevalence: RA affects 1% UK population; gout affects 2.5%
-
Anaemia classification by MCV:
- Microcytic (MCV <80 fL): iron deficiency, thalassaemia, chronic disease
- Normocytic (80-100 fL): acute blood loss, chronic disease, haemolysis
- Macrocytic (>100 fL): B12/folate deficiency, alcohol, hypothyroidism
-
Venous thromboembolism (VTE) risk stratification:
- Provoked vs unprovoked events determine anticoagulation duration
- Annual recurrence: 3-5% with treatment, 10% without
📌 Mnemonic for RA criteria (ACR/EULAR 2010): JSDAS - Joint involvement, Serology (RF/anti-CCP), Duration (≥6 weeks), Acute phase reactants, Score ≥6/10 confirms diagnosis
| Condition | Key Diagnostic Feature | Prevalence/Incidence |
|---|---|---|
| Rheumatoid arthritis | Anti-CCP antibody (98% specificity) | 1% UK population |
| Gout | Serum urate >360 μmol/L + crystals | 2.5% prevalence, M:F 3:1 |
| Iron deficiency anaemia | Ferritin <30 μg/L | Most common anaemia worldwide |
| Pulmonary embolism | Wells score + D-dimer/CTPA | 60-70 per 100,000/year |
Foundations: Defining Inflammatory and Haematological Disorders
2 - Pathophysiology: Understanding Disease Mechanisms
Rheumatoid arthritis exemplifies autoimmune-driven inflammation. Genetic susceptibility (HLA-DR4) combines with environmental triggers (smoking, infection) to breach immune tolerance. Anti-citrullinated protein antibodies (anti-CCP) target synovial proteins, initiating complement activation and cytokine release-particularly TNF-α, IL-6, and IL-1. This inflammatory cascade drives synovial hyperplasia, pannus formation, and cartilage destruction. Understanding this pathway explains why early DMARD therapy in prevents irreversible joint damage within the critical 3-month window.
Mechanistic Insights:
-
Crystal arthropathy pathogenesis :
- Hyperuricaemia (>360 μmol/L) → monosodium urate crystal deposition
- Crystals activate NLRP3 inflammasome → IL-1β release → neutrophil recruitment
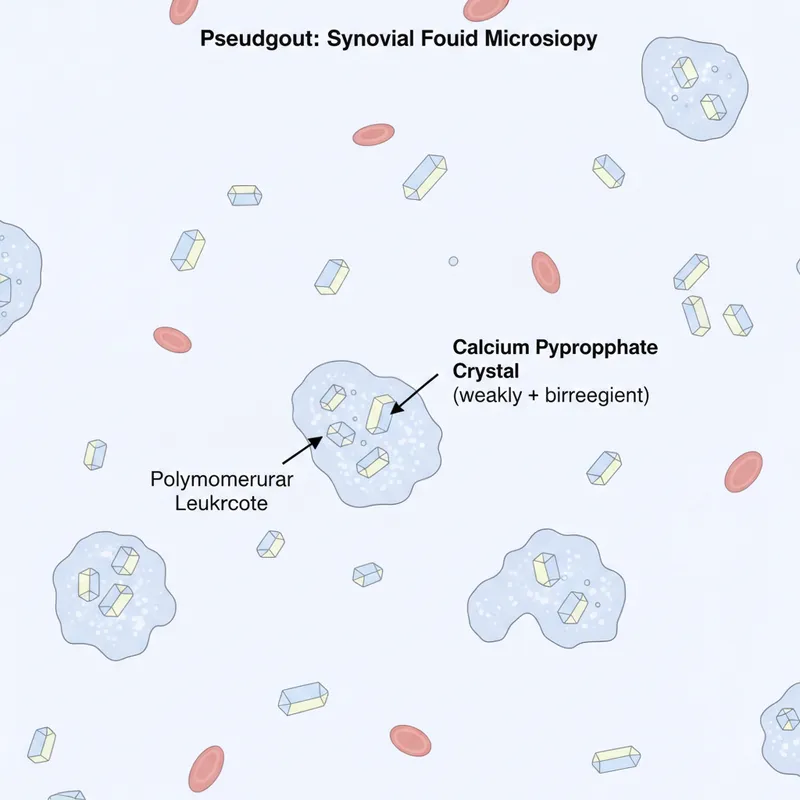
- Pseudogout: calcium pyrophosphate crystals, associated with hypercalcaemia, hyperparathyroidism
-
Anaemia mechanisms require systematic investigation :
- Iron deficiency: depleted stores → impaired haem synthesis → microcytosis
- Chronic disease: hepcidin ↑ → iron sequestration → functional deficiency
- Haemolysis: increased red cell destruction → elevated LDH, bilirubin, low haptoglobin
-
VTE pathophysiology (Virchow's triad):
- Stasis: immobility, surgery, long-haul travel
- Hypercoagulability: malignancy, thrombophilia, oestrogen
- Endothelial injury: trauma, surgery, central lines
| Mechanism | Clinical Manifestation | Therapeutic Target |
|---|---|---|
| TNF-α overproduction | Synovial inflammation, erosions | Anti-TNF biologics (adalimumab) |
| IL-1β in gout | Acute inflammatory flare | IL-1 inhibitors (canakinumab) |
| Hepcidin elevation | Anaemia of chronic disease | Treat underlying inflammation |
| Factor Xa activation | Thrombus propagation | DOACs (apixaban, rivaroxaban) |
2 — Pathophysiology: Understanding Disease Mechanisms
3 - Clinical Application: Diagnostic Approaches
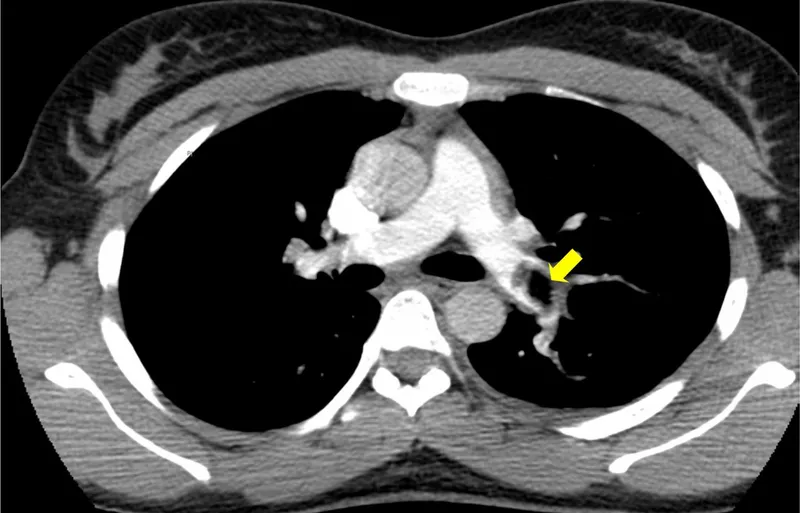
A 45-year-old presents with sudden-onset first metatarsophalangeal joint pain, erythema, and inability to weight-bear. Serum urate is 520 μmol/L, and joint aspiration reveals negatively birefringent needle-shaped crystals-confirming gout . Meanwhile, a 72-year-old with breathlessness has Wells score 6.5 (PE likely); D-dimer is 850 ng/mL, and CTPA confirms segmental pulmonary embolus . These cases demonstrate guideline-concordant diagnostic pathways.
Evidence-Based Investigation Sequences:
-
RA diagnostic workup (NICE NG100):
- Clinical suspicion: symmetrical small joint arthritis, morning stiffness >30 minutes
- Bloods: RF, anti-CCP, CRP/ESR, FBC, LFTs, U&Es (baseline for DMARDs)
- Imaging: X-ray hands/feet (erosions), ultrasound (synovitis, power Doppler)
- Refer to rheumatology within 3 working days if suspected
-
Anaemia investigation pathway :
- Step 1: FBC → identify MCV pattern
- Step 2: Microcytic → ferritin, transferrin saturation; if ferritin <30 μg/L → GI investigation
- Step 3: Macrocytic → B12, folate, TFTs, reticulocyte count
- Step 4: Normocytic → reticulocytes, blood film, haemolysis screen
-
VTE diagnosis (NICE NG158):
- Wells score: ≤1 (PE unlikely) → D-dimer; >1 (PE likely) → CTPA
- D-dimer sensitivity 95%, specificity 50% (age-adjusted threshold: age × 10 μg/L if >50 years)
3 — Clinical Application: Diagnostic Approaches
4 - Differential Diagnosis: Distinguishing Similar Conditions
Acute monoarthritis demands rapid differentiation between gout, pseudogout, septic arthritis, and trauma. Septic arthritis is the critical emergency-joint aspiration must occur before antibiotics if feasible. Synovial fluid WCC >50,000/mm³ with neutrophil predominance suggests sepsis, while crystals confirm crystal arthropathy . In anaemia, distinguishing iron deficiency from thalassaemia trait prevents unnecessary iron supplementation: ferritin differentiates, but both cause microcytosis .
Key Discriminators:
-
Monoarthritis differential:
- Gout: first MTP, rapid onset, urate crystals (negative birefringence)
- Pseudogout: knee/wrist, elderly, CPPD crystals (weak positive birefringence)
- Septic arthritis: systemically unwell, fever, WCC >50,000/mm³, Gram stain positive
- Trauma: clear history, normal inflammatory markers
-
Microcytic anaemia differentiation:
- Iron deficiency: low ferritin (<30 μg/L), high TIBC, low transferrin saturation
- Thalassaemia trait: normal/high ferritin, raised RBC count, HbA2 elevated
- Chronic disease: ferritin normal/high, low TIBC, normal HbA2
| Feature | Gout | Pseudogout | Septic Arthritis |
|---|---|---|---|
| Onset | Hours | Hours-days | Hours |
| Joint | 1st MTP (70%) | Knee (50%) | Any (knee commonest) |
| Crystals | Negative birefringent | Weak positive | None |
| Synovial WCC | 2,000-50,000 | 2,000-50,000 | >50,000 |
| Temperature | Usually afebrile | Usually afebrile | Fever >38°C |
4 — Differential Diagnosis: Distinguishing Similar Conditions
5 - Management: Evidence-Based Treatment Strategies
Early aggressive DMARD therapy in follows NICE NG100: methotrexate 15-25 mg weekly with folic acid 5 mg weekly (not on methotrexate day) is first-line. Treatment target is remission (DAS28 <2.6) or low disease activity. Failure after 3 months at therapeutic dose warrants escalation to dual/triple DMARD or biologic therapy (anti-TNF, rituximab, tocilizumab). For VTE , NICE NG158 recommends DOACs (apixaban 10 mg BD for 7 days, then 5 mg BD) over warfarin for most patients.
Specific Treatment Protocols:
-
Acute gout management (NICE CG177):
- First-line: NSAIDs (naproxen 750 mg stat, then 250 mg TDS) or colchicine (500 μg BD-QDS)
- Avoid allopurinol during acute flare (worsens inflammation)
- Urate-lowering therapy: start 2-4 weeks post-flare, target <300 μmol/L
- Allopurinol: start 100 mg OD, titrate to 300-900 mg based on response
-
Anaemia treatment depends on cause:
- Iron deficiency: ferrous sulphate 200 mg TDS (65 mg elemental iron per tablet)
- Recheck Hb after 2-4 weeks; continue 3 months post-normalisation to replete stores
- IV iron if intolerant/non-adherent/malabsorption/ongoing losses
-
VTE anticoagulation duration:
- Provoked: 3 months
- Unprovoked: consider extended (assess bleeding risk with HAS-BLED)
- Active cancer: LMWH preferred (dalteparin 200 units/kg OD for 1 month, then 150 units/kg)
| Drug | Dose | Monitoring | Key Adverse Effect |
|---|---|---|---|
| Methotrexate | 15-25 mg weekly | FBC, LFTs fortnightly × 6, then monthly | Hepatotoxicity, pneumonitis |
| Allopurinol | 100-900 mg OD | U&Es, LFTs baseline | Hypersensitivity (stop immediately) |
| Apixaban (PE) | 10 mg BD × 7d, then 5 mg BD | Renal function annually | Bleeding (no routine monitoring) |
| Ferrous sulphate | 200 mg TDS | Hb at 2-4 weeks | GI upset, constipation |
5 — Management: Evidence-Based Treatment Strategies
6 - Complex Integration: Managing Multimorbidity
A 78-year-old with RA, CKD stage 3b, and recent PE presents a management challenge. Methotrexate dose requires reduction (10 mg weekly if eGFR 30-50); apixaban needs dose adjustment (2.5 mg BD if ≥2 of: age ≥80, weight ≤60 kg, creatinine ≥133 μmol/L). Concurrent anaemia investigation reveals microcytosis; ferritin is 180 μg/L-suggesting anaemia of chronic disease from RA rather than iron deficiency. This case exemplifies real-world complexity requiring synthesis across rheumatology, haematology, and renal medicine.
Special Population Considerations:
-
Pregnancy and VTE :
- DOACs contraindicated; use LMWH throughout pregnancy and 6 weeks postpartum
- Prophylactic LMWH if previous unprovoked VTE or thrombophilia
-
Elderly with crystal arthropathy :
- NSAIDs high-risk (GI bleed, AKI, heart failure exacerbation)
- Colchicine dose-reduce in renal impairment (avoid if eGFR <10)
- Consider intra-articular steroid if monoarticular
-
Complications requiring vigilance:
- RA: cardiovascular disease (2× risk), interstitial lung disease, cervical myelopathy
- Long-term anticoagulation: major bleeding 2-3% per year, intracranial haemorrhage 0.5%
| Comorbidity | Drug Adjustment | Monitoring Frequency |
|---|---|---|
| CKD stage 3b (eGFR 30-44) | Methotrexate 10 mg weekly | FBC, LFTs, U&Es fortnightly |
| Age ≥80 + weight ≤60 kg | Apixaban 2.5 mg BD | Annual renal function |
| Heart failure | Avoid NSAIDs | Consider colchicine for gout |
6 — Complex Integration: Managing Multimorbidity
High Yield Summary
Key Take-Aways:
- RA requires urgent rheumatology referral within 3 working days if symmetrical small joint swelling with morning stiffness >30 minutes
- Anti-CCP antibody has 98% specificity for RA; methotrexate 15-25 mg weekly is first-line DMARD
- Acute monoarthritis demands joint aspiration before antibiotics to exclude septic arthritis (synovial WCC >50,000/mm³)
- Gout: treat acute flare first, delay allopurinol 2-4 weeks, target urate <300 μmol/L
- Microcytic anaemia: ferritin <30 μg/L confirms iron deficiency and mandates GI investigation in adults
- Wells score >1 for PE: proceed directly to CTPA; D-dimer only if Wells ≤1
- DOACs are first-line for VTE: apixaban 10 mg BD × 7 days, then 5 mg BD; 3 months if provoked
Essential Rheumatology & Haematology Numbers:
| Parameter | Threshold | Clinical Significance |
|---|---|---|
| Anti-CCP | >5 U/mL | 98% specificity for RA |
| Serum urate | >360 μmol/L | Hyperuricaemia, gout risk |
| Ferritin | <30 μg/L | Iron deficiency |
| MCV | <80 fL / >100 fL | Microcytic / macrocytic anaemia |
| D-dimer age-adjusted | Age × 10 μg/L (if >50 years) | Improves specificity in elderly |
| Wells score PE | >1 | PE likely, proceed to CTPA |
Key Principles/Pearls:
- Treat-to-target in RA: Aim for DAS28 <2.6; escalate therapy if not achieved within 3-6 months
- Never start allopurinol during acute gout flare-worsens inflammation by mobilizing crystals
- Ferritin is an acute phase reactant: May be falsely normal/elevated in inflammation; consider transferrin saturation
- DOACs contraindicated in pregnancy, antiphospholipid syndrome, mechanical heart valves-use LMWH or warfarin
Quick Reference:
| Condition | First-Line Treatment | Key Monitoring |
|---|---|---|
| Rheumatoid arthritis | Methotrexate 15-25 mg weekly + folic acid 5 mg weekly | FBC, LFTs monthly after stabilization |
| Acute gout | Naproxen 750 mg stat, then 250 mg TDS | Avoid allopurinol during flare |
| Iron deficiency anaemia | Ferrous sulphate 200 mg TDS | Hb at 2-4 weeks, continue 3 months post-normal |
| Pulmonary embolism | Apixaban 10 mg BD × 7d, then 5 mg BD | Annual renal function |
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Partner, to explain anything you didn't understand
Everything you need for UKMLA prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app








