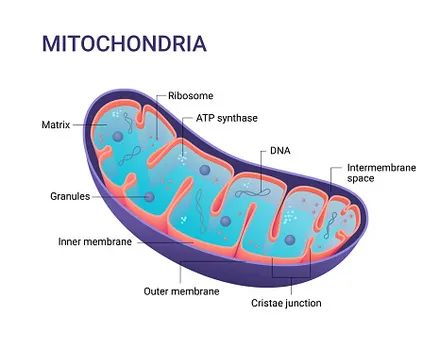
Mitochondrial Structure - Powerhouse Blueprint
- "Powerhouse": Primary site of ATP synthesis (oxidative phosphorylation).
- Double Membrane:
- Outer Membrane (OMM): Smooth, permeable (due to porins).
- Inner Membrane (IMM): Folded into cristae (↑ surface area). Houses ETC complexes & ATP synthase.
⭐ Cardiolipin in IMM makes it highly impermeable to ions, essential for maintaining the proton gradient.
- Intermembrane Space (IMS): Located between OMM & IMM. Contains cytochrome c (apoptosis).
- Matrix: Innermost space. Contains mtDNA, ribosomes, enzymes for TCA cycle, fatty acid oxidation.
Electron Transport Chain - Electron Hustle
- Location: Inner mitochondrial membrane (IMM).
- Goal: Oxidize NADH & FADH₂; transfer electrons to $O_2$; pump $H^+$ from matrix to intermembrane space (IMS), creating proton-motive force.
- Components:
- Complex I (NADH dehydrogenase): NADH → CoQ. Pumps 4$H^+$.
- Complex II (Succinate dehydrogenase): FADH₂ → CoQ. No $H^+$ pumping.
- Coenzyme Q (Ubiquinone): Mobile electron carrier.
- Complex III (Cytochrome bc₁ complex): CoQ → Cytochrome c. Pumps 4$H^+$.
- Cytochrome c: Mobile electron carrier.
- Complex IV (Cytochrome c oxidase): Cytochrome c → $O_2$. Pumps 2$H^+$. $O_2$ reduced to $H_2O$.
- Electron Flow:
- 📌 ETC Inhibitors: RotenONE (Complex I), AnAAmycin (Complex III), CO/CN⁻/H₂S IDE (Complex IV).
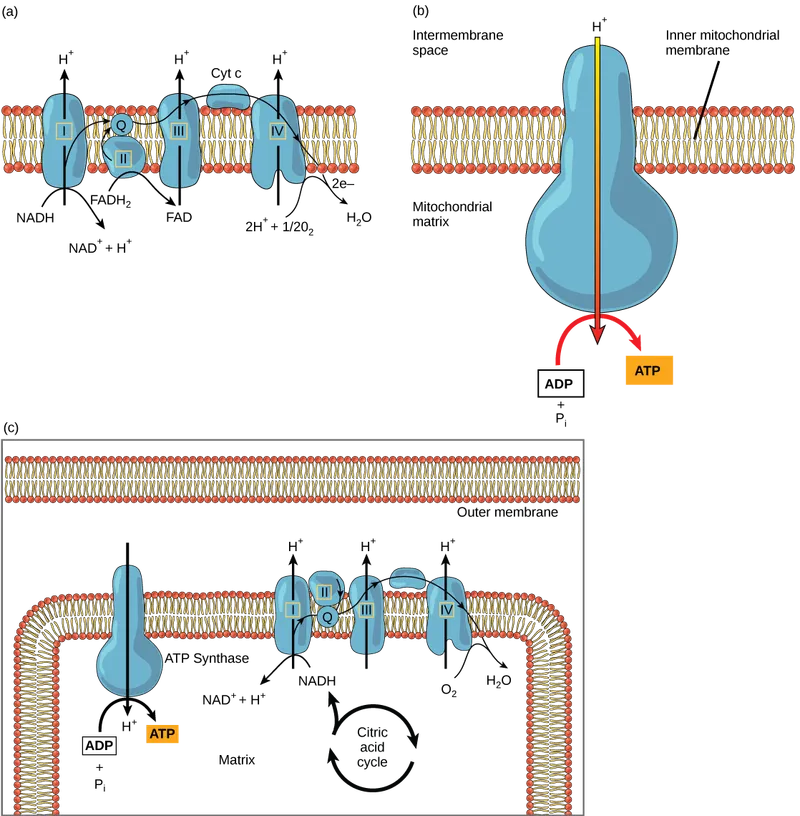
⭐ Cyanide (CN⁻) and Carbon Monoxide (CO) are potent inhibitors of Complex IV (Cytochrome c oxidase), halting electron transport and ATP synthesis, leading to rapid cellular hypoxia.
Oxidative Phosphorylation - Energy Mint
- Mechanism: ATP synthesis fueled by energy from electron transfer (NADH/FADH₂ → O₂) via ETC on Inner Mitochondrial Membrane (IMM).
- Chemiosmosis (Mitchell): ETC pumps H⁺ (matrix → Intermembrane Space/IMS), creating Proton-Motive Force (PMF; an electrochemical gradient).
- ATP Synthase (Complex V; F₀F₁ ATPase):
- F₀ (IMM): Proton channel for H⁺ re-entry to matrix.
- F₁ (matrix): Catalytic sites; $ADP + P_i + H^+{matrix} \rightarrow ATP + H_2O + H^+{IMS}$.
- P/O Ratios:
- NADH: ~2.5 ATP (enters Complex I).
- FADH₂: ~1.5 ATP (enters Complex II).
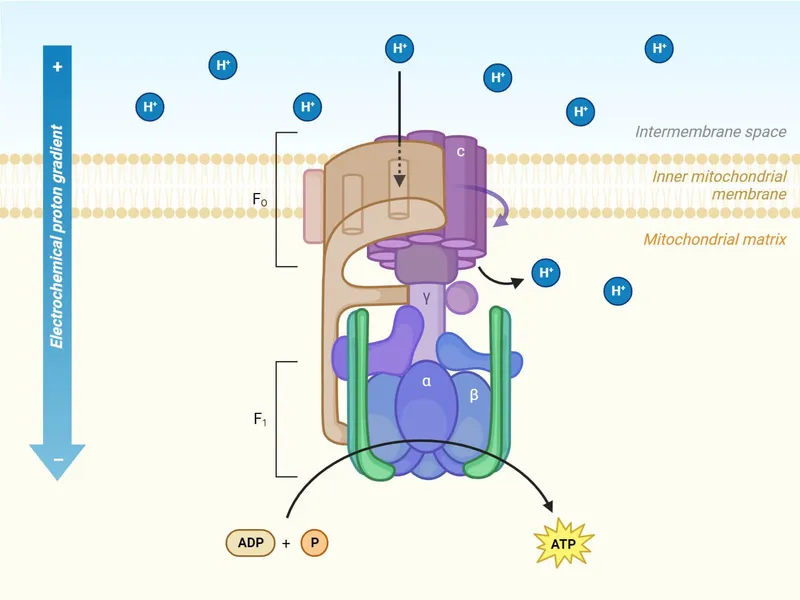
- ⭐ > Uncouplers (e.g., 2,4-DNP, aspirin overdose) dissipate proton gradient, ↑O₂ consumption, ↓ATP synthesis, produce heat. (Thermogenin is a natural uncoupler).
Mito Dysfunction & Roles - When Power Fails
- Dysfunction: Causes & Features
- Genetic: mtDNA mutations (e.g., MELAS, LHON, MERRF); nuclear DNA gene defects.
- Acquired: Ischemia/reperfusion, toxins (CN⁻, CO), drugs (zidovudine), ↑ROS.
- Results in impaired oxidative phosphorylation (OXPHOS).
- Consequences of Failure
- ↓ATP synthesis: Energy crisis, particularly in high-demand tissues (brain, heart, muscle).
- ↑ROS production: Oxidative stress, lipid peroxidation, further mtDNA damage.
- Apoptosis initiation: Release of cytochrome c, activating caspases.
- Beyond Energy: Critical Roles
- $Ca^{2+}$ homeostasis: Sequesters and releases $Ca^{2+}$, shaping signals.
- Metabolic pathways: Heme synthesis, steroid hormone synthesis (initial steps).
- Thermoregulation: Non-shivering thermogenesis via UCP1 in brown adipose tissue.
⭐ Mitochondrial DNA (mtDNA) is exclusively maternally inherited and has a high mutation rate.
oka
High‑Yield Points - ⚡ Biggest Takeaways
- Mitochondria: Powerhouses; main site of ATP synthesis via oxidative phosphorylation.
- Electron Transport Chain (ETC): Located on inner mitochondrial membrane (IMM).
- Chemiosmosis: Proton gradient across IMM drives ATP synthase (Complex V).
- Uncoupling proteins (e.g., thermogenin/UCP1) dissipate proton gradient, generating heat.
- Mitochondrial DNA (mtDNA): Maternally inherited, circular; codes for some ETC components.
- ETC Inhibitors: Rotenone (C-I), Antimycin A (C-III), CN-/CO (C-IV), Oligomycin (ATP synthase).
- Role in apoptosis (cytochrome c release) and Ca2+ homeostasis.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

