DRC Basics & Curve Types - Dose Makes the Difference
- Dose: Amount of drug. Response: Effect observed.
- Dose-Response Relationship: Correlates dose with effect magnitude.
- Graded Response:
- Effect intensity varies with dose in an individual.
- Example: Blood pressure change.
- Curve: Hyperbolic (linear dose) or sigmoid (log dose).
- Quantal Response:
- All-or-none effect in a population (e.g., sleep/cure).
- Shows frequency of response.
- Curve: Typically sigmoid.
- DRC Shapes (General):
- Hyperbolic: Response vs. linear dose.
- Sigmoid (S-shaped): Response vs. log dose; for $ED_{50}$, $E_{max}$ analysis.
⭐ Graded dose-response curves depict the intensity of effect in an individual, while quantal curves show the frequency of response in a population.
Graded Dose-Response Curves - Efficacy vs. Might-acy
- Graded DRCs plot drug effect (response) against dose or concentration.
- Efficacy ($E_{max}$): Maximum effect a drug can produce. Represents the drug's maximal therapeutic potential. Higher $E_{max}$ = ↑ efficacy.
- Potency ($EC_{50}$ or $ED_{50}$): Dose/concentration required to produce 50% of $E_{max}$. Lower $EC_{50}$/$ED_{50}$ = ↑ potency.
- Primarily reflects drug-receptor binding affinity.
- Slope of DRC: Indicates the range of doses over which response changes; steep slope = narrow therapeutic window.
- 📌 Mnemonic: Potency = Position (on X-axis, Left is more potent); Efficacy = Effect (Max Y-axis value).
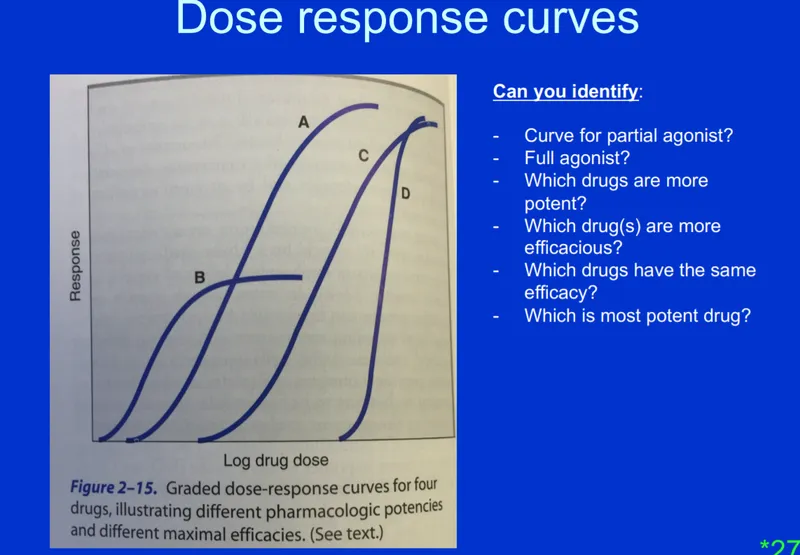
⭐ Efficacy (maximum effect) is often clinically more important than potency (amount of drug needed for an effect).
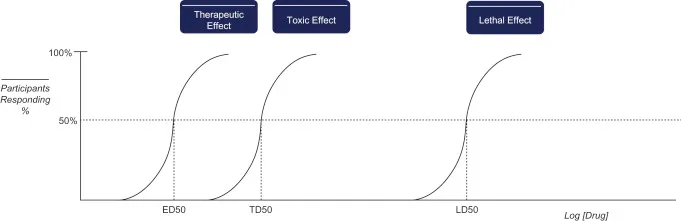
Quantal DRCs & Drug Safety - Safety Numbers Game
- Quantal DRCs: Dose vs. % population with all-or-none effect.
- Key Metrics:
- $ED_{50}$: Effective dose in 50% of population.
- $TD_{50}$: Toxic dose in 50% of population.
- $LD_{50}$: Lethal dose in 50% (animals).
- Drug Safety Indices (Margin of Safety indicators):
- Therapeutic Index ($TI$): $TI = \frac{TD_{50}}{ED_{50}}$ or $TI = \frac{LD_{50}}{ED_{50}}$.
- 📌 TILE: Toxic Is Lethal / Effective.
- Low $TI$ (narrow margin, requires monitoring): Warfarin, Lithium, Digoxin, Phenytoin, Theophylline.
- 📌 "Warning: These Drugs Can Leak Poison".
- Certain Safety Factor ($CSF$): $CSF = \frac{LD_1}{ED_{99}}$ (stricter; $LD_1$: lethal 1%, $ED_{99}$: effective 99%).
- Therapeutic Index ($TI$): $TI = \frac{TD_{50}}{ED_{50}}$ or $TI = \frac{LD_{50}}{ED_{50}}$.
⭐ A higher Therapeutic Index indicates a wider margin of safety for a drug.
Receptor Interactions & Response Variation - Receptor Roulette
- Agonists: Bind & activate receptors.
- Full: Elicits maximal response ($E_{max}$); high intrinsic activity.
- Partial: Submaximal response; lower intrinsic activity. Can act as antagonist with full agonist.
- Inverse: Opposite effect to agonist; reduces basal receptor activity.
- Antagonists: Bind receptors, block agonist. 📌 Competitive Right-shifts $EC_{50}$ ($E_{max}$ same); Non-competitive Lowers $E_{max}$ (NCLM).
Type $EC_{50}$ $E_{max}$ Overcome by ↑ Agonist? Site of Action Competitive (Reversible) ↑ (Right shift) Unchanged Yes Active Non-competitive Unchanged/↑ ↓ (Down shift) No Allosteric/Active Irreversible Unchanged/↑ ↓ (Down shift) No (covalent bond) Active ⭐ Competitive antagonists increase $EC_{50}$ (shift DRC right) but do not change $E_{max}$, while non-competitive antagonists decrease $E_{max}$ and may or may not change $EC_{50}$.
- Response Variation:
- Spare Receptors: $E_{max}$ achieved with fractional receptor occupancy (e.g., insulin).
- Tachyphylaxis: Rapid ↓ drug response on repeated dosing (acute tolerance; e.g., ephedrine).
- Tolerance: Gradual ↓ drug effectiveness with chronic use.
- Desensitization: ↓ Receptor responsiveness (e.g., downregulation, uncoupling).
- Idiosyncrasy: Genetically determined abnormal drug reaction (e.g., G6PD deficiency).
High‑Yield Points - ⚡ Biggest Takeaways
- Graded Dose-Response Curves (DRCs) relate drug dose to effect intensity.
- Efficacy (Emax) is the maximum possible effect a drug can produce.
- Potency (ED50): dose for 50% of Emax; lower ED50 means higher potency.
- Quantal DRCs show population response (all-or-none) to varying doses.
- Therapeutic Index (TI) = TD50/ED50; a wider TI indicates greater drug safety.
- Spare receptors allow Emax with submaximal receptor occupancy.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more