Quick Overview
Tuberculosis (TB) is a notifiable infectious disease caused by Mycobacterium tuberculosis, affecting primarily the lungs (pulmonary TB) but can involve any organ system (extrapulmonary TB). NICE NG33 provides comprehensive guidance on diagnosis, treatment, and contact tracing. Key priorities include identifying active disease early, managing latent TB infection (LTBI), and recognizing drug-resistant TB to prevent transmission and complications.
Core Facts & Concepts
Diagnostic Criteria (NICE NG33)
- Active pulmonary TB: Clinical symptoms + microbiological confirmation (culture/PCR) OR radiological evidence + clinical response to treatment
- Latent TB: Positive interferon-gamma release assay (IGRA) or tuberculin skin test (TST ≥6mm if BCG vaccinated, ≥5mm if not) WITHOUT active disease
- Microbiological confirmation: Sputum smear (Ziehl-Neelsen stain), culture (gold standard, 2-6 weeks), or nucleic acid amplification test (NAAT, results in 24-48 hours)
First-Line Treatment Regimens
-
Standard 6-month regimen (drug-sensitive TB):
- Intensive phase (2 months): RIPE daily
- Rifampicin 10mg/kg (max 600mg)
- Isoniazid 5mg/kg (max 300mg) + pyridoxine 10mg
- Pyrazinamide 25mg/kg (max 2g if <50kg, 2.5g if ≥50kg)
- Ethambutol 15mg/kg
- Continuation phase (4 months): RI daily
- Intensive phase (2 months): RIPE daily
-
CNS/bone/joint TB: Extend continuation phase to 10 months (12 months total)
-
Pyridoxine supplementation: Mandatory with isoniazid (prevents peripheral neuropathy)
Drug-Resistant TB Recognition
- Risk factors: Previous TB treatment, contact with drug-resistant case, birth/residence in high-prevalence area
- Send rapid molecular testing (e.g., GeneXpert for rifampicin resistance)
- MDR-TB: Resistant to rifampicin + isoniazid (requires specialist management, 18-24 month regimens)
Problem-Solving Approach
Step-by-Step Diagnostic Pathway
- Clinical suspicion: Persistent cough >3 weeks, fever, night sweats, weight loss, haemoptysis
- Initial investigations:
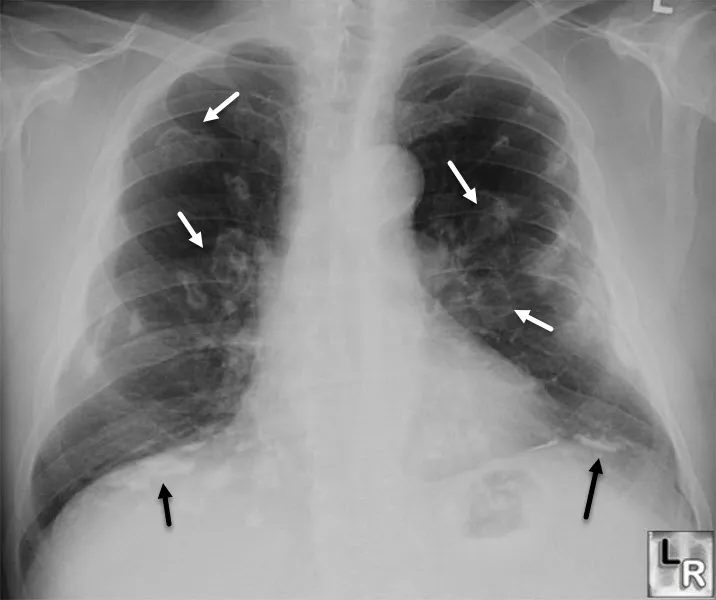
- Chest X-ray (upper lobe infiltrates, cavitation, lymphadenopathy)
- Three sputum samples (early morning) for smear, culture, and NAAT
- HIV test (mandatory in all TB cases)
- Extrapulmonary TB: Sample appropriate site (pleural fluid, CSF, lymph node biopsy)
- LTBI screening indications:
- Close contacts of active TB cases
- Immunosuppression planned (anti-TNF, transplant)
- Healthcare workers, migrants from high-prevalence countries

🚩 Red Flags Requiring Urgent Action
- Smear-positive pulmonary TB → respiratory isolation immediately
- CNS TB → dexamethasone 0.4mg/kg daily (reduces mortality)
- Drug-resistant TB suspected → specialist referral same day
- HIV co-infection → start ART within 2-8 weeks of TB treatment
Analysis Framework
| Feature | Active TB | Latent TB |
|---|---|---|
| Symptoms | Present (cough, fever, weight loss) | Absent |
| Chest X-ray | Abnormal | Normal |
| Sputum/culture | Positive | Negative |
| IGRA/TST | Usually positive | Positive |
| Treatment duration | 6-12 months (RIPE → RI) | 3 months (rifampicin + isoniazid) OR 6 months (isoniazid alone) |
| Infectivity | Contagious (if pulmonary, smear-positive) | Non-infectious |
Contact Tracing Protocol (NICE NG33)
- Close contacts: Screen with IGRA/TST and chest X-ray
- Timing: Test at initial contact; if negative and immunocompetent, repeat at 8 weeks
- Offer LTBI treatment if positive screening and active TB excluded
Visual Aid
Key Points Summary
✓ Standard treatment: 2 months RIPE (rifampicin 10mg/kg, isoniazid 5mg/kg + pyridoxine, pyrazinamide 25mg/kg, ethambutol 15mg/kg) → 4 months RI
✓ Extend to 12 months total for CNS, bone, or joint TB
✓ LTBI treatment: 3 months rifampicin + isoniazid OR 6 months isoniazid alone (prevents progression to active disease in 60-90%)
✓ Drug-resistant TB risk factors: Previous treatment, contact with resistant case, high-prevalence country origin → send rapid molecular testing
✓ Contact tracing: Screen close contacts with IGRA/TST + chest X-ray; repeat at 8 weeks if initially negative
✓ Mandatory notifications: Report all TB cases (active and LTBI treated) to local Health Protection Team within 3 days
✓ Directly observed therapy (DOT): Consider for non-adherence risk, homelessness, substance misuse, MDR-TB
⚠️ Warning: Never use monotherapy (risks resistance development). Always check HIV status and ensure pyridoxine supplementation with isoniazid.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

