Membrane Transport & Absorption - Getting In!
- Cell Membrane: Phospholipid bilayer, proteins; fluid mosaic model.
- Passive Transport Mechanisms:
- Simple Diffusion:
- Major route.
- Down conc. gradient; no energy.
- Favors lipid-soluble, unionized drugs.
- Fick's Law: Rate $\propto \frac{\text{Surface Area} \times (\text{C}_1-\text{C}_2) \times \text{Permeability}}{\text{Thickness}}$
- pH & Ionization: Henderson-Hasselbalch eq. for ionization.
- Acids: $pH - pKa = \log([\text{Ionized A}^-]/[\text{Unionized HA}])$
- Bases: $pKa - pH = \log([\text{Ionized BH}^+]/[\text{Unionized B}])$
- 📌 Unionized form is lipid-soluble & absorbed.
- Acidic drugs (Aspirin): ↑ unionized in acidic pH (stomach).
- Basic drugs (Morphine): ↑ unionized in alkaline pH (intestine).
- Filtration: Small water-soluble drugs (< 100-200 Da) via aqueous pores.
- Simple Diffusion:
- Carrier-Mediated Transport: Uses transporters; specific, saturable, competitive.
- Facilitated Diffusion: Down gradient, no energy (e.g., GLUT for glucose).
- Active Transport: Against gradient, needs ATP (e.g., Na+/K+ ATPase, SGLT1).
- Efflux: P-glycoprotein (MDR1) pumps out drugs, ↓ absorption.
- Vesicular Transport (Endo/Exocytosis): Large molecules (e.g., Vit B12-IF).
⭐ P-glycoprotein (MDR1), an efflux pump, significantly impacts drug absorption and bioavailability, contributing to multidrug resistance.
Routes & Bioavailability - The Entry Game
- Routes of Administration:
- Enteral: Oral (PO), Sublingual (SL), Rectal (PR).
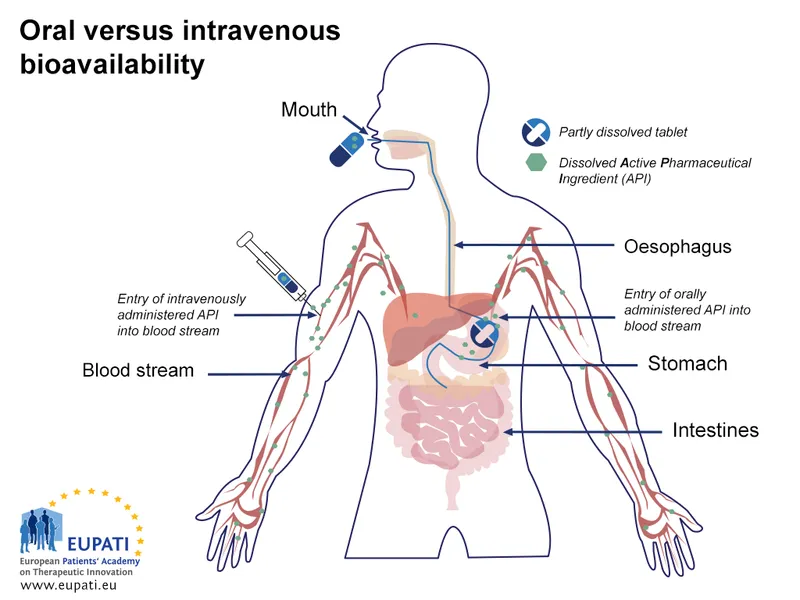
- PO: Common, convenient; subject to first-pass.
- SL/Buccal: Bypasses first-pass (Nitroglycerin); rapid.
- Rectal: Partial (≈50%) first-pass bypass; useful if N/V.
- Parenteral: IV, IM, SC.
- IV: 100% bioavailability; immediate effect.
- IM/SC: Depot possible; variable absorption.
- Inhalational: Rapid onset (anaesthetics, bronchodilators); large SA.
- Transdermal: Sustained delivery, bypasses first-pass (Fentanyl patch).
- Enteral: Oral (PO), Sublingual (SL), Rectal (PR).
- Bioavailability (F): Fraction of drug reaching systemic circulation unchanged.
- $F = (\frac{AUC_{oral}}{AUC_{IV}}) \times 100%$ (for same dose).
- IV route: $F = \textbf{100}%$.
- ↓F due to: First-pass metabolism, poor absorption, instability, formulation.
- First-Pass Metabolism: Pre-systemic metabolism (liver, gut wall) ↓F.
- Affects: Propranolol, Lignocaine, Nitroglycerin (oral).
- 📌 "LMNOP": Lignocaine, Morphine, Nitroglycerin, Oestrogens, Propranolol (high first-pass).
⭐ Drugs with high first-pass metabolism (e.g., Lignocaine) require significantly higher oral doses than IV doses to achieve comparable therapeutic effects due to extensive pre-systemic elimination.
Drug Distribution - Spreading Out!
- Drug movement: Blood → Tissues.
- Key Factors:
- Lipid Solubility: ↑ solubility → ↑ distribution.
- Ionization (pH-pKa): Non-ionized form crosses membranes.
- Plasma Protein Binding (PPB):
- Albumin (acidic drugs: warfarin), α1-acid glycoprotein (basic drugs: lidocaine).
- Only unbound drug is active & distributes. High PPB → ↓ $V_d$, longer $t_{1/2}$.
- Blood Flow/Perfusion: Brain, liver, kidney (rapid); Fat, bone (slow).
- Tissue Affinity: E.g., Iodine (thyroid), Digoxin (heart), Chloroquine (liver, retina).
- Volume of Distribution ($V_d$):
- Apparent volume drug occupies if concentration throughout body equals plasma concentration.
- Formula: $V_d = \frac{\text{Total amount of drug in body (Dose)}}{\text{Plasma drug concentration } (C_0)}$
- Low $V_d$ (< 5L): Confined to plasma (e.g., Heparin).
- Intermediate $V_d$ (15-20L): ECF (e.g., Aminoglycosides).
- High $V_d$ (> 40L): Accumulates in tissues (e.g., Chloroquine, Digoxin).
- Clinical use: Calculate Loading Dose ($LD = V_d \times C_{target}$).
- Redistribution: Highly lipid-soluble IV drugs (e.g., Thiopental) initially to brain, then to muscle/fat → rapid termination of CNS effect.
- Special Barriers:
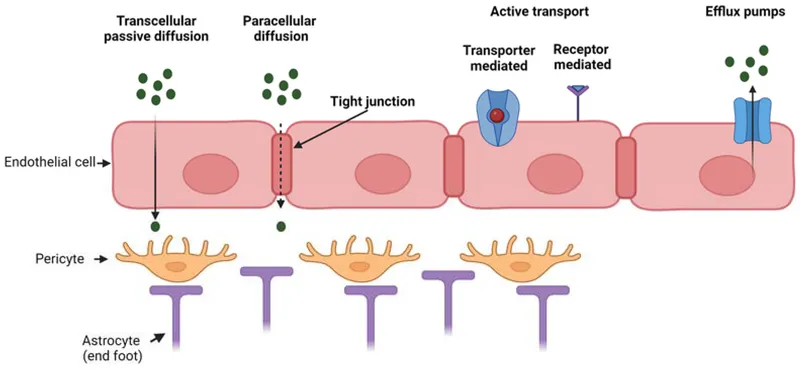
- Blood-Brain Barrier (BBB): Tight junctions; P-glycoprotein efflux. Restricts polar drugs. Lipid-soluble drugs pass.
- Placental Barrier: Most drugs cross to some extent, especially lipid-soluble.
⭐ Drugs with a very high $V_d$ (e.g., Chloroquine, Amiodarone) are not effectively removed by hemodialysis because they are extensively sequestered in tissues, away from the blood being dialyzed.
High‑Yield Points - ⚡ Biggest Takeaways
- Lipid solubility (↑) & low ionization favor drug absorption.
- First-pass metabolism (liver/gut) ↓ oral bioavailability.
- Bioavailability (F) = fraction of drug reaching systemic circulation unchanged.
- Volume of distribution (Vd) reflects tissue drug distribution; high Vd = more in tissues.
- Plasma protein binding: acidic drugs to albumin, basic drugs to α1-acid glycoprotein.
- Only unbound drug is active, distributed, metabolized, excreted.
- P-glycoprotein (MDR1): efflux pump, ↓ absorption, limits brain/placental entry_
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

