Oxygen Effect - O2 Boosts Zap
- Oxygen (O2) markedly increases cellular radiosensitivity. 📌 O2 Boosts Zap!
- Mechanism: O2 "fixes" radiation-induced DNA damage by reacting with free radicals, preventing repair (Oxygen Fixation Hypothesis).
- Oxygen Enhancement Ratio (OER):
- $OER = \frac{Dose_{hypoxic}}{Dose_{aerobic}}$ for same biological effect.
- Low-LET (X-rays, γ-rays): OER ≈ 2.5 - 3.5.
- High-LET (α-particles, neutrons): OER ≈ 1.0.
- Significance: Hypoxic tumor regions are radioresistant.
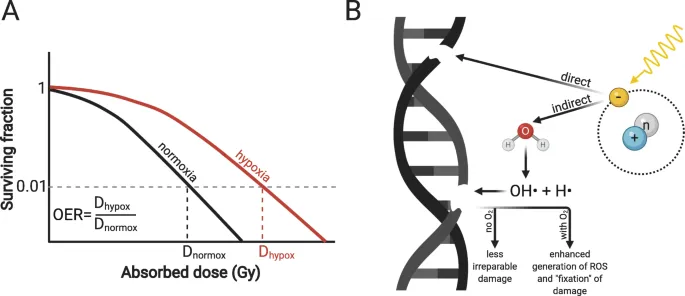
⭐ The OER for sparsely ionizing radiation (X-rays, gamma rays) is typically 2.5-3.5, while for densely ionizing radiation (alpha particles, neutrons), it approaches 1.0 due to more direct, irreparable damage.
Tumor Hypoxia - Tumors Resist Treatment
- Hypoxic tumor cells are significantly more radioresistant (Oxygen Enhancement Ratio, OER ≈ 2.5-3.5 for X-rays).
- Mechanism: Oxygen ($O_2$) "fixes" DNA damage from radiation-induced free radicals (indirect action). ↓$O_2$ → ↓damage fixation → ↑cell repair & survival.
- Types:
- Chronic (diffusion-limited): Cells distant from blood supply.
- Acute (perfusion-limited): Temporary vessel occlusion.
- Radioresistance critical if $pO_2$ < 10 mmHg.
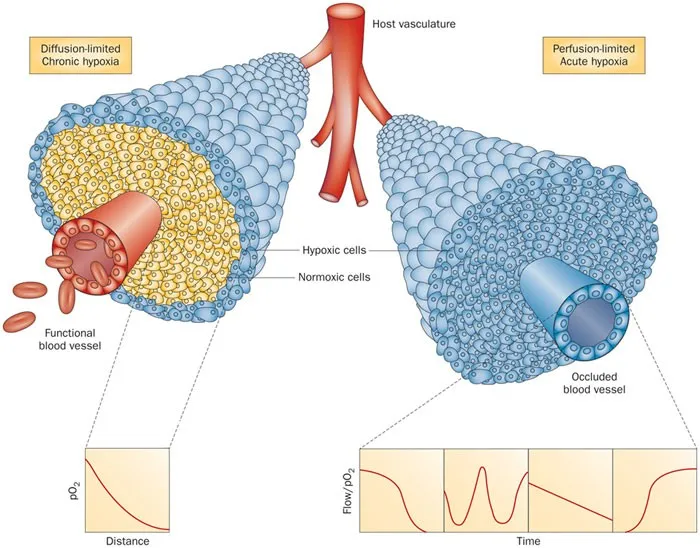
⭐ Reoxygenation, where hypoxic cells gain oxygen between radiation fractions, is key to overcoming chronic hypoxia's resistance.
Radiosensitizers Overview - Supercharging Radiation
- Compounds enhancing radiation's cell-killing effect.
- Primary aim: ↑ tumor control, especially for radioresistant hypoxic cells.
- Mechanisms vary:
- Mimic oxygen (e.g., Misonidazole).
- Inhibit DNA repair (e.g., PARP inhibitors).
- Perturb cell cycle.
- Goal: Improve therapeutic ratio - ↑ tumor damage, ↓ normal tissue injury.
⭐ Ideal radiosensitizers should be non-toxic at effective doses and selectively sensitize tumor cells, not normal tissues.
Hypoxic Cell Sensitizers - Targeting Low O2
- Reduce radioresistance of hypoxic cells (often 2-3 times > normoxic); mimic oxygen, "fix" DNA damage via bioreductive activation, preventing repair.
- Most are 2-nitroimidazole compounds. 📌 Metro Nida's Miso soup was Etan's Nimo-nic for Nitroimidazoles.
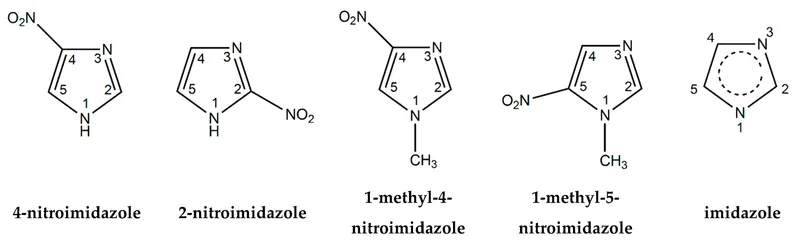
- Key Nitroimidazoles:
| Drug | Key Features | Toxicity Profile |
|---|---|---|
| Misonidazole | 1st gen; high sensitizing efficiency; benchmark compound | Peripheral neuropathy (dose-limiting) |
| Etanidazole | 2nd gen; less lipophilic than Miso, ↓ CNS penetration, ↓ toxicity | Less neurotoxic than Misonidazole |
| Nimorazole | 3rd gen; relatively low toxicity, orally active | Mildest side effects; some nausea |
Non-Hypoxic Sensitizers - Other Smart Drugs
- Halogenated Pyrimidines: (BrdU, IdU)
- Incorporate into DNA, ↑ radiation damage susceptibility.
- Thiol-Reactive Agents: (N-ethylmaleimide)
- Deplete thiols (glutathione), ↓ natural radioprotection.
- DNA Repair Inhibitors: (PARP inhibitors - Olaparib)
- Block radiation-induced DNA damage repair.
- Cell Cycle Modulators:
- Synchronize cells in radiosensitive G2/M phase.
⭐ Halogenated pyrimidines like 5-Bromodeoxyuridine (BrdU) act by incorporating into DNA, making it more susceptible to radiation damage.
High‑Yield Points - ⚡ Biggest Takeaways
- Oxygen Enhancement Ratio (OER) for low-LET radiation (X-rays, gamma rays) is typically 2.5-3.5.
- Oxygen acts as a potent radiosensitizer by fixing free-radical induced DNA damage.
- Hypoxic cells exhibit significant radioresistance, a major challenge in radiotherapy.
- Tumor hypoxia is a primary cause of local recurrence after radiation.
- Hypoxic cell sensitizers (e.g., Nimorazole, Misonidazole) mimic oxygen.
- Halogenated pyrimidines (e.g., 5-FU, BUdR) are incorporated into DNA, increasing radiosensitivity.
- Maximum oxygen effect is seen at pO2 of 20-40 mmHg; little gain above this.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

