Osmosis Fundamentals - Water's Ways
- Movement of water (solvent) across a semipermeable membrane (SPM).
- Driven by water potential gradient: from area of ↑ water potential (↓ solute conc.) to ↓ water potential (↑ solute conc.).
- SPM: Selectively permeable; allows water, restricts most solutes.
- 📌 Water always follows solutes!
- Osmotic Pressure ($\\pi$): Hydrostatic pressure required to stop net water movement across SPM.
- Quantified by Van't Hoff's Law: $\\pi = nCRT$.
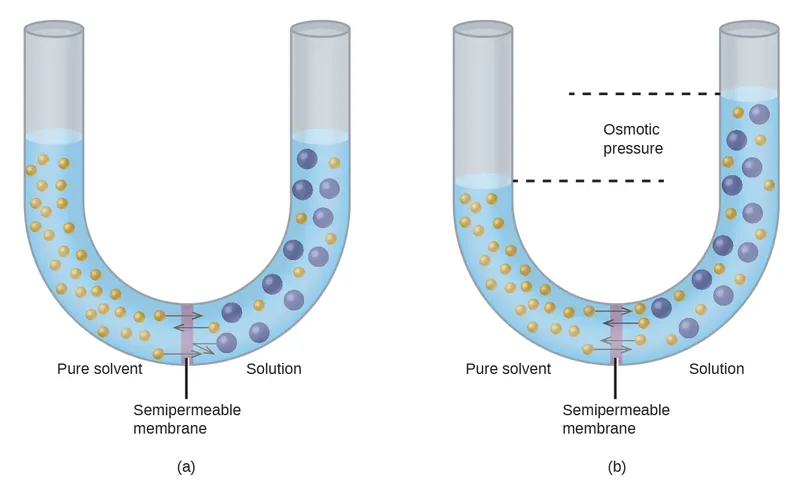
⭐ Osmosis is a colligative property; depends on the number of osmotically active particles, not their size or charge.
Osmotic Pressure - Push & Pull
- Osmotic Pressure (Π): "Pulling" pressure by solutes; pressure to prevent osmosis.
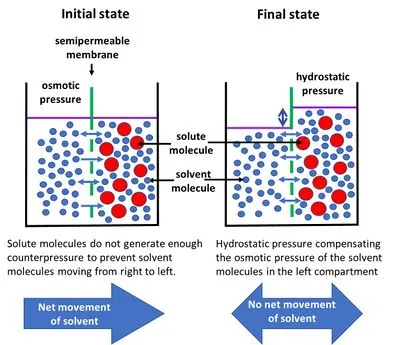
- Van't Hoff's Law: $Π = g \cdot C \cdot σ \cdot R \cdot T$
- $g$: particle count (NaCl≈2, glucose=1)
- $C$: molar concentration
- $σ$: reflection coefficient
- $R$: gas constant; $T$: absolute temp (K)
- Reflection Coefficient (σ): Solute permeability.
- $σ = 1$: Impermeable (albumin); full osmotic effect.
- $σ = 0$: Freely permeable (urea); no osmotic effect.
- Effective Osmotic Pressure: $Π_{eff} = σ \cdot Π$; dictates water flow.
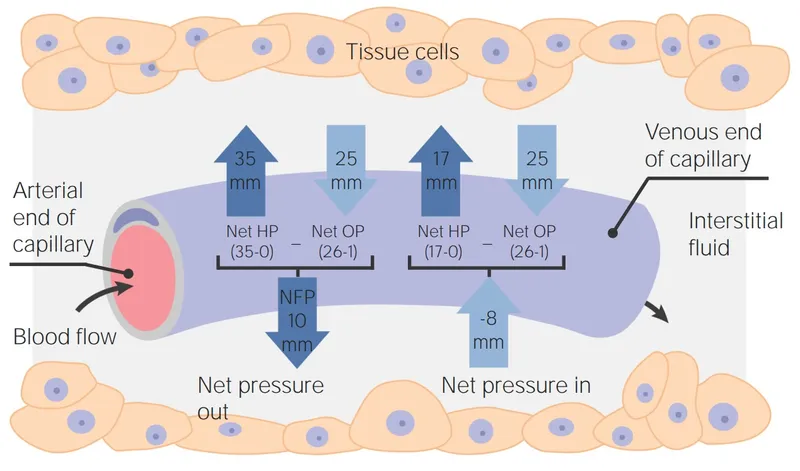
- Oncotic Pressure: Colloid osmotic pressure by plasma proteins (e.g., albumin), crucial for capillary fluid exchange. Normal: ~25 mmHg.
⭐ Plasma osmolality: 280-295 mOsm/kg H₂O. Key contributors: Na⁺, glucose, urea. Calculated: $2 \cdot [Na⁺] + [Glucose]/18 + [BUN]/2.8$ (units: Na⁺ mEq/L, Glucose mg/dL, BUN mg/dL).
Osmolarity & Tonicity - Solution Showdown
- Osmolarity: Total solute concentration in solution.
- Unit: mOsm/L.
- Plasma: 280-295 mOsm/L.
- Formula: Osmolarity = $g \times C$ ($g$=particles/mole, $C$=molar conc.).
- Osmolality: Solute concentration per kg solvent. Lab standard.
- Tonicity: Effective osmolarity; effect on cell volume. Depends on impermeable solutes.
- Isotonic: No cell volume change. E.g., 0.9% NaCl, Ringer's Lactate.
- Hypotonic: Cell swells (H₂O in). E.g., 0.45% NaCl.
- Hypertonic: Cell shrinks (H₂O out). E.g., 3% NaCl, Mannitol.
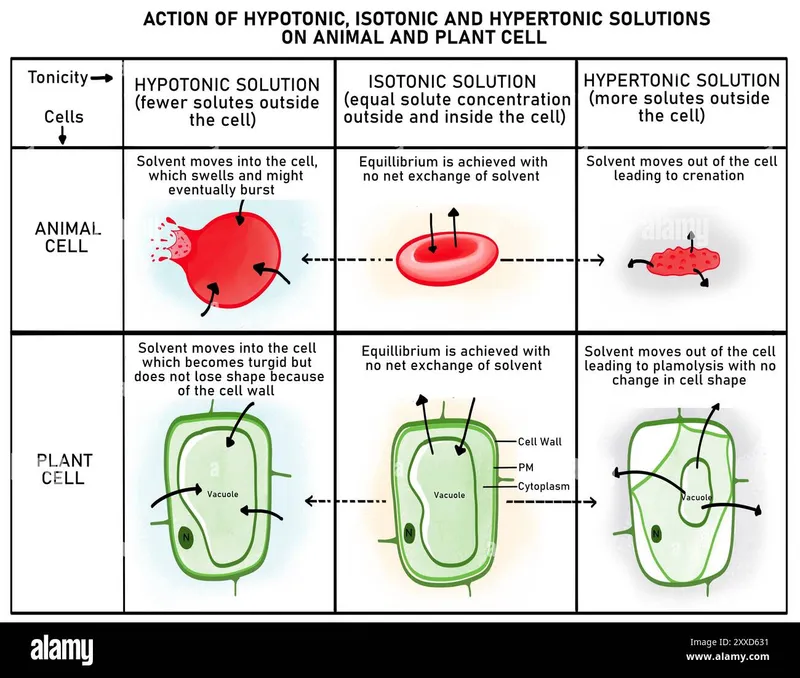
- Key Distinction:
- Osmolarity: All solutes (permeable/impermeable).
- Tonicity: Only impermeable solutes; predicts H₂O shift.
- 📌 E.g., Urea solution: isosmotic but hypotonic (urea enters cell).
⭐ D5W (5% Dextrose in water) is isotonic in vitro but physiologically hypotonic as dextrose is rapidly metabolized, leaving free water.
Clinical Correlations - Body Balance
Osmotic balance is crucial for cellular function and overall homeostasis. Disturbances lead to significant clinical issues.
- Fluid Imbalances:
- Dehydration: Water loss > intake (e.g., vomiting, diarrhea). Leads to ↑ECF osmolality, cell shrinkage.
- Water Intoxication: Excessive water intake or impaired excretion. Leads to ↓ECF osmolality, cell swelling (e.g., cerebral edema).
- Edema: Excess fluid in interstitial space.
- Caused by: ↑Capillary hydrostatic pressure, ↓Plasma oncotic pressure, ↑Capillary permeability, Lymphatic obstruction.
- IV Fluid Therapy:
- Isotonic (0.9% NaCl, Ringer's Lactate): No net water movement; for ECF volume expansion.
- Hypotonic (0.45% NaCl): Water moves into cells; for cellular dehydration. Use with caution (risk of hemolysis, cerebral edema).
- Hypertonic (3% NaCl, Mannitol): Water moves out of cells; for cerebral edema, severe hyponatremia. Use with caution (risk of osmotic demyelination).
⭐ In severe hyponatremia, rapid correction with hypertonic saline can lead to Osmotic Demyelination Syndrome (ODS). Correction rate should not exceed 8-12 mEq/L in 24h.
High‑Yield Points - ⚡ Biggest Takeaways
- Osmosis: Water movement via semipermeable membrane from low to high solute concentration.
- Osmotic pressure: Pressure to stop osmosis; depends on solute particle count.
- Van't Hoff's Law quantifies osmotic pressure; 'n' (dissociable particles) is key.
- Osmolality (mOsm/kg solvent) clinically preferred over osmolarity (mOsm/L solution).
- Tonicity (iso-, hypo-, hypertonic) dictates cell volume changes.
- Plasma osmolality: 275‑295 mOsm/kg; main solutes: Na+, glucose, BUN.
- Reflection coefficient (σ): 0 (freely permeable) to 1 (impermeable); impacts effective osmotic pressure.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

