Acid-Base Basics - Proton Power Play
- Acid: Proton ($H^+$) donor (Brønsted-Lowry).
- Base: Proton ($H^+$) acceptor (Brønsted-Lowry).
- pH: Measure of $[H^+]$.
- $pH = -log_{10}[H^+]$.
- Normal blood pH: 7.35-7.45.
- pKa: Strength of a weak acid.
- $pKa = -log_{10}K_a$ ($K_a$ = acid dissociation constant).
- ↓pKa = ↑Acid strength.
- pH & pKa Relationship:
- If $pH < pKa$: Protonated form (HA) predominates.
- If $pH > pKa$: Deprotonated form ($A^-$) predominates.
- If $pH = pKa$: $[HA] = [A^-]$.
- Strong Acids/Bases: Dissociate completely.
- Weak Acids/Bases: Dissociate partially.
- Water: Dissociates into $H^+$ and $OH^-$.
- $K_w = [H^+][OH^-] = 10^{-14}$ at 25°C.
⭐ A change of 1 pH unit represents a 10-fold change in $[H^+]$ concentration.
Buffer Systems - pH pHight Club
Buffers are solutions that resist changes in pH upon addition of an acid or base. Their effectiveness is governed by the Henderson-Hasselbalch equation: $pH = pKa + log_{10}([A^-]/[HA])$ For the bicarbonate system: $pH = 6.1 + log_{10}([HCO_3^-]/(0.03 \times PCO_2))$
-
Major Physiological Buffers: 📌 Big Problems Prevented:
- Bicarbonate Buffer System:
- Most important extracellular buffer.
- Components: $H_2CO_3$ (acid) / $HCO_3^-$ (base).
- pKa = 6.1.
- Regulated by lungs ($CO_2$) and kidneys ($HCO_3^-$).
- Phosphate Buffer System:
- Important intracellular and renal tubular buffer.
- Components: $H_2PO_4^-$ (acid) / $HPO_4^{2-}$ (base).
- pKa = 6.8.
- Protein Buffer System:
- Major intracellular buffer; includes hemoglobin (Hb) in RBCs.
- Amphoteric: carboxyl (-COOH) and amino (-$NH_2$) groups. Histidine residues are key.
- Bicarbonate Buffer System:
-
Isohydric Principle: All buffer systems in a common solution (e.g., blood) are in equilibrium with the same $[H^+]$.
⭐ The bicarbonate buffer system is the most important extracellular buffer due to its high concentration and the ability of lungs and kidneys to regulate its components (PCO2 and HCO3- respectively).
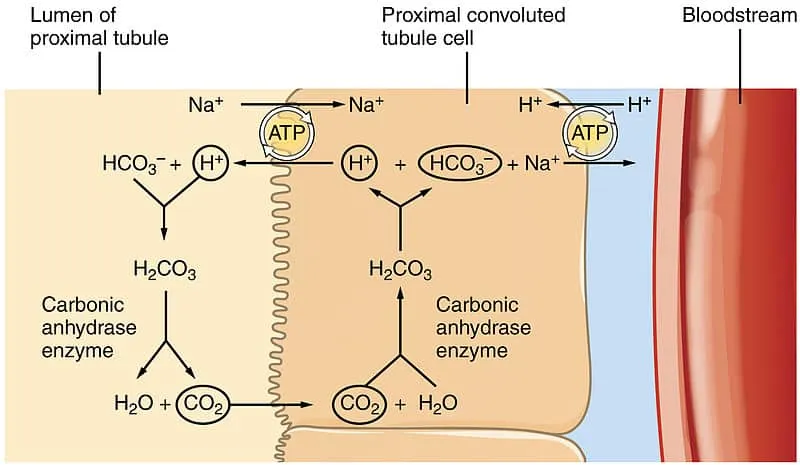
Physiological Regulation - Lungs & Kidneys Tango
Key reaction: $CO_2 + H_2O \rightleftharpoons H_2CO_3 \rightleftharpoons H^+ + HCO_3^-$ (carbonic anhydrase).
-
Lungs (Respiratory): Rapid $PCO_2$ Control (Volatile Acid)
- Adjust alveolar ventilation → alters blood $PCO_2$.
- Chemoreceptors: Central (medulla: $H^+$ in CSF from $CO_2$) & peripheral (carotid/aortic bodies: arterial $P_{O_2}$, $PCO_2$, $H^+$).
- Response: Minutes-hours; rapid, often partial.
-
Kidneys (Renal): Slow, Powerful $HCO_3^-$/$H^+$ Control (Fixed Acids)
- Regulate $HCO_3^-$ reabsorption/generation & $H^+$ secretion/excretion to manage fixed (non-volatile) acids.
- Response: Hours-days; slower onset, but provides more complete and sustained correction.
- Mechanisms:
- $H^+$ Secretion: Na-H exchanger (NHE3 in PCT), H-ATPase (collecting ducts).
- $HCO_3^-$ Reabsorption: Mainly PCT (~80-90%); also TAL, collecting ducts.
- New $HCO_3^-$ Generation: $NH_4^+$ excretion (glutamine metabolism) & titratable acid ($H_2PO_4^-$) formation.

⭐ Respiratory compensation for metabolic acid-base disturbances is rapid but often incomplete, while renal compensation for respiratory disturbances is slower but can be more complete.
High‑Yield Points - ⚡ Biggest Takeaways
- Acids are proton donors; bases are proton acceptors.
- Normal arterial pH is 7.35-7.45; $pH = -log[H^+]$.
- Henderson-Hasselbalch equation ($pH = pKa + log \frac{[Base]}{[Acid]}$) quantifies pH of buffer solutions.
- Key physiological buffers: bicarbonate (ECF), phosphate (ICF/renal), proteins (hemoglobin).
- pKa is the pH of 50% dissociation; a buffer is most effective near its pKa.
- Strong acids dissociate completely; weak acids dissociate partially, acting as buffers.
- Lungs excrete volatile acid (CO2); kidneys handle fixed acids and regenerate bicarbonate.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

