Opioid Receptors & Classification - Pain's Off-Switches
- Receptors: Mu (μ), Kappa (κ), Delta (δ) - G-protein coupled receptors (GPCRs) in CNS & periphery.
- Mechanism: ↓cAMP, ↑K+ efflux, ↓Ca2+ influx → neuronal inhibition.
- Endogenous Opioids: Endorphins (μ-affinity), Enkephalins (δ-affinity), Dynorphins (κ-affinity).
- Drug Classes (Examples):
- Agonists (e.g., Morphine, Fentanyl)
- Partial Agonists (e.g., Buprenorphine)
- Mixed Agonist-Antagonists (e.g., Pentazocine)
- Antagonists (e.g., Naloxone, Naltrexone)

⭐ Endogenous opioids like endorphins, enkephalins, and dynorphins are naturally occurring peptides that modulate pain and are targeted by opioid analgesics.
Opioid MOA & Pharmacokinetics - Body's Opioid Journey
- Mechanism of Action (MOA):
- Receptor binding: μ, κ, δ GPCRs.
- Presynaptic effects: ↓ Ca²⁺ influx → ↓ neurotransmitter release.
- Postsynaptic effects: ↑ K⁺ efflux → hyperpolarization → ↓ excitability.
⭐ Opioids exert their analgesic effect by binding to G-protein coupled receptors (GPCRs), primarily μ (mu), κ (kappa), and δ (delta), leading to decreased presynaptic Ca²⁺ influx and increased postsynaptic K⁺ efflux.
- Pharmacokinetics (ADME):
- Absorption: Variable; significant first-pass metabolism (e.g., Morphine).
- Distribution: Lipid solubility dictates CNS entry (Fentanyl > Morphine).
- Metabolism: Hepatic (CYP450, glucuronidation). Codeine → Morphine (CYP2D6).
- Excretion: Renal.
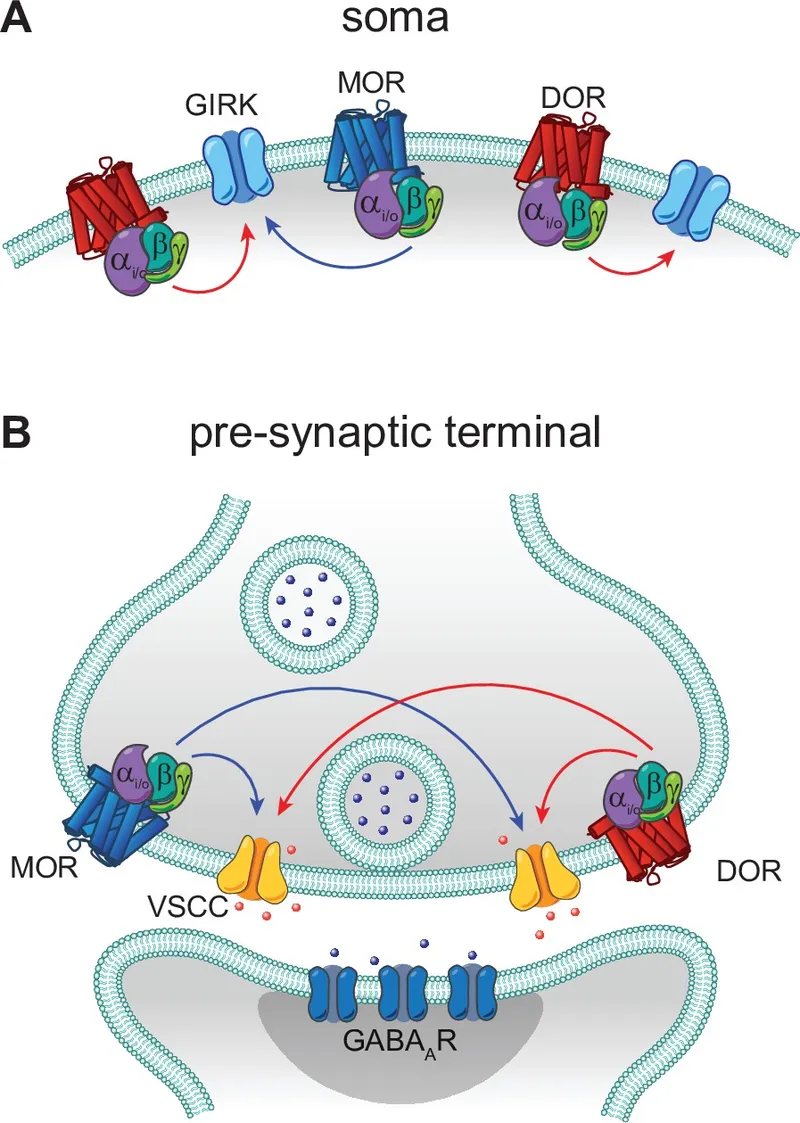
Key Opioid Agonists - The Relief Squad
- Morphine (Prototype µ-agonist):
- Strong analgesia (severe pain e.g. MI, cancer).
- AEs: Resp. depression, miosis, constipation, histamine.
- ⚠️ Avoid: Head injury, asthma.
- Pethidine (Meperidine):
- Synthetic µ-agonist; shorter action. Atropine-like (mydriasis).
- Norpethidine metabolite → seizures in renal failure.
⭐ Pethidine (Meperidine) is an opioid agonist that, unlike morphine, has atropine-like effects, may cause less biliary spasm, and its metabolite norpethidine can accumulate in renal failure leading to seizures.
- Fentanyl & congeners:
- Potent (~100x M); rapid onset, short duration.
- Uses: Anesthesia, pain patches. Risk: chest rigidity.
- Methadone:
- Long-acting µ-agonist; opioid dependence, chronic pain.
- Risk: QT prolongation.
- Codeine:
- Weak µ-agonist (→morphine by CYP2D6); antitussive.
- Tramadol:
- Weak µ-agonist + SNRI; moderate pain.
- Risk: Seizures, serotonin syndrome.
- Tapentadol:
- µ-agonist + NRI; less GI AEs.
- Buprenorphine:
- Partial µ-agonist, κ-antagonist; resp. depression ceiling.
- Uses: Dependence, pain. Can precipitate withdrawal.
Opioid ADRs & Toxicity - The Dark Side
- Common ADRs: Constipation (most persistent), N/V, sedation, dizziness, pruritus, urinary retention.
- Respiratory depression: Dose-dependent; most serious acute ADR.
- Tolerance: ↓ effect with repeated use.
- Dependence: Withdrawal syndrome on cessation.
- 📌 Mnemonic for opioid toxicity triad: CPR - Coma, Pinpoint pupils, Respiratory depression.
- Acute Toxicity:
- Triad: Coma, pinpoint pupils (miosis), respiratory depression.
- Management: Airway support, Naloxone (0.4-2 mg IV/IM/SC).
⭐ The classic triad of acute opioid poisoning is coma, pinpoint pupils (miosis), and respiratory depression; naloxone is the specific antidote.
Opioid Antagonists & Clinical Use - Counter & Cautions
- Naloxone: Reverses OD (respiratory depression). Short t½ (1-2h).
- Naltrexone: Oral, long-acting. For opioid/alcohol dependence.
- PAMORAs (Methylnaltrexone): For OIC, peripheral.
- OD Management:
- ⚠️ Cautions: Acute withdrawal; Naloxone: renarcotization; Naltrexone: hepatotoxic.
⭐ Naloxone is a pure opioid antagonist with a rapid onset and short duration of action (1-2 hours), primarily used to reverse opioid-induced respiratory depression in overdose situations.
High‑Yield Points - ⚡ Biggest Takeaways
- Morphine: Prototype μ-agonist; causes analgesia, euphoria, respiratory depression, miosis, constipation.
- Codeine: Weak opioid, metabolized to morphine by CYP2D6; effective antitussive.
- Fentanyl: Highly potent, rapid-acting μ-agonist; used in anesthesia, breakthrough pain.
- Tramadol: Dual action (weak μ-agonist, SNRI); risk of seizures, serotonin syndrome.
- Naloxone: Pure antagonist for acute opioid overdose reversal; short duration of action.
- Naltrexone: Orally active, long-acting antagonist for opioid and alcohol dependence treatment.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

