Definitions & Principles - Water's Ways
- Osmosis: H₂O movement, semipermeable membrane (low → high solute).
- Osmole: 1 mole non-dissociable solute.
- Osmolality: Osmoles/kg solvent. Plasma: 275-295 mOsm/kg H₂O.
- Plasma Osmolality $\approx 2 \times [Na^+]{pl} + \frac{[Glucose]{pl}}{18} + \frac{[BUN]_{pl}}{2.8}$ (glucose/BUN in mg/dL).
- Osmolarity: Osmoles/L solution.
- Tonicity: Effective osmolality; dictates H₂O shift.
- Isotonic: No H₂O shift.
- Hypotonic: H₂O enters cell. 📌 HypOtonic solutions make cells swell like an O.
- Hypertonic: H₂O exits cell.
- Effective osmoles: Non-penetrating (Na+, glucose); determine tonicity.
- Ineffective osmoles: Penetrating (urea).
- Oncotic Pressure: Osmotic pressure by plasma proteins (albumin).
| Aspect | Osmolality (mOsm/kg) | Osmolarity (mOsm/L) | Tonicity |
|---|---|---|---|
| Basis | /kg solvent | /L solution | Effective osmoles |
| Key | Temp independent | Temp dependent | Membrane dependent |
⭐ Tonicity is solely determined by solutes that do not cross the cell membrane (non-penetrating or effective osmoles), dictating water movement across the membrane.
Regulation Mechanisms - Body's Balancing Act
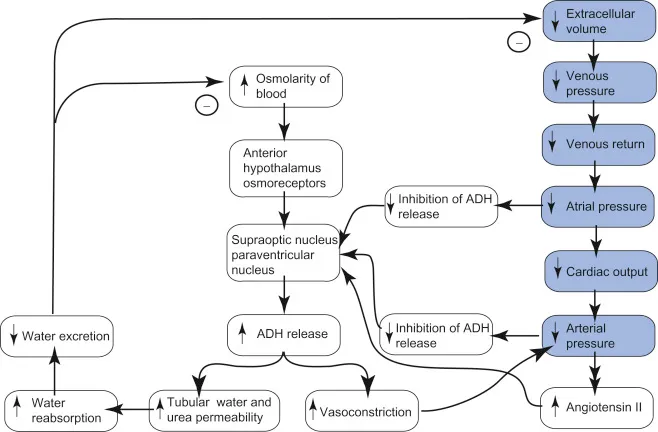
- Osmoregulation: Primarily via Antidiuretic Hormone (ADH) & thirst.
- Osmoreceptors: Located in the hypothalamus; detect changes in plasma osmolality.
- ADH release is triggered when plasma osmolality rises above ~280-285 mOsm/kg.
- Thirst is typically perceived when plasma osmolality reaches ~290-295 mOsm/kg.
- ADH (Vasopressin): Synthesized in hypothalamus, released from posterior pituitary.
- Acts on V2 receptors in the principal cells of the kidney's collecting ducts.
- Promotes insertion of aquaporin-2 water channels into the apical membrane.
- Result: ↑ water reabsorption, ↓ plasma osmolality, concentrated urine.
- 📌 Mnemonic: ADH = Adds Da H₂O (to body).
- Thirst Center: Located in the hypothalamus; stimulation leads to increased water intake.
- Osmoreceptors: Located in the hypothalamus; detect changes in plasma osmolality.
⭐ Plasma osmolality is the primary determinant of ADH secretion; a 1% change can significantly alter ADH levels.
Clinical States & Fluids - When Balance Breaks
-
Common IV Fluids & Effects:
Fluid Tonicity Use(s) ⚠️ Cautions 0.9% NaCl Isotonic ECF expansion Overload, ↑Cl⁻ LR Isotonic Burns, surgery HyperK⁺, liver fail 0.45% NaCl Hypotonic HyperNa⁺, H₂O deficit ↑ICP, rapid Na⁺↓ D5W Hypotonic Free H₂O, diluent ↑ICP, hyperglycemia 3% NaCl Hypertonic Severe hypoNa⁺, cerebral edema ODS, central line Mannitol Hypertonic Cerebral edema, ↑IOP AKI, fluid shifts -
📌 Mnemonic: 'Hypertonic: ENTERS vessel (cell shrinks); Hypotonic: GOES OUT to cell (cell swells)'.
-
Key Clinical States:
- Dehydration: ECF↓, ICF↓ (hypertonic).
- SIADH: HypoNa⁺, ICF↑ (cell swell).
- DI: HyperNa⁺, ICF↓ (cell shrink).
- Cerebral Edema: Brain swell; use hypertonic fluids.
- ODS: Risk if chronic hypoNa⁺ corrected fast. Max Na⁺ rate: 8-10 mEq/L/24h.
⭐
D5W: Isotonic in bag, hypotonic in vivo (dextrose used), yields free water.

Osmolal Gap Calculation - Detective Work
- Osmolal Gap: A diagnostic tool. Difference between:
- Measured serum osmolality (by osmometer)
- Calculated serum osmolality
- Formula: Osmolal Gap = Measured Osmolality - Calculated Osmolality
- Calculated Osmolality (estimate): $2 \times [Na^+] + \frac{[Glucose]}{18} + \frac{[BUN]}{2.8}$ (Na in mmol/L; Glucose, BUN in mg/dL)
- Normal Gap: < 10-15 mOsm/kg H₂O. An ↑ gap suggests presence of unmeasured osmoles.
- Causes of ↑ Osmolal Gap (📌 Mnemonic: 'ME DIE P'):
- Methanol
- Ethylene glycol
- Diuretics (osmotic, e.g., Mannitol), DKA (severe diabetic ketoacidosis)
- Isopropyl alcohol
- Ethanol (alcoholic ketoacidosis, acute intoxication)
- Propylene glycol (e.g., in IV medications)
- Sorbitol
⭐ An elevated osmolal gap in a patient with altered mental status and metabolic acidosis is highly suggestive of toxic alcohol ingestion.
High‑Yield Points - ⚡ Biggest Takeaways
- Osmolality is the measure of solute concentration per kilogram of solvent (mOsm/kg H₂O).
- Tonicity describes a solution's effect on cell volume, determined by impermeable solutes.
- Plasma osmolality is chiefly determined by sodium, glucose, and urea (BUN). Calculated as 2[Na⁺] + [Glucose]/18 + [BUN]/2.8.
- Effective osmolality (or tonicity) excludes freely permeable solutes like urea.
- Isotonic solutions (e.g., 0.9% NaCl, Ringer's lactate) cause no net water movement, hence no change in cell volume.
- Hypertonic solutions draw water out of cells (cell shrinks); hypotonic solutions cause water to move into cells (cell swells).
- Hypothalamic osmoreceptors are crucial, sensing plasma osmolality to regulate ADH secretion and thirst.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

