Radioisotope Techniques: Fundamentals - Radioactive Kickstart
- Radioisotopes: Unstable atomic nuclei that decay, emitting radiation (alpha, beta, gamma).
- Radioactive Decay Types:
- Alpha (α): Helium nucleus emission ($^{4}_{2}He$); low penetration.
- Beta (β): Electron (β⁻) or positron (β⁺) emission; moderate penetration.
- Gamma (γ): High-energy photon emission; high penetration.

- Units:
- Curie (Ci): $3.7 \times 10^{10}$ disintegrations per second (dps).
- Becquerel (Bq): 1 dps.
- Half-life ($T_{1/2}$): Time for 50% of a radioisotope to decay. Formula: $N(t) = N_0 (1/2)^{t/T}$ where $N_0$ is initial quantity, $N(t)$ is quantity at time $t$, and $T$ is half-life.
Common Radioisotopes in Medicine:
| Isotope | Emitted Particle(s) | Half-life | Common Clinical/Research Use |
|---|---|---|---|
| ³H | β⁻ | 12.3 yrs | Metabolic studies, autoradiography |
| ¹⁴C | β⁻ | 5730 yrs | Dating, metabolic tracer studies |
| ³²P | β⁻ | 14.3 days | DNA/RNA labeling, leukemia therapy |
| ³⁵S | β⁻ | 87.4 days | Protein labeling (e.g., methionine) |
| ¹²⁵I | γ, X-ray | 59.4 days | Radioimmunoassays (RIA), imaging |
Radioisotope Techniques: Detection - Isotope Spies
- Detection relies on ionisation (e.g., Geiger-Müller counter) or excitation (e.g., scintillation counters, autoradiography) caused by radiation.
| Detector | Principle | Radiation Detected | Sample Type | Pros | Cons |
|---|---|---|---|---|---|
| GM Counter | Radiation ionises gas; electrons cause avalanche, detected as a pulse. | $\beta$, $\alpha$, some $\gamma$ (low efficiency) | Solid, Liquid, Gas | Simple, robust, inexpensive. | High dead time, cannot identify energy/isotope. |
| LSC | Sample + scintillator cocktail; $\beta$-particles excite fluor, light detected by PMTs. | $\beta$ (esp. $^3H$, $^{14}C$), low E $\gamma$ | Liquid, Soluble | High efficiency for $\beta$, energy discrimination. | Quenching issues, sample prep, costly. |
| Gamma Counter | $\gamma$-rays interact with NaI(Tl) crystal, producing light detected by PMT. | $\gamma$, X-rays | Solid, Liquid | High efficiency for $\gamma$, energy discrimination. | Lower efficiency for low E $\gamma$, costly. |
| Autoradiography | Radiation from sample directly exposes photographic emulsion or phosphor screen. | $\alpha$, $\beta$, $\gamma$ | Tissue sections, Gels | Visualises isotope localisation. | Low sensitivity, long exposure, semi-quantitative. |

Radioisotope Techniques: Applications - Tracers & Assays
- Tracers in Metabolic Pathways:
- Follow biochemical reactions (e.g., $^{14}C$-glucose for glycolysis).
- Study DNA synthesis (e.g., $^{3}H$-thymidine).
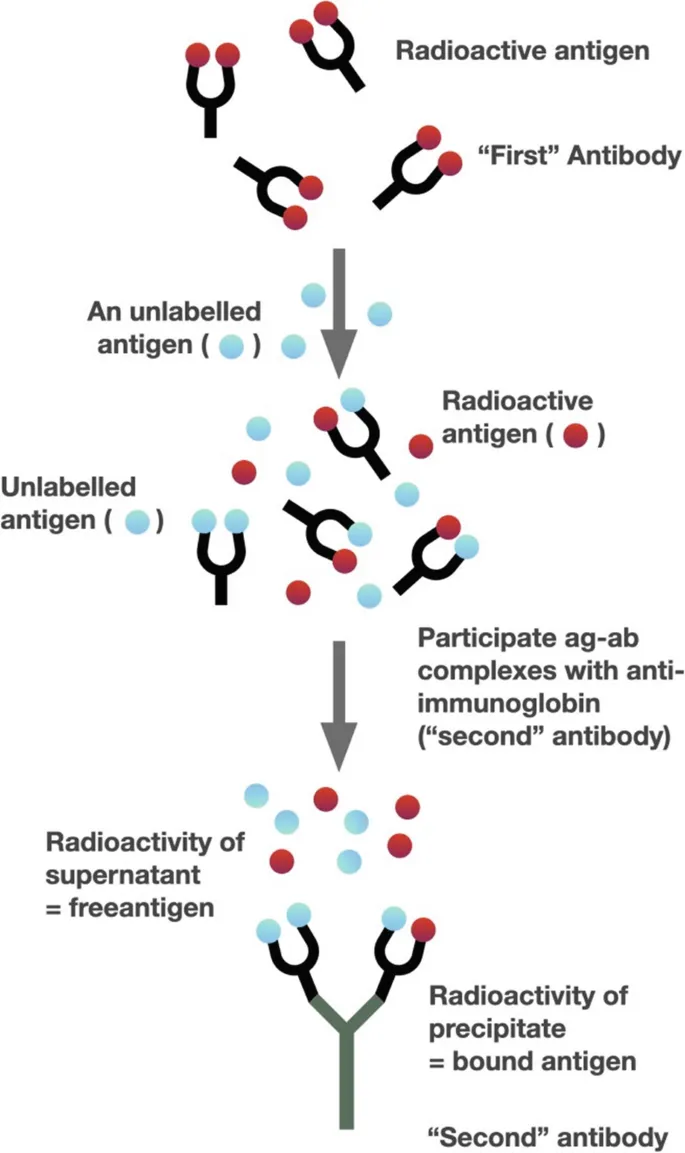
- Radioimmunoassay (RIA):
- Highly sensitive for quantifying antigens (hormones, drugs).
- Principle: Competitive binding of labeled (Ag*) and unlabeled (Ag) antigen to limited antibody.
- 📌 Mnemonic (RIA Components): Antibody, Antigen (labeled/unlabeled), Separation.
⭐ Radioimmunoassay (RIA) is a highly sensitive technique used to measure concentrations of antigens (e.g., hormones, drugs) by use of antibodies.
- Isotopic Dilution Analysis:
- Determines quantity of a substance in a sample.
- Known amount of labeled tracer added; change in specific activity measured.
- Enzyme Assays:
- Measure enzyme activity using radiolabeled substrates.
- Quantify radioactive product formation to determine reaction rates or enzyme levels.
Radioisotope Techniques: Safety - Handle With Care!
- Biological Effects of Radiation:
- Deterministic: Effects with a threshold, severity ↑ with dose (e.g., skin erythema, cataracts).
- Stochastic: Effects without a threshold, probability ↑ with dose (e.g., cancer, genetic mutations).
- Radiation Protection Principles:
- 📌 ALARA: As Low As Reasonably Achievable.
- Methods of Protection:
- Time: Minimize duration of exposure.
- Distance: Maximize distance from source (intensity $\propto 1/d^2$).
- Shielding: Use appropriate barriers (e.g., lead for X & $\gamma$-rays, perspex for $\beta$-particles).
- Safe Handling & Disposal:
- Strict adherence to protocols, proper labeling, designated areas.
- Segregation, decay storage, and authorized disposal of radioactive waste.

⭐ The ALARA principle (As Low As Reasonably Achievable) is a fundamental tenet of radiation safety, guiding practices to minimize exposure to ionizing radiation whenever possible without compromising diagnostic or therapeutic efficacy for patients or safety for workers and the public.
High‑Yield Points - ⚡ Biggest Takeaways
- Radioisotopes emit α, β, γ radiation; β-emitters like ³H, ¹⁴C, ³²P, ³⁵S are vital in biochemistry.
- Radioactivity units: Curie (Ci) and Becquerel (Bq), where 1 Ci = 3.7 x 10¹⁰ Bq.
- Detection methods include Geiger-Muller counters, Scintillation counters (liquid/solid), and Autoradiography.
- Half-life (t½) dictates experimental timing and isotope choice.
- Applications: Metabolic tracing, RIA, nucleic acid labeling, clinical diagnostics (e.g., ¹³¹I thyroid scans).
- Safety principle: ALARA - minimize exposure using time, distance, shielding.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

