Intro to Biotransformation - Body's Cleanup Crew
- Definition: Chemical alteration of xenobiotics (drugs, toxins) & endogenous compounds within the body.
- Primary Goal: Convert lipophilic (fat-soluble) compounds into more hydrophilic (water-soluble) metabolites for easier excretion, primarily via urine or bile.
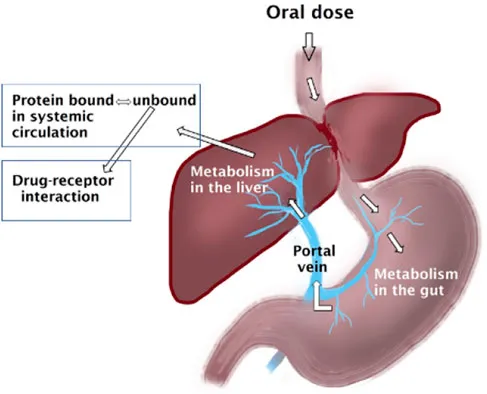
- Primary Site: Liver (smooth endoplasmic reticulum, cytosol).
- Other sites: GIT, lungs, kidneys, skin, plasma.
- Consequences:
- Inactivation of active drug (most common).
- Conversion of active drug to active metabolite.
- Conversion of inactive drug (prodrug) to active drug.
- Conversion to toxic metabolite.
⭐ Most drugs are lipid-soluble, weak acids or bases, non-polar, and therefore readily reabsorbed by renal tubules. Biotransformation makes them polar & water-soluble for excretion.
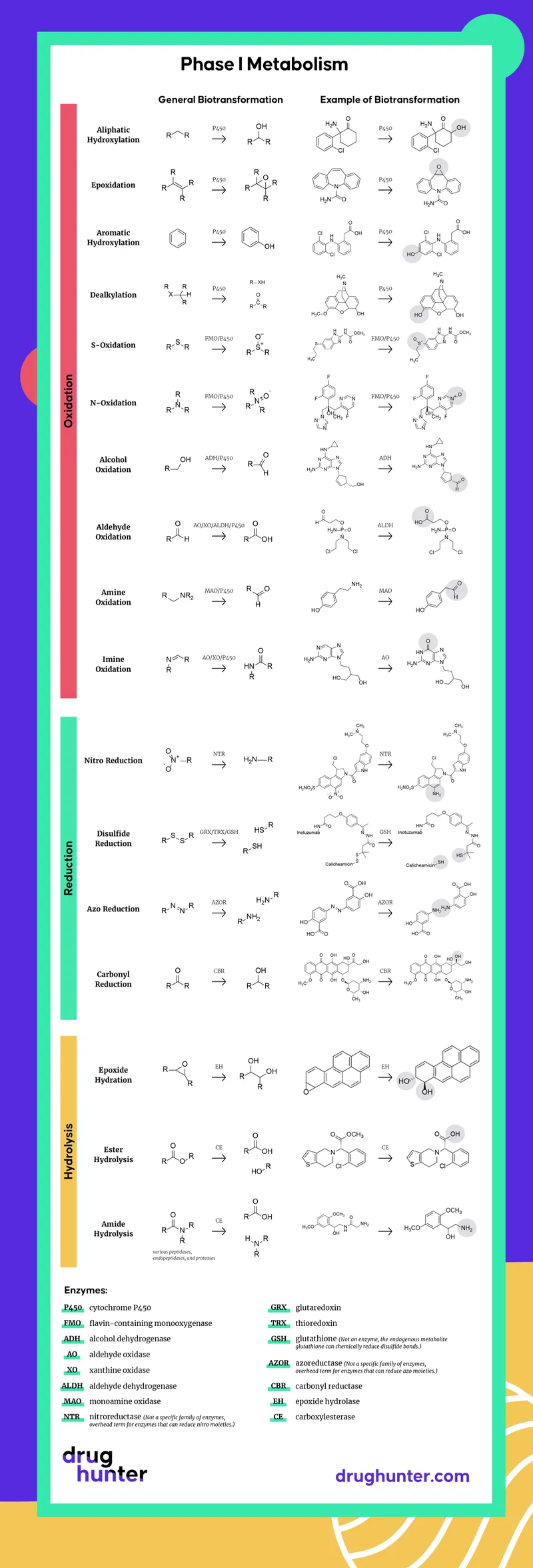
Phase I Reactions - Getting Functional
- Goal: Introduce/unmask polar functional groups (-OH, -NH2, -SH) → ↑ polarity & water solubility for excretion.
- Reaction Types: Oxidation (most common), Reduction, Hydrolysis. (📌 Mnemonic: HOR).
- Cytochrome P450 (CYP450) System:
- Primary enzymes in liver Smooth Endoplasmic Reticulum (microsomes).
- Requires: CYP450 (hemoprotein), NADPH-CYP450 reductase, NADPH, O2.
- Reaction: $Drug + O_2 + NADPH + H^+ \rightarrow Drug-Metabolite + H_2O + NADP^+$
- Key isoforms: CYP3A4/5 (metabolizes ~50% drugs), CYP2D6, CYP2C9, CYP2C19, CYP1A2.
- Non-CYP450 Enzymes:
- Alcohol/aldehyde dehydrogenase, xanthine oxidase, Monoamine Oxidase (MAO), esterases, amidases.
- Outcome: Metabolites: active, inactive, or toxic. Prodrugs often activated.
⭐ Grapefruit juice potently inhibits intestinal CYP3A4, leading to ↑ bioavailability & risk of toxicity of drugs like statins and calcium channel blockers (CCBs).
Phase II Reactions - Conjugation Junction
- Drug/metabolite + endogenous molecule → polar, excretable conjugate.
- Goal: ↑ water solubility, ↑ molecular weight, ↓ pharmacological activity.
- Reactions primarily cytosolic (except glucuronidation: ER).
- Require activated cofactors (e.g., UDP-glucuronic acid).
- Major Pathways:
- Glucuronidation: Most common. Enzyme: UDP-glucuronosyltransferase (UGT). Substrates: Bilirubin, morphine, paracetamol.
⭐ Neonates have deficient UGT activity, risking Gray Baby Syndrome with chloramphenicol.
- Sulfation: Enzyme: Sulfotransferase (SULT). Substrates: Steroids, paracetamol.
- Acetylation: Enzyme: N-acetyltransferase (NAT). Substrates: Isoniazid, sulfonamides. (📌 Slow/Fast acetylators).
- Glutathione Conjugation: Enzyme: Glutathione S-transferase (GST). Detoxifies NAPQI (paracetamol metabolite).
- Methylation: Enzyme: Methyltransferase (e.g., TPMT). Substrates: Azathioprine, 6-MP.
- Amino Acid Conjugation: With glycine, taurine. Substrates: Salicylates.

- Glucuronidation: Most common. Enzyme: UDP-glucuronosyltransferase (UGT). Substrates: Bilirubin, morphine, paracetamol.
Metabolism Modulators & Clinical Impact - Influencers & Effects
- Enzyme Induction: Process where drug ↑ enzyme synthesis/activity → ↑ its own or other drugs' metabolism.
- Inducers: Rifampicin, Phenytoin, Barbiturates, Carbamazepine, Griseofulvin, Smoking, Chronic alcohol. (📌 CRAP GPS)
- Clinical Impact: ↓ active drug efficacy, ↑ active metabolite toxicity, drug tolerance.
- Enzyme Inhibition: Process where drug ↓ enzyme activity → ↓ its own or other drugs' metabolism.
- Inhibitors: Cimetidine, Omeprazole, Ketoconazole, Macrolides (Erythromycin), Grapefruit juice, Valproate, Isoniazid, Allopurinol. (📌 G-VICK MA)
- Clinical Impact: ↑ active drug toxicity, ↓ prodrug activation & efficacy.
- Genetic Factors (Pharmacogenomics):
- CYP450 gene polymorphisms (e.g., CYP2D6, CYP2C19, CYP2C9) → variable enzyme activity.
- E.g., Slow acetylators of Isoniazid → ↑ risk of neuropathy/hepatotoxicity.
- Rapid metabolizers → may require ↑ doses for therapeutic effect.
- Other Influencers:
- Age: Neonates & elderly → ↓ metabolic capacity.
- Disease: Liver disease (e.g., cirrhosis) → significantly ↓ metabolism.
- Diet & Environment: Charcoal broiled food (inducer), grapefruit juice (inhibitor).
⭐ Grapefruit juice inhibits intestinal CYP3A4, significantly ↑ bioavailability and risk of toxicity of drugs like statins (Simvastatin, Atorvastatin), Nifedipine, and Cyclosporine. This interaction is clinically significant and often tested.
High‑Yield Points - ⚡ Biggest Takeaways
- Phase I reactions (oxidation, reduction, hydrolysis) primarily involve CYP450 enzymes, making drugs more polar.
- Phase II reactions (conjugation, e.g., glucuronidation) further ↑ water solubility for renal excretion.
- CYP450 inducers (e.g., Rifampicin, Phenobarbital) ↓ drug effect; inhibitors (e.g., Ketoconazole, Grapefruit juice) ↑ drug effect/toxicity.
- First-pass metabolism, mainly in the liver, significantly ↓ bioavailability of many oral drugs.
- Genetic polymorphisms (e.g., CYP2D6, NAT2) cause variable drug metabolism and patient responses.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

