Oncogenes & Growth Signals - Go-Go Growth Genes
- Oncogenes: Mutated proto-oncogenes; drive uncontrolled cell growth (gain-of-function). "Accelerator ON".
- Activation:
- Point mutation: RAS (KRAS, HRAS, NRAS).
- Amplification: HER2/neu (breast), NMYC (neuroblastoma).
- Translocation: BCR-ABL (CML), MYC-IgH (Burkitt's).
- Pathway Hijack:
- Key Examples by Class:
- Receptors: EGFR (lung adeno); HER2/neu (ERBB2) (breast, ovary, gastric ca). 📌 "HER 2 much growth".
- Transducers: RAS (pancreas, colon, lung); BRAF (melanoma); ABL (CML).
- Nuclear Factors: MYC (Burkitt's, neuroblastoma); drives growth, angiogenesis.
⭐ RAS mutations (KRAS, HRAS, NRAS) affect ~15-20% of human cancers; a key oncogene family.
Tumor Suppressors & Genome Guardians - Stop Signs & Fix-It Crew
- Function: Genes that normally inhibit cell proliferation or promote DNA repair. Loss of function (LOF) → cancer.
- Knudson's "two-hit" hypothesis: both alleles inactivated for cancer development.
- Key Examples:
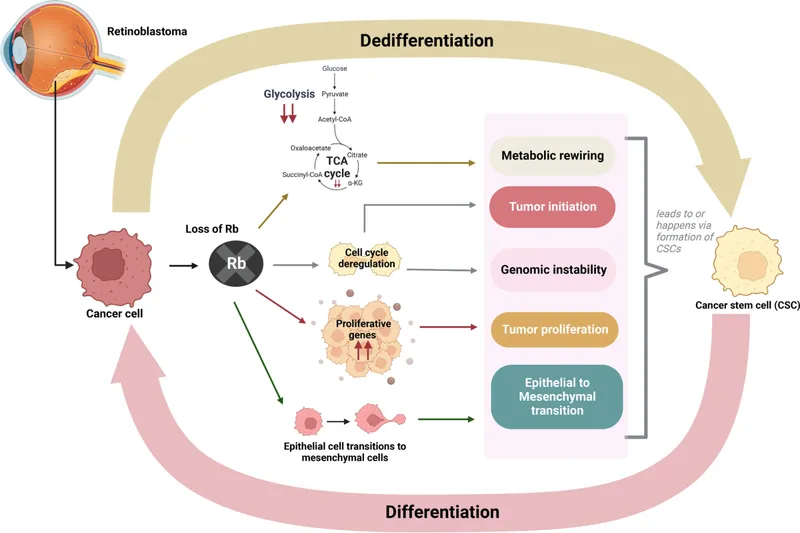
- RB (Retinoblastoma): "Governor". Controls G1/S checkpoint by binding E2F.
- Inactive when hyperphosphorylated by CyclinD-CDK4/6.
- Associated: Retinoblastoma, osteosarcoma.
- TP53 (p53): "Guardian". Responds to DNA damage.
- Actions: Cell cycle arrest (via p21), DNA repair, Apoptosis (via BAX).
- Li-Fraumeni syndrome (germline).
- RB (Retinoblastoma): "Governor". Controls G1/S checkpoint by binding E2F.
- **APC**: Degrades β-catenin (WNT pathway). Familial Adenomatous Polyposis (FAP).
- **BRCA1/BRCA2**: DNA repair (homologous recombination). ↑Breast, ovarian cancer risk.
- **CDKN2A (p16<sup>INK4a</sup>)**: Inhibits CDK4/6 → keeps RB active.
⭐ TP53 is mutated in >50% of human cancers, leading to genomic instability.
Epigenetics & Non-Coding RNAs - Silent Gene Tweaks
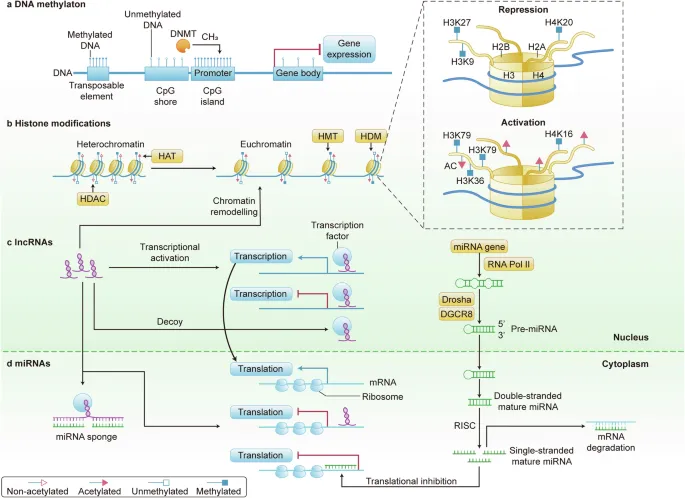
- Epigenetics: Heritable gene expression changes without DNA sequence alteration.
- DNA Methylation:
- Promoter hypermethylation → silences Tumor Suppressor Genes (TSGs) (e.g., CDKN2A (p16), MLH1).
- Global hypomethylation → activates oncogenes.
- Histone Modification: Acetylation (↑activity), deacetylation (↓activity). Dysregulated 'histone code'.
- DNA Methylation:
- Non-Coding RNAs (ncRNAs): Regulatory RNAs; not translated.
- MicroRNAs (miRNAs): ~22 nt. Post-transcriptional gene silencing.
- OncomiRs (promote cancer): e.g., miR-21, miR-17-92 cluster.
- Tumor suppressor miRNAs: e.g., let-7, miR-34a.
- Long Non-Coding RNAs (lncRNAs): >200 nt. Diverse roles. E.g., HOTAIR, MALAT1.
- MicroRNAs (miRNAs): ~22 nt. Post-transcriptional gene silencing.
⭐ Promoter hypermethylation of key tumor suppressor genes (e.g., CDKN2A (p16), MLH1, BRCA1) is a common epigenetic event driving carcinogenesis.
Immortality, Angiogenesis, Metastasis - Advanced Aggression
- Immortality (Limitless Replication):
- Telomerase (hTERT) activation: prevents telomere shortening.
- Evasion of apoptosis: e.g., p53 mutations, ↑BCL-2 family proteins.
- Bypass senescence signals.
- Angiogenesis (New Blood Vessel Formation):
- "Angiogenic switch": balance shifts to pro-angiogenic factors (VEGF, bFGF).
- Hypoxia (via HIF-1α) is a potent inducer.
- Tumor vessels are often abnormal: leaky, tortuous.

- Metastasis (Distant Spread):
- Sequential steps: local invasion → intravasation → systemic transport → extravasation → colonization.
- Epithelial-Mesenchymal Transition (EMT): crucial for motility; ↓E-cadherin, ↑N-cadherin, ↑vimentin.
- Matrix Metalloproteinases (MMPs) degrade ExtraCellular Matrix (ECM).
⭐ The "cadherin switch" (loss of E-cadherin, gain of N-cadherin) is a hallmark of EMT, facilitating tumor cell invasion.
High‑Yield Points - ⚡ Biggest Takeaways
- Proto-oncogenes (e.g., RAS, MYC) and Tumor Suppressor Genes (e.g., TP53, RB) are critical.
- TP53 ("guardian of the genome") is the most commonly mutated gene, vital for apoptosis.
- RB gene controls the G1/S checkpoint; its loss drives proliferation.
- RAS mutations cause constitutive activation of growth signaling pathways.
- BRCA1/BRCA2 defects impair DNA repair, predisposing to breast and ovarian cancers.
- Telomerase reactivation enables cancer cell immortality_._
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more