Intro & CO - Silent Killers
- Chemical Asphyxiants: Substances that cause asphyxia by interfering with O₂ uptake, transport, or cellular utilization.
- Classification by Mechanism:
- Interference with O₂ transport (e.g., Carbon Monoxide).
- Inhibition of cellular respiration (e.g., Cyanides, H₂S).
- Classification by Mechanism:
- Carbon Monoxide (CO):
-
Sources: Incomplete combustion (fires, car exhaust, faulty heaters, generators), metabolism of methylene chloride.
-
Mechanism:
- Binds hemoglobin (Hb) with affinity ~200-250x greater than O₂ → forms carboxyhemoglobin (COHb). $CO + Hb \rightleftharpoons COHb$.
- Inhibits cytochrome c oxidase (impairs cellular respiration).
-
Clinical Features: Headache, dizziness, nausea, confusion, coma.
- Classic cherry-red discoloration of skin, lips, and viscera (often post-mortem or in severe, acute poisoning; may be absent).
-
Post-mortem Findings:
- Cherry-red tissues.
- Bilateral symmetrical necrosis of globus pallidus and hippocampus.
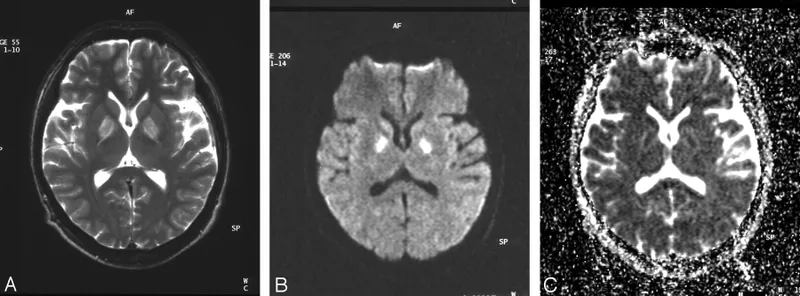
-
Fatal Levels: COHb > 50-60% in healthy adults; lower in individuals with pre-existing cardiac/respiratory conditions.
-
📌 Mnemonic for severe CO poisoning (COMA): Convulsions, Oliguria, Myocardial damage, Acidosis.
-
⭐ Methylene chloride (found in paint strippers) is metabolized in the liver to carbon monoxide, leading to delayed CO toxicity.
Cyanide Poisoning - Bitter Almonds Tale
- Sources: HCN gas, KCN/NaCN, industrial (electroplating), fires (plastics/wool), cyanogenic glycosides (apricot seeds, cassava).
- Mechanism: Inhibits mitochondrial cytochrome c oxidase (Complex IV) → blocks cellular respiration → histotoxic anoxia.
- Clinical Features: Rapid onset: headache, dyspnea, convulsions, coma. Bitter almond odor (~60% detect). Bright red venous blood.
- Post-mortem Findings: Bitter almond smell, bright red/pink lividity, gastric erosions (if ingested).
- Fatal Dose: KCN/NaCN and HCN gas fatal doses vary significantly based on individual susceptibility, route of exposure, and chemical form. ~200-300 mg orally and ~270 ppm for 30 min are approximate values only.
- Antidotes: 📌 Hydroxocobalamin is now the preferred first-line antidote, especially in smoke inhalation cases.
- Hydroxocobalamin (IV) → directly binds cyanide to form cyanocobalamin (non-toxic, renally excreted).
- Sodium thiosulfate (IV) → often co-administered, converts cyanide to thiocyanate via Rhodanese.
- Nitrites (amyl nitrite, sodium nitrite) → induce methemoglobinemia but carry significant risks (hypotension, tissue hypoxia).
- Mechanism: $CN^- + Hydroxocobalamin \rightarrow Cyanocobalamin$; $CN^- + S_2O_3^{2-} \xrightarrow{Rhodanese} SCN^-$.
⭐ In fire victims, co-oximetry should be performed to check for both COHb and methemoglobin, as cyanide poisoning is common alongside CO poisoning.
H₂S & Other Asphyxiants - Rotten Egg Menace
- Hydrogen Sulfide ($H_2S$)
- Sources: Decomposition of sulfur-containing organic matter (sewers, manure pits), industrial (petroleum refining, paper mills), natural gas.
- Mechanism: Tight binding to heme a3 in cytochrome c oxidase, inhibiting mitochondrial respiration. Cellular H₂S levels increase sharply for signaling responses before returning to low steady-state levels to prevent respiratory poisoning.
- Clinical: Rotten egg odor (detectable at 0.02-0.03 ppm); olfactory fatigue >100-150 ppm. Bimodal response dependent on H₂S concentration. Low conc: eye/respiratory irritation. High: 'knockdown', respiratory paralysis, death.
- Fatal conc: >500-700 ppm can be rapidly fatal.
- PM: Greenish tissues (esp. brain - sulfhemoglobin), transient rotten egg odor, pulmonary edema.
⭐ Olfactory fatigue with Hydrogen Sulfide is extremely dangerous as it removes the warning sign of exposure at high, life-threatening concentrations.
- Other Chemical Asphyxiants
- Hydrogen Chloride (HCl): Causes mild irritation to severe burns. Long-term exposure: respiratory problems, RADS (reactive airways dysfunction syndrome). Effects include shock, circulatory collapse, metabolic acidosis.
- Sulfur Dioxide ($SO_2$): Highly dangerous - causes convulsive airway narrowing, sudden death from respiratory arrest. Survivors suffer severe endocrine, lung, GI damage, paralysis, mental disorders.
- Phosphine ($PH_3$): Fumigation source (aluminum/zinc phosphide). Garlic/decaying fish odor. Inhibits mitochondrial respiration.
- Carbon Dioxide ($CO_2$): Simple asphyxiant, displaces $O_2$ in enclosed spaces.
High‑Yield Points - ⚡ Biggest Takeaways
- Carbon Monoxide (CO): Cherry-red PM staining; forms carboxyhemoglobin (Hb affinity 200-300x > O2).
- Hydrogen Cyanide (HCN): Bitter almond smell (inconsistent); brick-red PM staining; inhibits cytochrome oxidase.
- Hydrogen Sulfide (H2S): Rotten egg smell; greenish discoloration (tissues/brain); inhibits cytochrome oxidase.
- Phosphine (AlP): Garlicky/fishy odor; severe GI symptoms & cardiac toxicity.
- All cause histotoxic hypoxia by impairing cellular oxygen utilization.
- CO from incomplete combustion; Cyanide from burning nitrogenous compounds (wool, silk, plastics).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

