Hepatobiliary Surgery

On this page
🗺️ The Hepatobiliary Blueprint: Architectural Mastery
The liver's functional anatomy determines every surgical approach, complication pattern, and resection strategy. Couinaud's segmental classification divides the liver into 8 functionally independent segments, each with dedicated portal pedicle and hepatic vein drainage-understanding this architecture enables precise anatomical resections with <5% mortality in experienced centers.
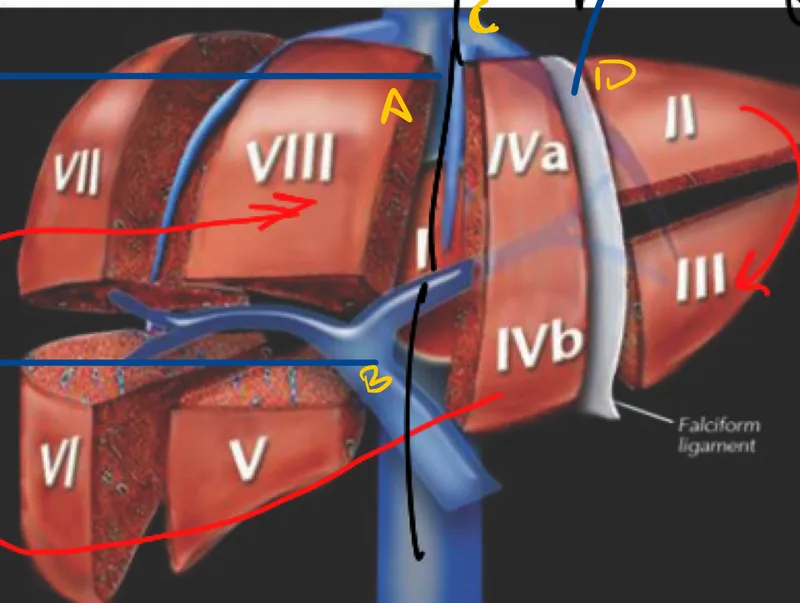
Segmental Architecture & Surgical Planes
The liver divides along three hepatic veins (right, middle, left) into hemilivers, then into 4 sectors and 8 segments. The principal plane (Cantlie's line) runs from the gallbladder fossa to the inferior vena cava, separating functional right and left lobes-this plane contains the middle hepatic vein and marks the boundary for major hepatectomies.
- Right Hemiliver (segments V-VIII)
- Anterior sector: segments V (inferior) and VIII (superior)
- Posterior sector: segments VI (inferior) and VII (superior)
- Receives right portal pedicle and drains via right hepatic vein
- Comprises 60-65% of total liver volume
- Left Hemiliver (segments II-IV)
- Left lateral section: segments II (superior) and III (inferior)
- Left medial section: segment IV (quadrate lobe)
- Receives left portal pedicle, drains via left and middle hepatic veins
- Comprises 35-40% of total liver volume
- Caudate Lobe (segment I)
- Independent blood supply from both portal pedicles
- Direct venous drainage to IVC via multiple short hepatic veins
- Hypertrophies in cirrhosis due to preserved perfusion
📌 Remember: "SALT" for hepatic vein drainage-Superior segments (II, VII, VIII) drain to hepatic veins; Anterior-right segments (V, VIII) share middle vein territory; Lateral-left segments (II, III) use left vein; The caudate goes straight to IVC
⭐ Clinical Pearl: The middle hepatic vein runs within Cantlie's line and drains portions of both hemilivers (segments V, VIII from right; IV from left)-injury during right hepatectomy causes left-sided congestion in 12-18% of cases
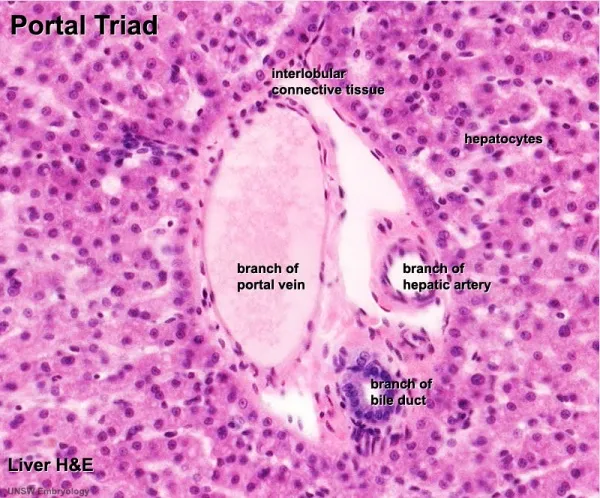
Vascular & Biliary Architecture
The liver receives dual blood supply: 75% from portal vein (nutrient-rich, deoxygenated) and 25% from hepatic artery (oxygenated). This dual perfusion protects against ischemia but creates unique injury patterns.
Portal Venous System
- Main portal vein forms behind pancreatic neck from SMV-splenic confluence
- Bifurcates at hepatic hilum into right and left branches
- Right branch: short (1-2 cm), divides into anterior and posterior sectoral branches
- Left branch: longer (3-4 cm), horizontal then umbilical portions
- Portal pressure normally 5-10 mmHg; >12 mmHg defines portal hypertension
Hepatic Arterial Supply
- Common hepatic artery from celiac trunk in 75% of cases
- Replaced/accessory right hepatic from SMA in 15-20%
- Replaced/accessory left hepatic from left gastric in 10-15%
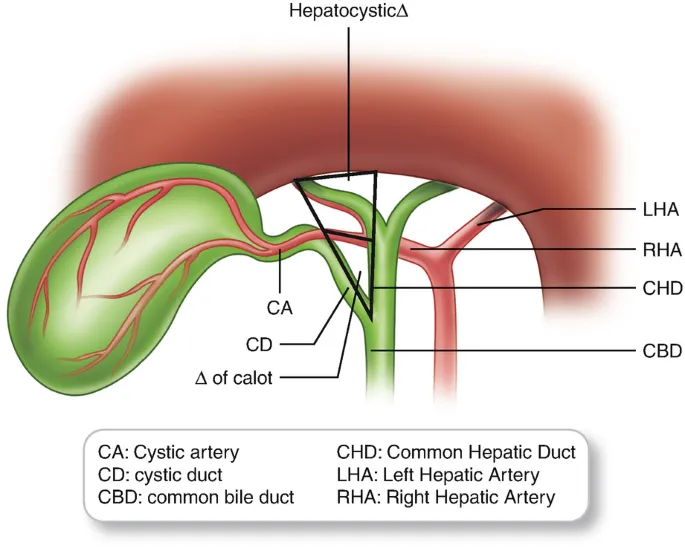
- Cystic artery typically from right hepatic artery in 75%
💡 Master This: Anatomical variants occur in 45% of patients-preoperative CT angiography identifies replaced arteries, preventing inadvertent ligation during cholecystectomy or hepatectomy that causes liver necrosis
Biliary Drainage System
- Right and left hepatic ducts join at hilum forming common hepatic duct
- Common hepatic duct + cystic duct = common bile duct (CBD)
- CBD length 6-8 cm, diameter ≤6 mm (≤8 mm post-cholecystectomy)
- Four anatomical portions: supraduodenal, retroduodenal, pancreatic, intraduodenal
- Drains at ampulla of Vater with pancreatic duct in 70%
📌 Remember: "ABCD" for bile duct dimensions-Abnormal if >6 mm; Bile duct dilates 2 mm per decade after age 60; Cholecystectomy allows 8 mm; Dilatation suggests obstruction
| Structure | Normal Diameter | Pathological Threshold | Clinical Significance |
|---|---|---|---|
| Common bile duct | 4-6 mm | >6 mm (>8 mm post-cholecystectomy) | Suggests distal obstruction |
| Portal vein | 10-13 mm | >13 mm | Portal hypertension marker |
| Hepatic artery | 4-6 mm | >7 mm | Suggests proximal stenosis/collateralization |
| Gallbladder wall | 2-3 mm | >4 mm | Acute cholecystitis if >3 mm |
| Intrahepatic ducts | <2 mm | >3 mm | Biliary obstruction |
Functional Liver Zones & Metabolic Gradients
The hepatic acinus (functional unit) demonstrates metabolic zonation from portal triad (zone 1) to central vein (zone 3), creating vulnerability gradients for different injuries.
- Zone 1 (Periportal)
- Highest oxygen tension (60-65 mmHg)
- Oxidative metabolism: gluconeogenesis, β-oxidation, cholesterol synthesis
- Resistant to ischemia, vulnerable to viral hepatitis
- Site of bile duct proliferation in cholestasis
- Zone 2 (Mid-acinar)
- Intermediate metabolism
- Mixed enzymatic activity
- Less specific injury patterns
- Zone 3 (Centrilobular)
- Lowest oxygen tension (30-35 mmHg)
- Glycolysis, lipogenesis, cytochrome P450 activity
- Most vulnerable to ischemia, toxins (acetaminophen, alcohol)
- Congestion site in right heart failure ("nutmeg liver")
⭐ Clinical Pearl: Acetaminophen toxicity preferentially destroys zone 3 hepatocytes due to concentrated P450 2E1 activity-this explains centrilobular necrosis pattern on biopsy and why >10 g ingestion causes acute liver failure in 60% of untreated cases
Connect this architectural foundation through to understand how segmental organization enables precise surgical resections and predicts complication patterns in hepatobiliary emergencies.
🗺️ The Hepatobiliary Blueprint: Architectural Mastery
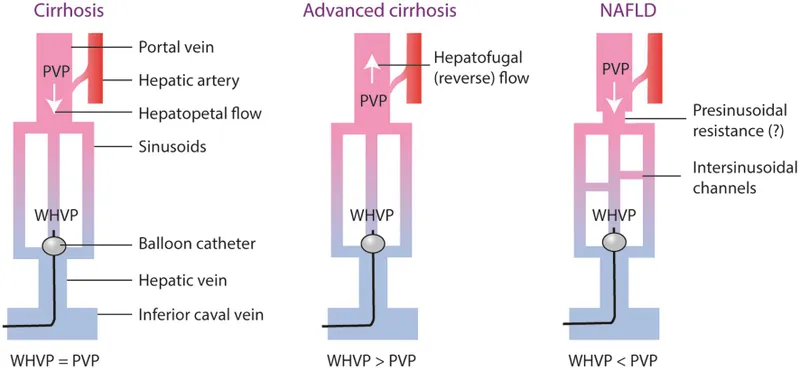
📚 Portal Hemodynamics: The Pressure System Decoded
Portal hypertension develops when portal pressure exceeds 12 mmHg (normal 5-10 mmHg), creating a pressure gradient that drives 90% of cirrhosis complications. Understanding the resistance equation-portal pressure = (portal flow × hepatic resistance) + hepatic vein pressure-reveals why reducing either flow or resistance treats variceal bleeding.
Pressure Gradient Physiology & Measurement
The hepatic venous pressure gradient (HVPG) measures the pressure difference between wedged and free hepatic vein pressures, reflecting sinusoidal portal pressure in sinusoidal portal hypertension (cirrhosis, veno-occlusive disease).
HVPG Clinical Thresholds
- ≥6 mmHg: Portal hypertension present
- ≥10 mmHg: Varices form (90% probability)
- ≥12 mmHg: Variceal bleeding risk (15%/year)
- ≥16 mmHg: Refractory ascites, hepatorenal syndrome risk
- ≥20 mmHg: Acute bleeding mortality 20% vs <5% if <20 mmHg
📌 Remember: "6-10-12-16-20" for HVPG milestones-6 = portal hypertension; 10 = varices appear; 12 = bleeding begins; 16 = refractory complications; 20 = mortality doubles
Collateral Pathway Development
When portal pressure rises, blood diverts through 4 major collateral systems, each creating distinct clinical manifestations and bleeding risks.
- Gastroesophageal Varices (left gastric → esophageal veins → azygos)
- Most clinically significant: 30% of cirrhotic patients bleed
- Esophageal varices: lower 5 cm, highest bleeding risk (25% mortality per episode)
- Gastric varices: fundal (GOV2, isolated IGV1) bleed more severely but less frequently
- Variceal size correlates with bleeding: large varices bleed at 15%/year
- Hemorrhoidal Plexus (superior rectal → middle/inferior rectal veins)
- Anorectal varices in 40% of portal hypertension
- Rarely cause significant bleeding (<5% of cases)
- Distinguished from hemorrhoids by location above dentate line
- Paraumbilical Collaterals (umbilical vein → abdominal wall veins)
- Caput medusae: dilated periumbilical veins in 15%
- Cruveilhier-Baumgarten syndrome: loud venous hum at umbilicus
- Rarely bleeds unless traumatized
- Retroperitoneal Collaterals (splenic/mesenteric → renal/lumbar veins)
- Splenorenal shunts develop spontaneously in 20-30%
- Reduce portal pressure but worsen encephalopathy
- Complicate surgical shunt procedures
⭐ Clinical Pearl: Gastric varices (GOV2/IGV1) bleed less frequently than esophageal (10% vs 25%/year) but cause more severe hemorrhage with 45% mortality due to higher flow rates and difficult endoscopic access
| Collateral System | Bleeding Frequency | Bleeding Severity | Mortality per Episode | Treatment Success |
|---|---|---|---|---|
| Esophageal varices | 25%/year | Moderate | 15-25% | 85-90% (EVL) |
| Gastric varices (GOV1) | 15%/year | Moderate-severe | 25-35% | 75-80% (EVL/glue) |
| Gastric varices (GOV2/IGV1) | 10%/year | Severe | 35-45% | 60-70% (glue/TIPS) |
| Anorectal varices | <5%/year | Mild | <5% | 90% (local measures) |
| Portal gastropathy | Chronic low-grade | Mild | <5% | 70% (beta-blockers) |
Splanchnic Hemodynamic Changes
Portal hypertension triggers systemic vasodilation through nitric oxide overproduction, creating a hyperdynamic circulation that paradoxically worsens portal pressure despite systemic hypotension.
Hemodynamic Alterations
- Splanchnic vasodilation increases portal inflow by 30-50%
- Cardiac output rises 30-40% (hyperdynamic state)
- Systemic vascular resistance drops 20-30%
- Effective arterial blood volume decreases → RAAS activation
- Sodium retention and water retention → ascites formation
💡 Master This: Beta-blockers (propranolol, carvedilol) reduce portal pressure through dual mechanisms-β1 blockade decreases cardiac output (15% reduction), β2 blockade causes splanchnic vasoconstriction (20% flow reduction)-combined effect lowers HVPG by 10-15% and reduces bleeding risk 40-50%
Pharmacological Targets
- Non-selective beta-blockers: Reduce portal pressure 12-15 mmHg → 8-10 mmHg
- Propranolol: 20-40 mg BID, titrate to heart rate 55-60 bpm
- Carvedilol: 6.25-12.5 mg daily, superior HVPG reduction vs propranolol
- Primary prophylaxis reduces first bleed from 25% → 15% over 2 years
- Nitrates (isosorbide mononitrate): Reduce intrahepatic resistance
- Combined with beta-blockers in non-responders
- Alone: limited efficacy, hypotension risk
- Vasopressin analogues (terlipressin): Splanchnic vasoconstriction
- Acute bleeding: 2 mg IV Q4h × 48-72 hours
- Controls bleeding in 75-80%, reduces mortality 35% → 25%
📌 Remember: "TIPS for HVPG" thresholds-Ten starts varices; I (roman numeral I = 1, so 1+1 = 12) bleeds; Pressure Sixteen makes refractory; every 4 mmHg rise doubles complication risk
Connect portal pressure physiology through to understand how pressure gradients determine variceal formation, bleeding risk, and guide prophylactic interventions that reduce mortality in decompensated cirrhosis.
📚 Portal Hemodynamics: The Pressure System Decoded
🔬 Biliary Stone Disease: The Crystallization Cascade
Gallstone formation follows predictable biochemical pathways-80% are cholesterol stones from bile supersaturation, 15% are black pigment stones from hemolysis, and 5% are brown pigment stones from biliary infection. Understanding nucleation factors, crystal growth promoters, and biliary stasis mechanisms predicts stone composition and guides dissolution strategies.
Cholesterol Stone Pathogenesis
Cholesterol stones form when biliary cholesterol exceeds the solubilizing capacity of bile salts and phospholipids, creating supersaturated bile that nucleates into crystals. Three defects drive this process: hepatic hypersecretion of cholesterol, decreased bile salt synthesis, and gallbladder hypomotility.
Lithogenic Triangle Components
- Cholesterol supersaturation
- Normal bile: cholesterol <6% of total lipids
- Lithogenic bile: cholesterol >10%, exceeds micellar capacity
- Cholesterol saturation index >1.0 indicates supersaturation
- Risk factors: obesity (↑HMG-CoA reductase), rapid weight loss, TPN
- Nucleation acceleration
- Mucin glycoproteins promote crystal aggregation
- Gallbladder mucosa secretes pronucleating factors in 80% of stone formers
- Antinucleating proteins (apolipoprotein A1) deficient
- Crystal formation time: 2-5 days in lithogenic bile vs >14 days in normal
- Gallbladder stasis
- Fasting residual volume >20 mL (normal <10 mL)
- Ejection fraction <40% after CCK stimulation (normal >60%)
- Pregnancy: progesterone impairs contractility, stones in 5-10%
- Vagotomy, somatostatin, octreotide reduce motility
📌 Remember: "CSN" for cholesterol stone formation-Cholesterol supersaturation; Stasis from hypomotility; Nucleation by mucin glycoproteins-all three required for stone development
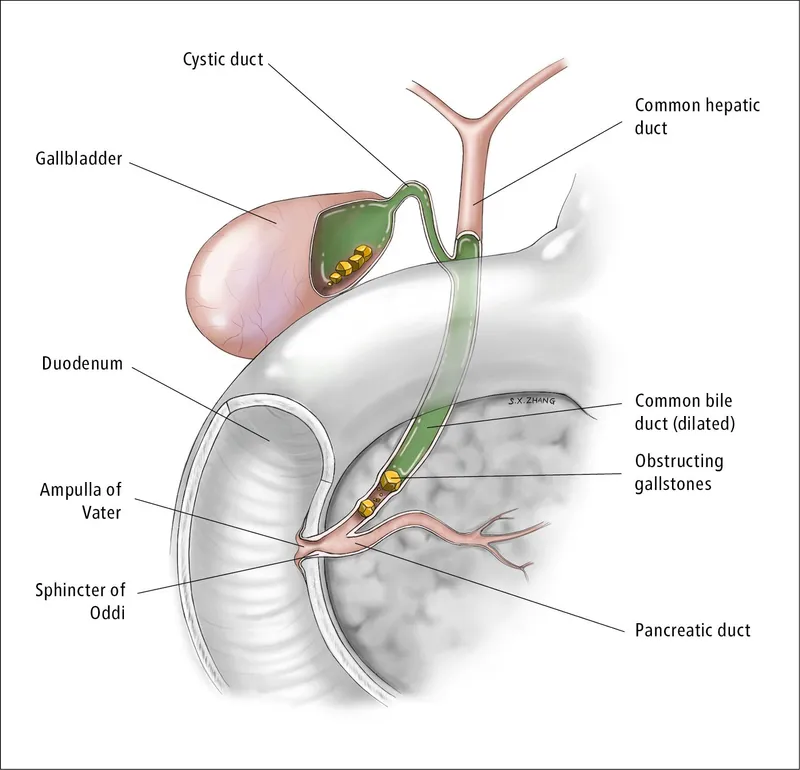
Pigment Stone Mechanisms
Pigment stones form through distinct mechanisms: black stones from unconjugated bilirubin precipitation in sterile bile, brown stones from bacterial deconjugation of bilirubin in infected bile.
- Black Pigment Stones
- Composition: calcium bilirubinate (50-70%), calcium carbonate, phosphate
- Hemolytic conditions: sickle cell (30% prevalence), hereditary spherocytosis, thalassemia
- Cirrhosis: unconjugated bilirubin ↑ from hepatocyte dysfunction (25% prevalence)
- Ileal disease/resection: bile salt malabsorption → ↓bile salt pool
- Hard, radiopaque in 50%, form in gallbladder
- Brown Pigment Stones
- Composition: calcium bilirubinate, fatty acid calcium salts, cholesterol
- Bacterial β-glucuronidase deconjugates bilirubin diglucuronide
- Associated organisms: E. coli (60%), Klebsiella, Enterococcus
- Biliary stasis: strictures, choledochal cysts, periampullary diverticula
- Soft, radiolucent, form in bile ducts, recur after extraction in 15-30%
- Endemic in East Asia: Clonorchis sinensis, Ascaris lumbricoides infections
⭐ Clinical Pearl: Black stones occur in hemolysis and cirrhosis (sterile bile), brown stones in biliary infection and stasis (bacterial deconjugation)-this distinction guides antibiotic selection and predicts recurrence risk
| Stone Type | Composition | Location | Radiopacity | Primary Mechanism | Associated Conditions |
|---|---|---|---|---|---|
| Cholesterol | Cholesterol >70% | Gallbladder | 10-15% | Supersaturation | Obesity, female, age >40 |
| Black pigment | Ca bilirubinate 50-70% | Gallbladder | 50% | Hemolysis | Sickle cell, cirrhosis, TPN |
| Brown pigment | Ca bilirubinate + fatty acids | Bile ducts | Radiolucent | Bacterial infection | Strictures, parasites, stasis |
| Mixed | Cholesterol 30-70% | Gallbladder | 20% | Combined factors | Metabolic syndrome |
Clinical Pattern Recognition
Stone location and composition determine clinical presentation patterns-gallbladder stones cause biliary colic and cholecystitis, CBD stones cause obstructive jaundice and cholangitis, intrahepatic stones cause recurrent pyogenic cholangiohepatitis.
Gallbladder Stone Presentations
- Asymptomatic stones: 80% remain silent, 1-2%/year become symptomatic
- Biliary colic: RUQ pain >30 minutes, often post-prandial, 80% recur within 2 years
- Acute cholecystitis: persistent pain >6 hours, fever, Murphy's sign in 95%
- Mirizzi syndrome: CBD compression from impacted cystic duct stone (<1%)
Common Bile Duct Stone Presentations
- Asymptomatic choledocholithiasis: 10-15% of gallstone patients, 50% pass spontaneously
- Obstructive jaundice: bilirubin >2 mg/dL, alkaline phosphatase >3× normal
- Cholangitis: Charcot triad (50-70%), Reynolds pentad (<10%, mortality 50%)
- Gallstone pancreatitis: 40% of acute pancreatitis cases, amylase >3× normal
💡 Master This: CBD diameter >6 mm on ultrasound (>8 mm post-cholecystectomy) has 73% sensitivity for choledocholithiasis-combine with elevated bilirubin >1.8 mg/dL and alkaline phosphatase >240 U/L to achieve >95% positive predictive value for CBD stones requiring ERCP
Connect stone pathophysiology through and to understand how biochemical defects translate into clinical presentations and guide timing of endoscopic versus surgical interventions.
🔬 Biliary Stone Disease: The Crystallization Cascade
🏥 Hepatic Injury Classification: Grading the Damage
Liver trauma grading follows the American Association for the Surgery of Trauma (AAST) scale from grade I (subcapsular hematoma <10% surface area) to grade VI (hepatic avulsion)-each grade predicts mortality, transfusion requirements, and need for operative intervention with >90% accuracy.
AAST Liver Injury Scale & Clinical Correlations
The AAST scale combines anatomical injury severity with hemodynamic stability to guide management-grades I-III typically manage non-operatively (85-90% success), grades IV-V require selective operative intervention (40-60%), grade VI is nearly uniformly fatal without immediate surgery.
| Grade | Injury Description | Mortality | Transfusion Need | Operative Rate | Non-op Success |
|---|---|---|---|---|---|
| I | Hematoma <10% surface, laceration <1 cm | 1-2% | 10% | 5% | 95% |
| II | Hematoma 10-50% surface, laceration 1-3 cm | 3-5% | 25% | 10% | 90% |
| III | Hematoma >50% or expanding, laceration >3 cm | 8-12% | 50% | 25% | 75% |
| IV | Parenchymal disruption 25-75% lobe | 20-30% | 80% | 60% | 40% |
| V | Parenchymal disruption >75% lobe, juxtahepatic vein injury | 40-60% | 95% | 85% | 15% |
| VI | Hepatic avulsion | >90% | 100% | 100% | 0% |
📌 Remember: "1-3-10-25-75" for AAST liver grades-grade I = 1 cm laceration; grade II = 10-50% hematoma; grade III = >3 cm laceration; grade IV = 25-75% lobe destroyed; grade V = >75% lobe destroyed
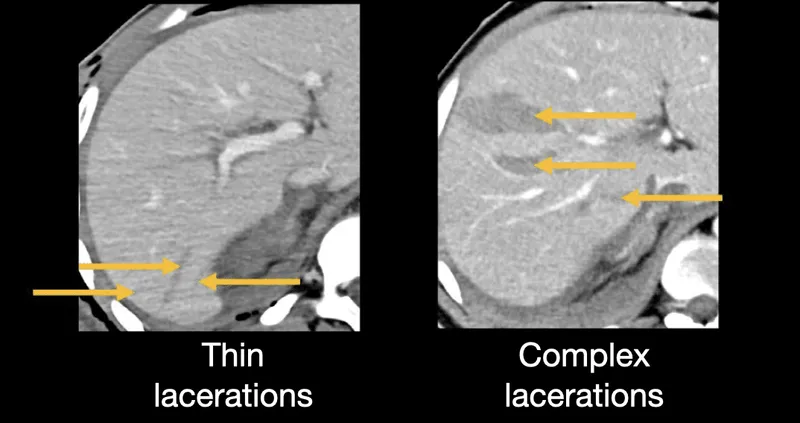
Hemodynamic Response Patterns
Patient hemodynamic status at presentation predicts management pathway better than anatomical grade alone-transient responders (stabilize with <4 units PRBC, then deteriorate) have 70% operative rate regardless of CT grade.
ATLS Shock Classification Applied to Liver Trauma
- Class I (blood loss <15%, <750 mL)
- HR <100, BP normal, mental status normal
- Non-operative management >95% success
- Observe 24-48 hours, serial Hb Q6h
- Class II (blood loss 15-30%, 750-1500 mL)
- HR 100-120, BP normal, anxious
- Non-operative management 80-85% success if transient responder
- Resuscitate, CT if stable, consider angioembolization
- Class III (blood loss 30-40%, 1500-2000 mL)
- HR 120-140, BP ↓, confused
- Operative rate 60%, angioembolization in 40% of non-operative attempts
- Damage control surgery if unstable after 4 units PRBC
- Class IV (blood loss >40%, >2000 mL)
- HR >140, BP severely ↓, lethargic
- Operative rate >90%, mortality 40-60%
- Immediate laparotomy, damage control principles
⭐ Clinical Pearl: Transient responders (initial stabilization then deterioration) have 3-fold higher mortality than sustained responders-early angioembolization within 6 hours reduces operative rate from 70% → 30% in grades III-IV injuries
Non-Operative Management Criteria & Failure Predictors
Non-operative management (NOM) succeeds in 85-90% of hemodynamically stable patients with blunt liver trauma, but 15 clinical and radiographic factors predict failure requiring delayed operation.
Absolute Contraindications to NOM
- Hemodynamic instability despite 2 L crystalloid + 4 units PRBC
- Peritonitis on physical exam (hollow viscus injury)
- Grade VI injury (hepatic avulsion)
- Associated injuries requiring laparotomy (bowel perforation)
Relative Contraindications (Higher Failure Risk)
- Grade IV-V injuries: NOM failure 15-25% vs <5% for grades I-III
- Contrast extravasation ("blush") on CT: failure 30-40% vs <10% without
- Moderate-large hemoperitoneum: failure 20% vs 5% with minimal fluid
- Coagulopathy (INR >1.5, platelets <50,000): failure 35%
- Age >55 years: failure rate doubles (20% vs 10%)
💡 Master This: Contrast blush on CT indicates active arterial bleeding-immediate angioembolization achieves hemostasis in 85-95% and reduces NOM failure from 40% → <10%, avoiding laparotomy in 75% of grade IV injuries
NOM Protocol Components
- ICU admission 24-48 hours for grades I-II, 48-72 hours for grades III-V
- Serial Hb Q6h × 24 hours, then Q12h if stable
- Bed rest 24 hours, advance activity if Hb stable
- Repeat CT if clinical deterioration or Hb drop >2 g/dL
- Transfusion threshold: Hb <7 g/dL (restrictive strategy reduces complications)
Connect trauma grading and management algorithms through to understand how anatomical injury patterns, hemodynamic responses, and imaging findings integrate into evidence-based decision pathways that optimize survival.
🏥 Hepatic Injury Classification: Grading the Damage
🔍 Endoscopic Intervention Mastery: ERCP Techniques & Complications
ERCP combines diagnostic and therapeutic capabilities-successful biliary cannulation in 90-95%, stone extraction in 85-90%, but carries 5-10% complication rate including post-ERCP pancreatitis (5-7%), bleeding (1-2%), perforation (0.3-0.6%), and cholangitis (1%). Understanding cannulation techniques, sphincterotomy principles, and complication prevention strategies optimizes outcomes.
Cannulation Techniques & Success Optimization
Selective biliary cannulation requires understanding ampullary anatomy-the bile duct enters at 11 o'clock position (viewed endoscopically) in 85%, with pancreatic duct at 5 o'clock. Failed cannulation after >10 attempts or >10 minutes increases pancreatitis risk 3-fold.
Standard Cannulation Approach
- Position sphincterotome at 11 o'clock orientation
- Advance with gentle upward pressure toward bile duct axis
- Inject contrast: immediate drainage confirms biliary position
- Success rate 80-85% on first attempt
- Wire-guided technique improves success to 90% vs contrast-guided 75%
Difficult Cannulation Strategies
- Pancreatic duct stent-assisted: Place 5-Fr × 5-cm pancreatic stent, then cannulate bile duct alongside-success 70-80%, reduces post-ERCP pancreatitis 10% → 3%
- Precut sphincterotomy: Needle-knife fistulotomy or septotomy after failed standard attempts-success 75-85%, pancreatitis risk 10-15% vs 5% standard
- Transpancreatic septotomy: Incise septum between ducts after pancreatic wire placement-success 80%, pancreatitis 8-10%
- Rendezvous technique: Percutaneous transhepatic wire placement through ampulla, snared endoscopically-success >95%, reserved for failed ERCP
📌 Remember: "5-5-5" for pancreatic stent prevention-5-French diameter, 5-cm length, reduces pancreatitis risk 5-fold (from 15% → 3%) in difficult cannulation cases
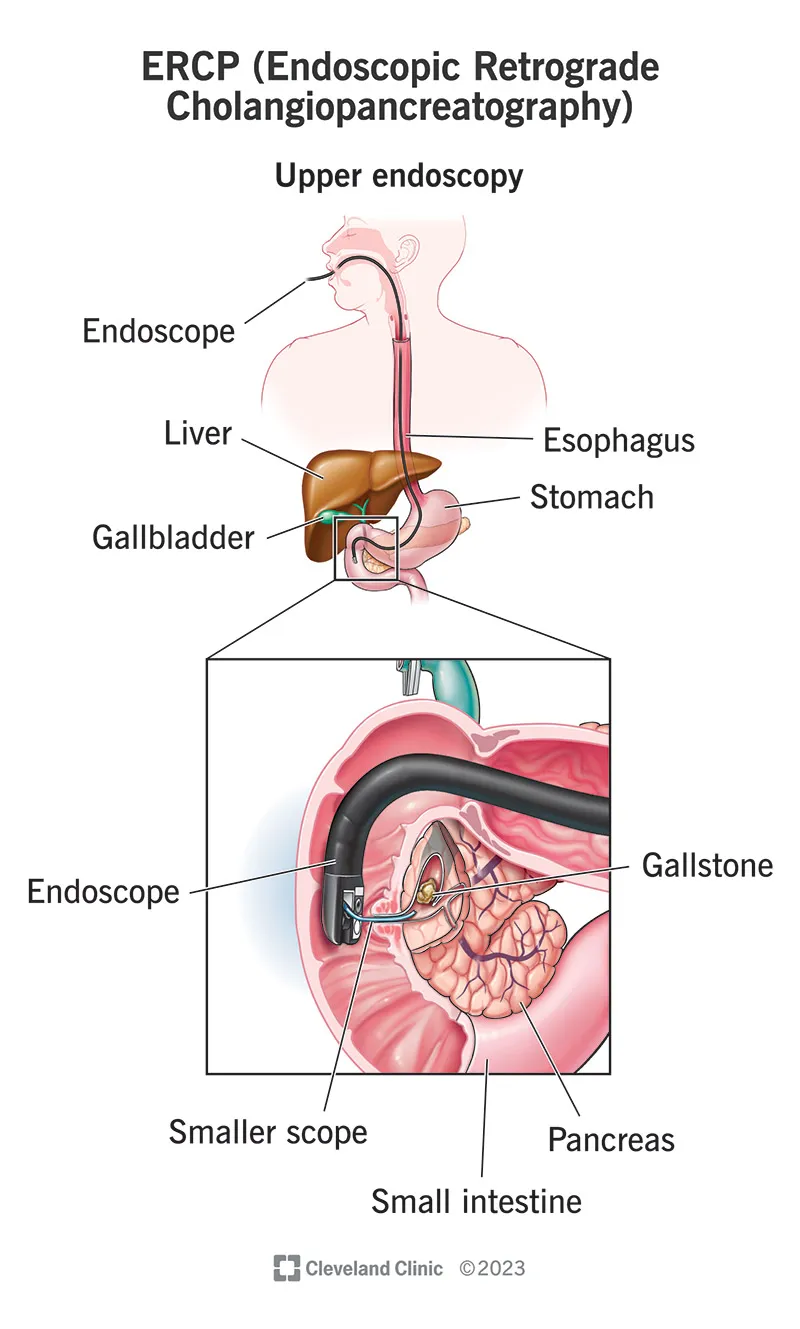
Sphincterotomy Execution & Stone Extraction
Biliary sphincterotomy creates a controlled incision in the sphincter of Oddi, extending 10-15 mm in the 11 o'clock direction to avoid duodenal perforation (risk if cutting toward 9 o'clock) or pancreatic duct injury (risk if cutting toward 1 o'clock).
Sphincterotomy Technique
- Optimal incision length: 1/3 to 1/2 of intramural bile duct
- Pure cutting current reduces bleeding vs blended current (1% vs 3%)
- Small incremental cuts safer than single large incision
- Bleeding during sphincterotomy: pause, inject epinephrine 1:10,000, apply bipolar coagulation
- Completion confirmed by easy passage of extraction balloon
Stone Extraction Methods
- Balloon extraction: stones <10 mm, success 85-90%
- Basket extraction: stones 10-15 mm, success 80-85%, fragmentation if impacted
- Mechanical lithotripsy: stones >15 mm, success 75-80%, multiple sessions often needed
- Large balloon dilation: dilate sphincterotomy to 12-15 mm for stones >15 mm-success 85%, pancreatitis risk 5-8%
⭐ Clinical Pearl: Stones >15 mm require large balloon dilation (up to 18 mm) or mechanical lithotripsy-balloon dilation achieves single-session clearance in 85% vs 60% with standard sphincterotomy alone, but carries 5% bleeding risk vs 2% standard
| Stone Size | First-line Method | Success Rate | Sessions Needed | Complication Rate |
|---|---|---|---|---|
| <10 mm | Balloon sweep | 90-95% | 1 session | 3-5% |
| 10-15 mm | Basket extraction | 80-85% | 1-2 sessions | 5-7% |
| 15-20 mm | Large balloon dilation | 85% | 1-2 sessions | 8-10% |
| >20 mm | Mechanical lithotripsy | 75-80% | 2-3 sessions | 10-15% |
| Impacted | Extracorporeal lithotripsy or surgery | 60-70% | Multiple | 15-20% |
Post-ERCP Pancreatitis Prevention
Post-ERCP pancreatitis (PEP) occurs in 5-7% overall, 15-20% in high-risk patients (young age, female sex, suspected SOD, difficult cannulation, pancreatic duct injection). Prophylactic pancreatic stenting and rectal NSAIDs reduce risk by 50-60%.
Risk Stratification
- High-risk factors (each doubles risk)
- Age <50 years, female sex (baseline risk 8-10% vs 3-4% male >60)
- Suspected sphincter of Oddi dysfunction (20-30% PEP risk)
- History of prior PEP (15-20% recurrence)
- Difficult cannulation: >10 attempts or >10 minutes (15% risk)
- Pancreatic duct injection >3 times (12% risk)
- Precut sphincterotomy (10-15% risk)
- Ampullary balloon dilation (10% risk)
Prophylactic Strategies
- Rectal indomethacin or diclofenac: 100 mg suppository immediately pre- or post-procedure
- Reduces PEP from 10% → 5% (NNT = 20)
- Most effective in high-risk patients: 20% → 8% (NNT = 8)
- Pancreatic duct stent: 5-Fr × 5-cm unflanged stent
- Reduces PEP in high-risk from 15% → 5% (NNT = 10)
- Spontaneous passage in 85% by 2 weeks
- Combined with NSAIDs: additive benefit, PEP <3%
💡 Master This: Prophylactic rectal indomethacin 100 mg plus pancreatic stent placement reduces post-ERCP pancreatitis from 20% → <3% in high-risk patients-cost-effective when baseline risk exceeds 10%, preventing 1 PEP case per 8-10 high-risk procedures
Connect ERCP techniques and complication management through to understand how procedural modifications, prophylactic interventions, and risk stratification integrate into evidence-based protocols that optimize therapeutic success while minimizing adverse events.
🔍 Endoscopic Intervention Mastery: ERCP Techniques & Complications
⚖️ Hepatocellular Carcinoma Staging: The Barcelona Blueprint
Barcelona Clinic Liver Cancer (BCLC) staging integrates tumor burden, liver function (Child-Pugh), and performance status to guide treatment-very early/early stage (0/A) candidates for curative resection or transplant (5-year survival 50-70%), intermediate stage (B) for transarterial chemoembolization (median survival 20-30 months), advanced stage (C) for systemic therapy (median survival 6-12 months), terminal stage (D) for supportive care (median survival <3 months).
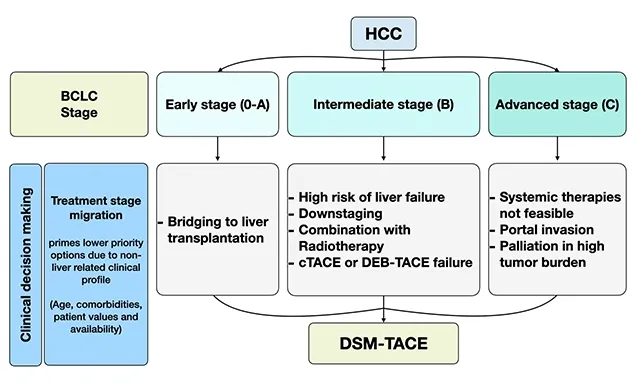
Multi-System Treatment Integration
HCC management requires coordinating hepatology (optimize liver function), oncology (systemic therapy), interventional radiology (locoregional therapy), and surgery (resection/transplant)-multidisciplinary tumor boards improve treatment selection and increase 5-year survival from 35% → 50% compared to single-specialty management.
Curative Treatment Eligibility
- Resection criteria: Single tumor any size, Child-Pugh A, portal hypertension absent
- 5-year survival 50-70% if well-selected
- Recurrence 70% at 5 years, half intrahepatic metastases, half de novo tumors
- Contraindications: Child-Pugh B/C, portal hypertension (platelets <100,000, varices), bilirubin >2 mg/dL
- Transplant criteria (Milan): Single tumor ≤5 cm OR ≤**3 tumors each ≤3 cm, no vascular invasion
- 5-year survival 65-75%, recurrence only 10-15%
- Expanded criteria (UCSF): single ≤6.5 cm OR ≤**3 tumors ≤4.5 cm, total diameter ≤8 cm-survival 60% at 5 years
- Downstaging with locoregional therapy: 40% achieve Milan criteria, similar post-transplant survival
📌 Remember: "5-3-3" for Milan criteria-5 cm single tumor; 3 tumors maximum; 3 cm each tumor-exceeding these doubles recurrence from 15% → 30% and reduces 5-year survival 75% → 50%
Locoregional Therapy Selection *
Continue reading on Oncourse
Sign up for free to access the full lesson, plus unlimited questions, flashcards, AI-powered notes, and more.
CONTINUE READING — FREEor get the app
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Practice Questions: Hepatobiliary Surgery
Test your understanding with these related questions
Which of the following statements regarding a patient of liver trauma are correct? 1. Liver is the most common organ injured following abdominal trauma. 2. Surgical exploration (laparotomy) is required in haemodynamically unstable patients and patients with free intraperitoneal fluid on FAST. 3. Contrast enhanced CT abdomen should be done in haemodynamically stable patients. 4. Blunt injuries have a higher mortality as compared to penetrating injuries.






