General Principles of Age-Related PK - Tiny Tots & Golden Oldies
- Pharmacokinetics (ADME) vary significantly at age extremes.
- Tiny Tots: Immature organs (liver, kidney); ↑Total Body Water (TBW), ↓protein binding.
- Golden Oldies: ↓Organ function (renal, hepatic); ↓TBW, ↑fat; ↓albumin.
- Impacts drug dosing, efficacy, and toxicity.

⭐ Reduced renal clearance in geriatrics is a major cause of adverse drug reactions (ADRs).
Pediatric PK: Absorption & Distribution - Sponge Bob & Skinny Jeans
Absorption:
- Gastric pH: ↑ (alkaline) in neonates (adult by 2-3 yrs).
- Gastric emptying: ↓, irregular (adult by 6-8 mo).
- Percutaneous: ↑ (thin skin, ↑hydration, ↑SA:weight). ⚠️Toxicity.
Distribution:
- TBW: ↑ (75-80%); ECF: ↑.
- ↑ Vd water-soluble drugs (gentamicin). 📌 Sponge Bob
⭐ Neonates have higher Vd for water-soluble drugs.
- Body Fat: ↓ (preterm 1%, term 15%).
- ↓ Vd lipid-soluble drugs (diazepam). 📌 Skinny Jeans
- Protein Binding: ↓ (↓albumin, ↑bilirubin) → ↑free drug.
Pediatric PK: Metabolism & Excretion - Liver & Kidney Kickstart
- Liver Metabolism (Hepatic Clearance):
- Phase I (e.g., CYP450 oxidation): Activity significantly ↓ in neonates/infants; matures by 1-2 years.
- Phase II (Conjugation):
- Glucuronidation: Markedly ↓ in neonates (matures 2-4 yrs).
- Sulfation: Relatively well-developed at birth.
- Implication: Slower metabolism → prolonged drug half-life ($t_{1/2}$), ↑ toxicity risk. Careful dosing needed.
- Kidney Excretion (Renal Clearance):
- GFR: Significantly ↓ (approx. 30-40% of adult values); matures by 6-12 months.
- Tubular Secretion: Also ↓; matures over the first year.
- Implication: Reduced renal drug elimination → prolonged $t_{1/2}$. Dose/interval adjustments critical.
⭐ Glucuronidation pathways mature late in neonates, increasing risk of Gray Baby Syndrome with chloramphenicol.
Geriatric PK: Absorption & Distribution - Senior Shifts & Stashes
- Absorption:
- ↓ Gastric acid (↑pH), ↓GI motility & blood flow.
- Rate may ↓; extent usually unchanged.
- Distribution:
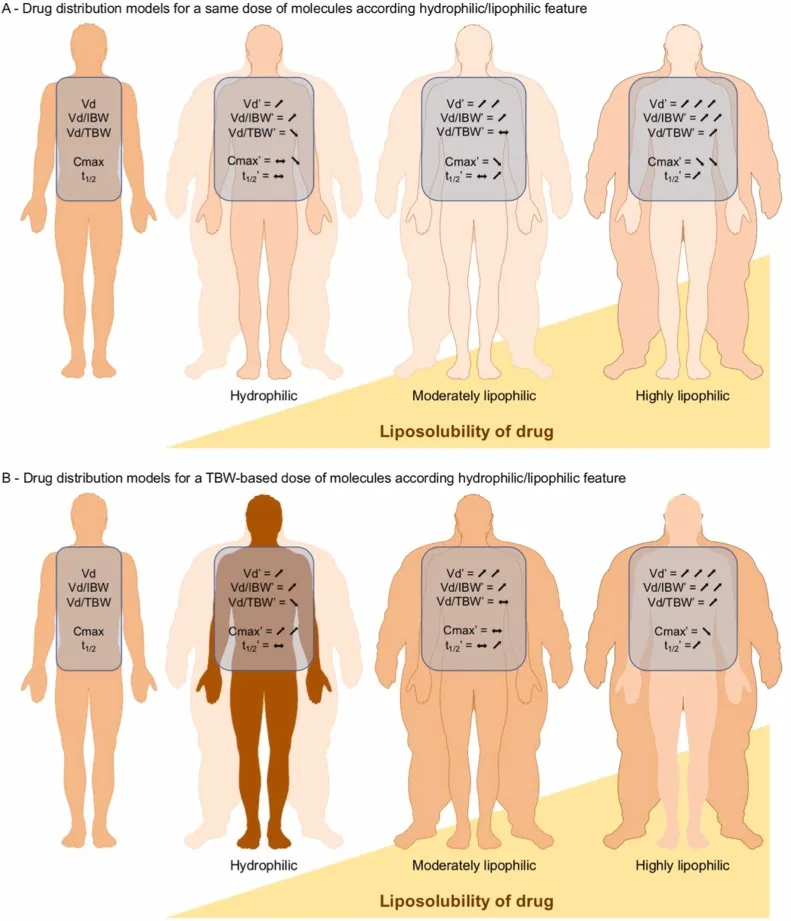
- Body Composition: ↓Total body water, ↓lean mass, ↑total body fat. (📌 Seniors Stash Fat!)
- Protein Binding: ↓Albumin (→ ↑free acidic drugs e.g. warfarin); ↑Alpha-1-acid glycoprotein (AAG).
- Volume of Distribution (Vd): ↓ for water-soluble (e.g. lithium); ↑ for lipid-soluble (e.g. diazepam).
⭐ Increased body fat in elderly means larger Vd for lipophilic drugs, prolonging effects & toxicity risk.
Geriatric PK: Metabolism & Excretion - System Slowdown Saga
- Metabolism (Liver):
- ↓ Hepatic blood flow & liver mass.
- ↓ Phase I (CYP450) reactions (oxidation, reduction) significantly.
- Phase II (conjugation) relatively preserved.
- Leads to: ↓ first-pass effect, ↑ drug $t_{1/2}$.
- Excretion (Kidney):
- ↓ Renal blood flow & GFR (by ~1% per year after 40).
- ↓ Tubular secretion.
- Leads to: ↓ drug clearance, ↑ drug accumulation.
- 📌 Estimate $CrCl$ (e.g., Cockcroft-Gault); serum creatinine unreliable alone.
⭐ Phase I (oxidative) metabolism declines more significantly than Phase II (conjugation) in the elderly.
Clinical Implications & Dosing Adjustments - Age-Wise Dosing Dilemmas
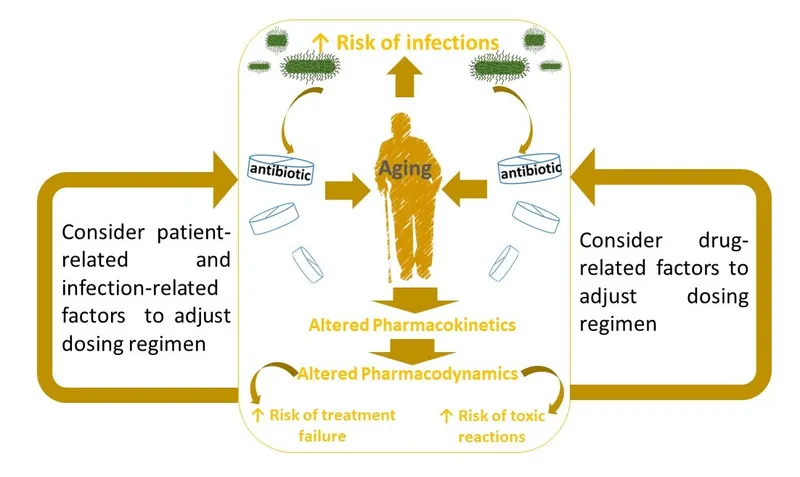
- Pediatrics: Immature organs affect drug handling; dose per kg/BSA. Monitor closely for efficacy & toxicity.
- Geriatrics: ↓ Renal function common (Cockcroft-Gault: $CrCl = \frac{(140 - Age) \times Wt (kg)}{72 \times SCr (mg/dL)} (\times 0.85 \text{ if female})$); ↑ drug sensitivity.
⭐ Polypharmacy is a major risk factor for ADRs in the elderly.
High‑Yield Points - ⚡ Biggest Takeaways
- Neonates: Higher gastric pH, slower gastric emptying, ↑ transdermal absorption.
- Pediatrics: ↑ Total Body Water (↑ Vd water-soluble drugs); ↓ protein binding (↑ free drug).
- Pediatrics: Immature liver enzymes (↓ metabolism); ↓ GFR (prolonged drug half-life).
- Geriatrics: ↓ Total Body Water, ↑ body fat (alters Vd); ↓ serum albumin (↑ free drug).
- Geriatrics: ↓ Hepatic metabolism (especially Phase I); reduced liver blood flow.
- Geriatrics: Most critical: ↓ Renal GFR leads to drug accumulation and toxicity.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

