Hypothalamic Nuclei - Brain's Appetite Command
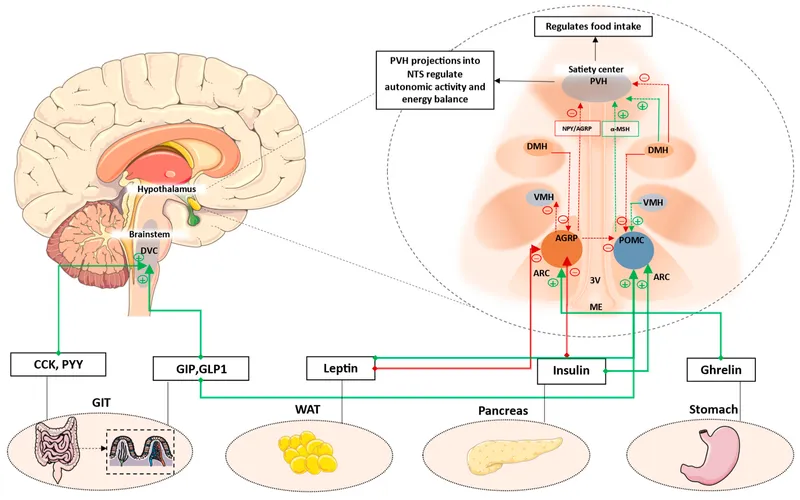
- Central Integrator: Hypothalamus, especially Arcuate Nucleus (ARC), processes peripheral hunger/satiety signals.
- Key Nuclei & Roles:
- Arcuate Nucleus (ARC): Dual neuron populations.
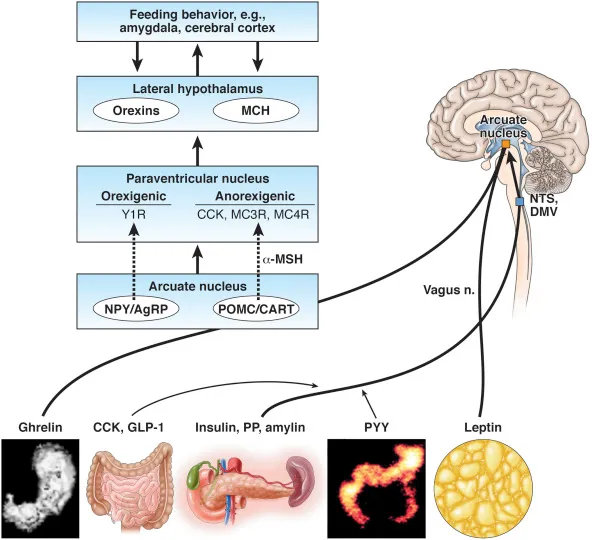
- Orexigenic: NPY/AgRP → ↑ appetite. Project to LHA.
- Anorexigenic: POMC/CART (α-MSH) → ↓ appetite. Project to VMH.
- Lateral Hypothalamic Area (LHA): "Hunger center".
- Stimulated by NPY/AgRP. Produces orexin, MCH.
- 📌 LHA lesion → Lean (aphagia).
- Ventromedial Nucleus (VMH): "Satiety center".
- Stimulated by α-MSH.
- 📌 VMH lesion → Very Massive (hyperphagia).
- Arcuate Nucleus (ARC): Dual neuron populations.
- Brainstem Link: Nucleus of Solitary Tract (NTS) relays gut satiety signals (CCK, GLP-1) via vagus nerve to hypothalamus.
⭐ Lesions in the VMH lead to hyperphagia and obesity, while LHA lesions cause aphagia and weight loss. This highlights their critical roles as satiety and hunger centers, respectively.
Gut-Brain Axis Hormones - Belly's Broadcast
GI signals to hypothalamus; regulate meal start/end.
- Orexigenic (Stimulate Eating):
- Ghrelin: 📌 'Grrr-helin' for hunger.
- Source: Stomach (P/D1 cells).
- Action: ↑ Hunger, stimulates NPY/AgRP.
- Levels: ↑ pre-meal, ↓ post-meal.
- Ghrelin: 📌 'Grrr-helin' for hunger.
- Anorexigenic (Inhibit Eating):
- Cholecystokinin (CCK):
- Source: Duodenum/Jejunum (I-cells); fats, proteins.
- Action: ↓ Intake, ↑ satiety (vagal).
- Peptide YY (PYY$_{3-36}$):
- Source: Ileum/Colon (L-cells); fats.
- Action: ↓ Intake, inhibits NPY/AgRP.
- Glucagon-Like Peptide-1 (GLP-1):
- Source: Ileum/Colon (L-cells).
- Action: ↓ Intake, ↑ satiety, incretin. (POMC/CART).
- Oxyntomodulin (OXM):
- Source: L-cells (with GLP-1/PYY).
- Action: ↓ Intake.
- Amylin:
- Source: Pancreatic β-cells (with insulin).
- Action: ↓ Intake, slows gastric emptying.
- Cholecystokinin (CCK):

⭐ Ghrelin is the only major orexigenic gut hormone; others (CCK, PYY, GLP-1, OXM, Amylin) are anorexigenic.
Adiposity & Pancreatic Signals - Long-Term Regulators
- Leptin ("Satiety Hormone"):
- Source: Adipose tissue; proportional to fat mass.
- Action: ↓Appetite (anorexigenic), ↑Energy expenditure.
- Mechanism: Hypothalamic (ARC nucleus - ↑POMC/CART, ↓NPY/AgRP).
- Deficiency/Resistance: Leads to hyperphagia, obesity.
- Insulin:
- Source: Pancreatic β-cells.
- Action: Anorexigenic signal to CNS (long-term adiposity).
- Mechanism: Acts on hypothalamus; transport across BBB.
- Amylin (IAPP):
- Source: Pancreatic β-cells (co-secreted with insulin).
- Action: ↓Gastric emptying, ↓Glucagon secretion, ↑Satiety.
- Mechanism: Acts on hindbrain (Area Postrema, NTS).
⭐ Leptin resistance, not deficiency, is the more common scenario in human obesity, characterized by high leptin levels but reduced central response.
Key Neurotransmitters & Dysregulation - Chemical Messengers & Misfires
Central neurotransmitters balance hunger/satiety. Dysregulation causes obesity/eating disorders.
- Orexigenic (Appetite Stimulating):
- NPY (Neuropeptide Y): Potent stimulator from ARC.
- AgRP (Agouti-Related Peptide): Co-released with NPY; antagonizes MC3/4R.
- Endocannabinoids (e.g., Anandamide, 2-AG): ↑ appetite (CB1R). 📌 "Munchies".
- Anorexigenic (Appetite Suppressing):
- POMC/α-MSH: ARC neurons make POMC → α-MSH. Acts on MC4R.
- CART (Cocaine- and Amphetamine-Regulated Transcript): Co-expressed with POMC; anorexigenic.
- Serotonin (5-HT): ↑ satiety, ↓ meal size (5-HT2C R).
- Dopamine: Complex role: food reward, motivation, feeding.
Dysregulation: Imbalances: Obesity (leptin resistance, MC4R mutations), Eating disorders (serotonin/dopamine pathway alterations).

⭐ α-MSH (from POMC) is key anorexigenic via MC4R. MC4R mutations: most common monogenic cause of human obesity.
High‑Yield Points - ⚡ Biggest Takeaways
- Hypothalamus is central: Lateral (LH) for hunger, Ventromedial (VMH) for satiety.
- Ghrelin (stomach) is the primary orexigenic hormone, stimulating appetite.
- Leptin (adipose tissue) is a key anorexigenic hormone, signaling long-term satiety.
- Insulin, PYY 3-36, and GLP-1 are important anorexigenic signals reducing food intake.
- Cholecystokinin (CCK) from the duodenum promotes short-term satiety.
- Arcuate nucleus (ARC) integrates signals via NPY/AgRP (hunger) and POMC/CART (satiety) neurons_
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

