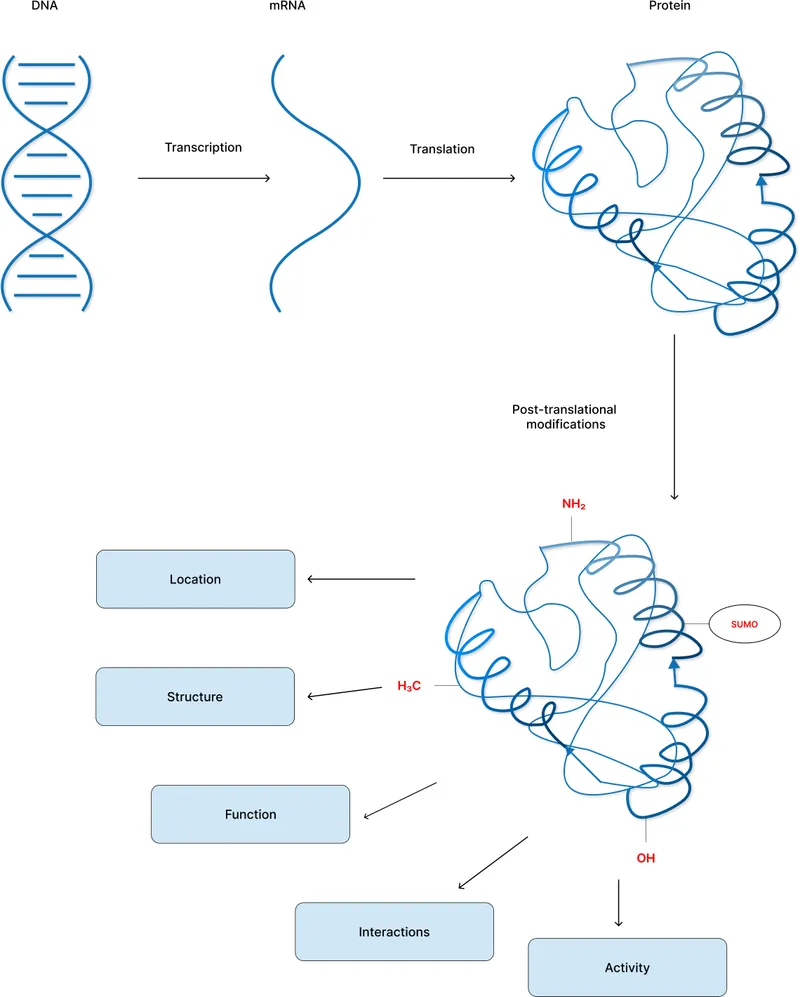
Introduction to PTMs - Protein Polish & Purpose
- Covalent chemical alterations to proteins after their synthesis on ribosomes.
- "Protein polish": Critical for transforming nascent polypeptides into mature, functional proteins.
- Vastly expand proteome complexity; >200 distinct PTM types identified.
- Essential for:
- Regulating protein activity (e.g., enzyme kinetics).
- Determining subcellular localization.
- Modulating protein-protein/protein-ligand interactions.
- Controlling protein half-life and degradation pathways.
- Occur in various cellular locations like ER, Golgi, cytoplasm.
⭐ PTMs are fundamental to cellular signaling, allowing rapid responses to stimuli.
Major PTMs I - Kinase Kisses & Sugar Coats
Phosphorylation: Reversible addition of phosphate ($PO_4^{3-}$)
- Enzymes: Kinases (add $PO_4^{3-}$ from ATP), Phosphatases (remove $PO_4^{3-}$).
- Targets: -OH of Ser, Thr, Tyr (key in eukaryotes).
- Functions: Regulates protein activity (on/off), signaling (MAPK), metabolism.
⭐ Protein kinases constitute one of the largest enzyme families, phosphorylating ~30% of all human proteins.
Glycosylation: Covalent attachment of oligosaccharides (sugars).
- Functions: Protein folding, stability, cell adhesion, immunity, signaling.
- Types:
- N-linked:
- To: Asn in Asn-X-Ser/Thr (X ≠ Pro).
- Location: ER (core, on dolichol-P) → Golgi (modify).
- Example: Immunoglobulins.
- O-linked:
- To: Ser, Thr (-OH).
- Location: Golgi, cytoplasm, nucleus.
- Example: ABO antigens, mucins. 📌 Mnemonic: N-linked to AsparagiNe. O-linked to -OH of Ser/Thr.
- N-linked:
Major PTMs II - Tag, Tuck & Tweak
- Ubiquitination: Attaching ubiquitin protein to targets.
- Pathway: 📌 E1 (activates Ub), E2 (conjugates Ub), E3 (ligase, specificity).
- **Functions**: K48-polyUb → proteasomal degradation. MonoUb/K63-polyUb → signaling, DNA repair, endocytosis.
> ⭐ Bortezomib, a proteasome inhibitor, treats multiple myeloma by preventing degradation of pro-apoptotic factors.
-
Acetylation: Adds acetyl ($CH_3CO-$) to lysine.
- Enzymes: Histone Acetyltransferases (HATs) add; Histone Deacetylases (HDACs) remove.
- Effect: Neutralizes lysine's (+) charge → loosens chromatin (euchromatin) → ↑ gene transcription.

-
Methylation: Adds methyl ($-CH_3$) to lysine/arginine.
- Enzymes: Methyltransferases add; Demethylases remove.
- Effect: Context-dependent; H3K4me3 → active transcription; H3K9me3/H3K27me3 → repressive. DNA methylation (cytosine) → gene silencing.
PTMs in Disease & Therapy - Clinical Connections
- Pathogenesis:
- Cancer: Dysregulated phosphorylation (kinases like EGFR), ubiquitination (p53, cyclins), histone PTMs (acetylation, methylation).
- Neurodegeneration: Tau hyperphosphorylation (Alzheimer's), α-synuclein PTMs (Parkinson's), huntingtin PTMs (Huntington's).
- Diabetes: Non-enzymatic glycosylation → Advanced Glycation End-products (AGEs) → complications.
- Cystic Fibrosis: CFTR protein misfolding (glycosylation defects).
- Inflammation: Altered SUMOylation, nitrosylation.
- Biomarkers:
- $HbA1c$: Glycated hemoglobin for diabetes monitoring (long-term glucose).
- Phosphorylated STAT3 (pSTAT3): Cancer prognosis.
- Circulating tumor DNA (ctDNA) PTMs: Emerging.
- Therapeutic Targets:
- Kinase inhibitors: Imatinib (BCR-ABL in CML), erlotinib (EGFR in lung cancer).
- Proteasome inhibitors: Bortezomib (multiple myeloma) - targets ubiquitination pathway.
- HDAC inhibitors: Vorinostat (lymphoma) - targets acetylation.
- Monoclonal antibodies against PTM-modified proteins.
⭐ Imatinib, a tyrosine kinase inhibitor targeting the BCR-ABL fusion protein (a result of PTM pathway dysregulation), revolutionized CML treatment.
 oka
oka
High-Yield Points - ⚡ Biggest Takeaways
- Phosphorylation by kinases (vs. phosphatases) critically regulates enzyme activity and cell signaling.
- Glycosylation: N-linked (ER, Asn) and O-linked (Golgi, Ser/Thr) affect protein folding, stability, and targeting.
- Ubiquitination: Polyubiquitin signals proteasomal degradation; monoubiquitin has other regulatory roles.
- Histone acetylation by HATs activates gene transcription; HDACs reverse this.
- Collagen synthesis requires Vitamin C for proline/lysine hydroxylation.
- Clotting factor activation needs Vitamin K for gamma-carboxylation of glutamate.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

