Principles of Clinical Pharmacology - Drug's Journey
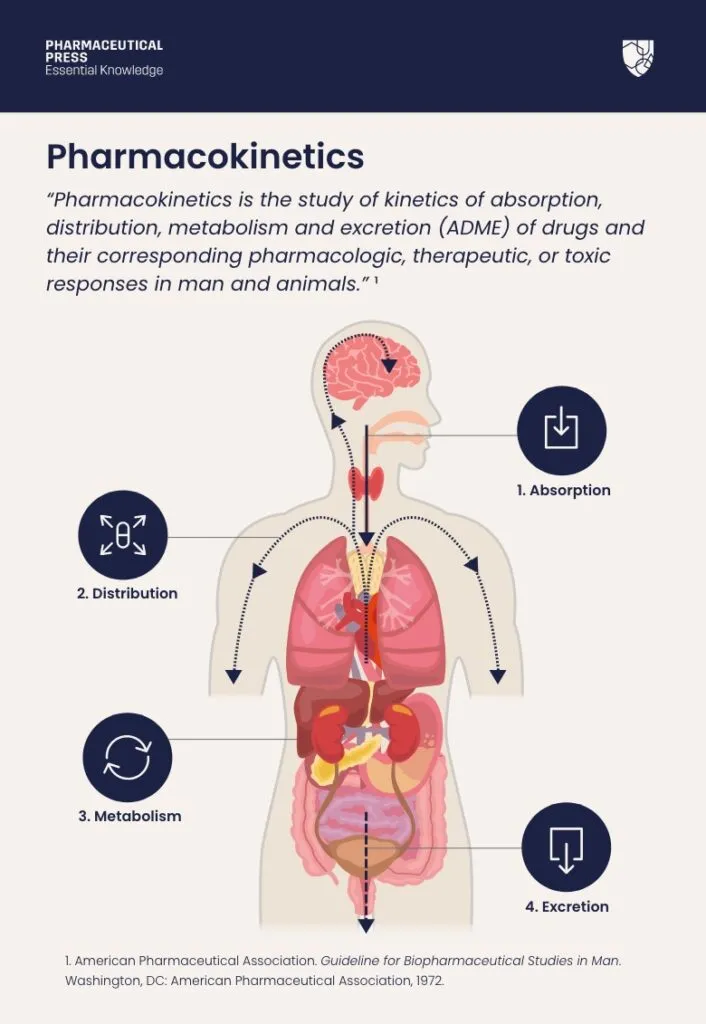
Pharmacokinetics (PK): What body does to drug (ADME).
- Absorption: Drug entry to systemic circulation.
- Bioavailability ($F$): Fraction of unchanged drug reaching systemic circulation. $F = (\text{AUC}\text{oral} / \text{AUC}\text{IV}) \times (\text{Dose}\text{IV} / \text{Dose}\text{oral})$.
- First-pass metabolism: Pre-systemic hepatic metabolism (↓$F$ oral).
- Distribution: Drug movement from blood to tissues.
- Volume of Distribution ($V_d$): Apparent volume drug occupies. $V_d = \text{Dose} / C_0$.
- Protein binding: Influences free drug.
- Metabolism: Chemical drug alteration.
- Phase I: Oxidation, reduction, hydrolysis (CYP450).
- Phase II: Conjugation (glucuronidation).
- Excretion: Drug removal.
- Routes: Renal, hepatic.
- Clearance ($Cl$): Plasma volume cleared per unit time. $Cl = k \times V_d$.
- Half-life ($t_{1/2}$): Time for drug conc. to halve. $t_{1/2} = (0.693 \times V_d) / Cl$.
⭐ Zero-order kinetics drugs: Warfarin, Aspirin, Phenytoin, Ethanol, Theophylline - 📌 WAP-ET Zero.
Principles of Clinical Pharmacology - Drug's Power Play
- Pharmacodynamics (PD): Drug's action on body; mechanisms.
- Receptors: Cellular targets for drugs.
- Types: Ion channels, GPCRs, Enzyme-linked, Intracellular.
- Drug-Receptor Interactions:
- Agonist: Activates receptor (Full, Partial, Inverse).
- Antagonist: Blocks agonist action (Competitive, Non-competitive).
- Dose-Response Curve (DRC): Relates dose to effect.
- Graded: Effect intensity vs. dose.
- Efficacy ($E_{max}$): Maximum possible effect.
- Potency ($ED_{50}$): Dose for 50% of $E_{max}$.
- Quantal: All-or-none response (e.g., $ED_{50}$, $LD_{50}$).
- Therapeutic Index (TI): $TI = TD_{50} / ED_{50}$; drug safety margin.
- Graded: Effect intensity vs. dose.
⭐ Spare receptors increase sensitivity to agonists, allowing maximal response without occupying all receptors.
- Key Concepts:
- Tolerance: Decreased drug effect with repeated use.
- Tachyphylaxis: Rapidly diminishing response.
- Synergism: Enhanced combined effect (1+1 > 2).
and potency (ED50))
Principles of Clinical Pharmacology - It Depends!
Drug response varies significantly due to multiple factors, necessitating individualized therapy.
- Age:
- Pediatrics: Immature organ function (e.g., Gray baby syndrome - chloramphenicol).
- Geriatrics: ↓ renal/hepatic function, polypharmacy, ↑ sensitivity (e.g., benzodiazepines).
- Pharmacogenomics: Genetic variants impact drug metabolism/response.
- E.g., CYP2D6 (codeine), TPMT (azathioprine), NAT2 (isoniazid).
- Disease States:
- Renal/Hepatic dysfunction: Requires dose adjustments (e.g., ↓ digoxin in renal failure).
- Drug Interactions: One drug alters another's effect.
Type Mechanism Example Pharmacokinetic Altered ADME (e.g., CYP enzyme activity) Rifampicin (inducer) ↓ warfarin effect Pharmacodynamic Additive/Opposing effects at target Beta-blockers + Verapamil → severe bradycardia
⭐ Grapefruit juice is a potent CYP3A4 inhibitor, significantly increasing levels of drugs like statins and calcium channel blockers.
Principles of Clinical Pharmacology - Safety First
- Adverse Drug Reactions (ADRs): Harmful drug effects.
- 📌 Mnemonic: Augmented, Bizarre, Chronic, Delayed, End-of-use, Failure.
- Types:
Type Description Example A Augmented, dose-dependent Insulin hypoglycemia B Bizarre, non-dose-dependent Penicillin allergy C Chronic, dose & time-related NSAID nephropathy D Delayed, time-related Carcinogenesis E End-of-use, withdrawal Opioid withdrawal F Failure of therapy Antibiotic resistance
⭐ Type B (Bizarre) ADRs are unpredictable, not dose-dependent, and often immune-mediated (e.g., penicillin allergy).
- Pharmacovigilance (PV): ADR detection, assessment, understanding, prevention. Report to PvPI.
- Therapeutic Drug Monitoring (TDM): Optimizes dose via drug levels. For NTI drugs (Digoxin, Lithium, Phenytoin).
- Rational Drug Use (RDU): Right drug, dose, duration, cost.
High‑Yield Points - ⚡ Biggest Takeaways
- Pharmacokinetics (ADME) defines drug journey; Pharmacodynamics its action and effects.
- Bioavailability (F): fraction reaching circulation; reduced by first-pass metabolism.
- Half-life (t½) determines dosing frequency and steady state achievement (typically 4-5 half-lives).
- Therapeutic Index (TI): crucial for safety; narrow TI drugs demand close monitoring.
- Volume of Distribution (Vd) indicates tissue penetration; Clearance (CL) reflects elimination efficiency.
- Drug action involves receptors: agonists activate, antagonists inhibit.
- Dose-response relationships quantify drug efficacy and potency.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

