Atomic Structure & Radioactivity - Decay Dance
- Atom: Nucleus (protons Z, neutrons N), electrons. Mass no. A = Z+N.
- Nuclide types: 📌 (P=Protons, N=Neutrons, R=Mass numbeR)
- Isotopes: Same Z.
- Isotones: Same N.
- Isobars: Same A.
- Isomers: Same Z, N, A; diff energy state (e.g., $^{99m}Tc$).
- Nuclide types: 📌 (P=Protons, N=Neutrons, R=Mass numbeR)
- Radioactivity: Unstable nuclei decay.
- Activity = $\lambda N$. Units: Bq (1 dps); Ci ($3.7 \times 10^{10}$ dps).
- Half-life ($T_{1/2}$) = $0.693/\lambda$.
- Decay Modes (Parent $\rightarrow$ Daughter):
- $\alpha$: Emits $^{4}_{2}He$. $A \downarrow 4, Z \downarrow 2$.
- $\beta^-$: $n \rightarrow p + e^-$. $A$ same, $Z \uparrow 1$.
- $\beta^+$: $p \rightarrow n + e^+$. $A$ same, $Z \downarrow 1$. (Needs >1.022 MeV).
- EC (Electron Capture): $p + e^- \rightarrow n$. $A$ same, $Z \downarrow 1$.
- $\gamma$/IT (Isomeric Transition): Photon from excited nucleus (e.g., $^{99m}Tc \rightarrow ^{99}Tc + \gamma$). $A, Z$ same.

⭐ $^{99m}Tc$ (metastable Technetium-99) is an isomer that decays via Isomeric Transition, emitting a 140 keV gamma ray, making it ideal for SPECT imaging.
Radionuclides & Radiopharmaceuticals - Isotope Arsenal
- Radionuclide: Unstable nuclide; emits radiation.
- Ideal for imaging: Short T½, pure $\gamma$-emitter (e.g., $^{\text{99m}}\text{Tc}$).
- Ideal for therapy: Particle emitter (e.g., $\beta^-$ in $^{\text{131}}\text{I}$).
- Key Isotopes:
- $^{\text{99m}}\text{Tc}$: T½ 6 hrs, 140 keV $\gamma$. Most common. From $^{\text{99}}\text{Mo}$/$^{\text{99m}}\text{Tc}$ generator.
- $^{\text{131}}\text{I}$: T½ 8 days. $\beta^-$ & $\gamma$. Thyroid therapy/imaging.
- $^{\text{123}}\text{I}$: T½ 13.2 hrs. Thyroid imaging.
- $^{\text{18}}\text{F}$-FDG: T½ 110 mins. PET imaging.
- $^{\text{67}}\text{Ga}$: T½ 78 hrs. Tumor/inflammation.
- $^{\text{201}}\text{Tl}$: T½ 73 hrs. Myocardial perfusion.
- Radiopharmaceutical: Radionuclide + pharmaceutical (carrier). Localizes to target organ.
- E.g., $^{\text{99m}}\text{Tc}$-MDP (bone scan), $^{\text{131}}\text{I}$-NaI (thyroid).

⭐ Technetium-99m is the workhorse isotope, used in over 80% of nuclear medicine procedures.
Radiation-Matter Interaction - Energy Exchange
- Energy Transfer:
- Excitation: e⁻ to higher shell.
- Ionization: e⁻ ejected → ion pair.
- Directly ionizing: Charged particles (α, β).
- Indirectly ionizing: Uncharged (γ, X-rays).
- Linear Energy Transfer (LET): Energy/path ($keV/\mu m$).
- High LET (α): Dense ionization, short range.
- Low LET (γ, β): Sparse ionization.
- Charged Particle Interactions:
- α: Ionization/excitation.
- β: Ionization, excitation, Bremsstrahlung (X-ray production, $\propto Z^2 \times E$).
- Photon Interactions (γ, X-rays):
- Photoelectric Effect (PEA): Photon absorbed, e⁻ out. Dominant low E. $P \propto Z^3/E^3$.
⭐ PEA is crucial for contrast in diagnostic X-rays (e.g., bone vs. tissue) due to its strong Z dependence.
- Compton Scattering: Photon scatters off e⁻, loses E. Dominant at diagnostic/NM E; causes scatter.
- Pair Production: Photon > 1.022 MeV → e⁻ + e⁺. Basis of PET (→ 2 x 0.511 MeV photons).

Radiation Detection & Imaging - Photon Sniffers
-
Core Principle: Detectors convert $\gamma$-ray energy into a measurable electrical signal.
-
Detector Types:
- Gas-Filled:
- Geiger-Müller (GM) Counter: Detects radiation, area monitoring. Not for imaging.
- Scintillation Detectors:
- NaI(Tl) Crystal + PMT: Workhorse for imaging. $\gamma$-ray $\rightarrow$ light $\rightarrow$ $e^-$ $\rightarrow$ signal.
- Semiconductor Detectors:
- CZT (Cadmium Zinc Telluride): Direct conversion $\gamma$-ray $\rightarrow$ signal. $\uparrow$Energy resolution.
- Gas-Filled:
-
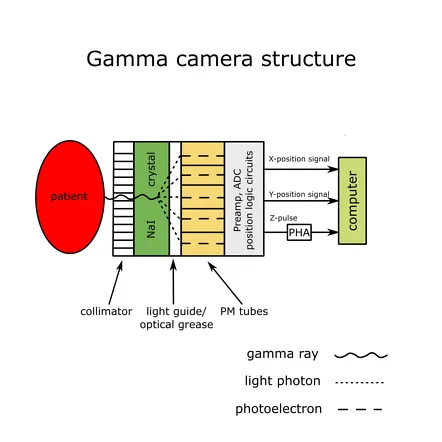
Gamma Camera (Anger Camera):
-
Key Parameters:
- Energy Resolution: Distinguishes energies (e.g., $ ^{99m}Tc $ photopeak at 140 keV). Better for scatter rejection.
- Spatial Resolution: Image detail. Collimator & crystal thickness dependent.
- Sensitivity: Detection efficiency. Thicker crystal $\uparrow$ sensitivity.
⭐ The Pulse Height Analyzer (PHA) is crucial for rejecting scattered photons, improving image contrast by selecting only photopeak events.
High‑Yield Points - ⚡ Biggest Takeaways
- Technetium-99m (Tc-99m): workhorse isotope, 140 keV gamma energy, 6-hour half-life.
- PET imaging: uses positron emitters (e.g., F-18), produces 511 keV annihilation photons.
- Gamma cameras: use NaI(Tl) scintillation crystals and photomultiplier tubes (PMTs).
- Collimators: crucial for SPECT image quality by selecting photon direction.
- Effective half-life: combines physical decay and biological clearance rates.
- ALARA principle: key for radiation safety (minimize time, maximize distance, use shielding).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

